Video capsule endoscopy and balloon-assisted enteroscopy are complementary diagnostic methods in the study of small bowel bleeding, and different factors can affect their diagnostic yield.
AimsTo define the level of agreement between video capsule endoscopy and enteroscopy in small bowel bleeding, according to the type of lesion, in a cohort of patients at a tertiary care referral center.
Materials and methodsA retrospective study was conducted that included 428 capsule endoscopies performed within the time frame of 2011 and 2019 at our healthcare institution. Seventy-four video capsule endoscopies, followed by enteroscopy, in 71 patients suspected of presenting with small bowel bleeding, were analyzed.
ResultsMean patient age was 63.9 ± 13.5 years and 42 patients were women. The two diagnostic procedures were performed. Overall diagnostic yield of positive findings between video capsule endoscopy and enteroscopy was 86.5% vs. 58.1%, respectively (p = 0.0527). Agreement between video capsule endoscopy and enteroscopy for positive pathologic findings was weak (Ik = 0.17, 95% CI: -0.0097-0.3543), but according to lesion type, it was good for inflammatory lesions (Ik = 0.71, 95% CI: 0.5182-0.9119) and moderate for angiectasias (Ik = 0.45, 95% CI: 0.2469-0.6538) and tumors (Ik = 0.40, 95% CI: 0.1217-0.6794). The results between the two methods differed in 38 patients (51.3%). There was complete intestinal capsule retention in one patient (1.4%) and active bleeding in 13 (17.6%).
ConclusionsThe present study showed that the two techniques had a similar overall detection rate for small bowel lesions, but the type of lesion was the main factor that could modify diagnostic agreement.
La videocápsula endoscópica (VCE) y la enteroscopia asistida por balón son métodos diagnósticos complementarios en el estudio de la hemorragia de intestino delgado, donde diversos factores pueden afectar su rendimiento diagnóstico.
ObjetivosDefinir el grado de concordancia entre la VCE y la enteroscopia en la hemorragia de intestino delgado según el tipo de lesión, en una cohorte de pacientes de un centro referencia de alta complejidad. Materiales y Métodos: estudio retrospectivo; 428 VCE se realizaron entre el año 2011 a 2019 en la institución. Se analizaron total de 74 VCE con posterior realización de enteroscopia en 71 pacientes con sospecha de hemorragia de intestino delgado.
ResultadosEdad media: 63,9 ± 13,5 años, 42 mujeres; realizamos ambos procedimientos diagnósticos. El rendimiento diagnóstico global de hallazgos positivos entre VCE y enteroscopia fue (86.5% vs. 58.1%, respectivamente p = 0.0527), la concordancia entre VCE y enteroscopia para hallazgos positivos patológicos fue débil Ik = 0,17 (IC 95% -0.0097-0.3543); sin embargo, según el tipo de lesión fue buena para lesiones inflamatorias Ik = 0,71 (IC 95% 0.5182-0.9119), moderada para angiectasias Ik = 0,45 (IC 95% 0.2469-0.6538) y tumores Ik = 0,40 (IC 95% 0.1217-0.6794). Los resultados entre ambos estudios difirieron en 38 pacientes (51.3%). Retención completa intestinal de la VCE en 1 paciente (1.4%). Hemorragia activa se evidencio en 13 pacientes (17.6%).
ConclusionesEl presente estudio evidencia que la tasa de detección global de lesiones del intestino delgado de ambas técnicas es similar; sin embargo, el principal factor que puede modificar la concordancia diagnóstica es el tipo de lesión.
The main indication for video capsule endoscopy (VCE) is small bowel bleeding in patients with previous negative upper gastrointestinal endoscopy and colonoscopy. The utility of enteroscopy has been shown to be similar to that of VCE in small bowel bleeding, albeit false negatives have been described with both techniques, depending on the type of lesion.1 The diagnostic yield of VCE in the context of small bowel bleeding is reported in different studies to be between 32-83%2,3 and said procedure helps define the insertion route for enteroscopy.4 The majority of studies on the agreement between the two techniques in relation to the different types of lesions are small case series with scant scientific evidence. There are few meta-analyses that comparatively analyze their diagnostic yield. Enteroscopy has shown a better diagnostic yield (68.2%) when performed after VCE, compared with enteroscopy alone (43.8%), suggesting that the two modalities are complementary.5 Enteroscopy is indicated when VCE results in positive findings that require intervention or diagnostic confirmation.6
The aim of the present study was to define the levels of agreement between VCE and enteroscopy, according to the type of small bowel lesion and the findings from a prior VCE performed to study small bowel bleeding, in a cohort of patients from a tertiary referral hospital.
Materials and methodsAll the patients suspected of presenting with small bowel bleeding that had undergone VCE and balloon-assisted enteroscopy at the Hospital Universitario San Ignacio, within the time frame of June 2011 to June 2019, were included in the study. Small bowel bleeding was defined in accordance with the definition of the American College of Gastroenterology,7 and the type of bleeding (overt or occult) was considered. All the patients underwent upper gastrointestinal endoscopy and colonoscopy, as well as other imaging techniques, with no significant findings to explain the bleeding. Patients under 18 years of age and those that did not undergo VCE prior to the enteroscopy were excluded from the study.
DefinitionsPositive pathologic findings in the VCE and enteroscopy were considered when lesions were detected that could explain the bleeding and were reported in both studies at the same anatomic site. Those findings were classified as groups of inflammatory lesions (defined by the presence of ulcerated enteritis), angiectasias, and tumors, as potential causes of bleeding.
Procedures: VCE and single or double balloon enteroscopyVCE for small bowel study (Given Imaging Ltd, Yoqueam, Israel) was performed with the PillCam SB2 and SB3, in the majority of cases, and with the EndoCapsule, (Olympus, Japan) in a smaller number of cases at the beginning of the study. The patients underwent the 6 -h fasting protocol and bowel preparation with two packets of polyethylene glycol. The capsule was swallowed with water. Some patients required its endoscopic advancement. Complete small bowel examination was defined as a VCE reaching the cecum with the adequate preparation. VCEs performed prior to enteroscopy were taken into account. Complete retention was established by a period greater than 14 days. All readings were viewed by three experienced gastroenterologists and the reading speed was increased or decreased, according to physician criteria.
Antegrade and/or retrograde enteroscopy was performed under general anesthesia, with a single and/or double balloon. For the antegrade approach, a 12 h fast was carried out, and for the retrograde approach, bowel preparation with four packets of polyethylene glycol was utilized. Total enteroscopy was defined as the complete examination of the small bowel through the combination of the two routes. Single balloon enteroscopy was performed with SIF-Q180 equipment (Olympus, Japan) and double balloon enteroscopy with EN-450P5 and EN-450T5 equipment (Fuji Film, Japan). The approach, lesion location, and procedure time were considered. The approach was selected according to the lesion site documented in the VCE, determining the percentage of VCE transit time. If the lesion was found within 60% of the transit time from the pylorus to the ileocecal valve, access was antegrade, and if it was found in the remaining 40% of transit time, access was retrograde.
Statistical analysisThe descriptive analysis of the qualitative variables was carried out through absolute and relative frequencies. The quantitative variables were described using the mean (X¯) or median (Me) measures of central tendency, with their respective dispersion measures of standard deviation (SD) and interquartile range (IQR), according to the normality criteria calculated by the Kolmogorov-Smirnov test. The Cohen’s kappa statistic was calculated to estimate the agreement between the positive findings, tumor, angiectasias, and inflammatory lesions identified through VCE and enteroscopy. Ninety-five percent confidence intervals were calculated in all the kappa indexes and agreement strength was indicated according to the kappa values as poor (< 0), weak (0-0.2), low (0.21-0.40), moderate (0.41-0.60), good (0.61-0.80), and very good (0.81-1.00). Statistical significance was set at a p < 0.05, with a 95% confidence interval. The STATA 12.1 (Data Analysis and Statistical Software, USA) software was employed.
Ethical considerationsThe project was approved by the ethics committees of the Hospital Universitario San Ignacio and the Pontificia Universidad Javeriana. Informed consent was obtained from all the participants for the performance of procedures, maintaining the principles of privacy and confidentiality regarding patient identification.
ResultsWithin the time frame of June 2011 to June 2019, 428 VCEs and 313 enteroscopies (201 antegrade and 112 retrograde) were performed at the gastroenterology unit of the Hospital Universitario San Ignacio. Seventy-four (23.6%) VCEs were followed by single balloon or double balloon enteroscopy with antegrade and/or retrograde access in 71 patients suspected of presenting with small bowel bleeding. The mean time between the two procedures was 19.5 (IQR 5.0-94) days.
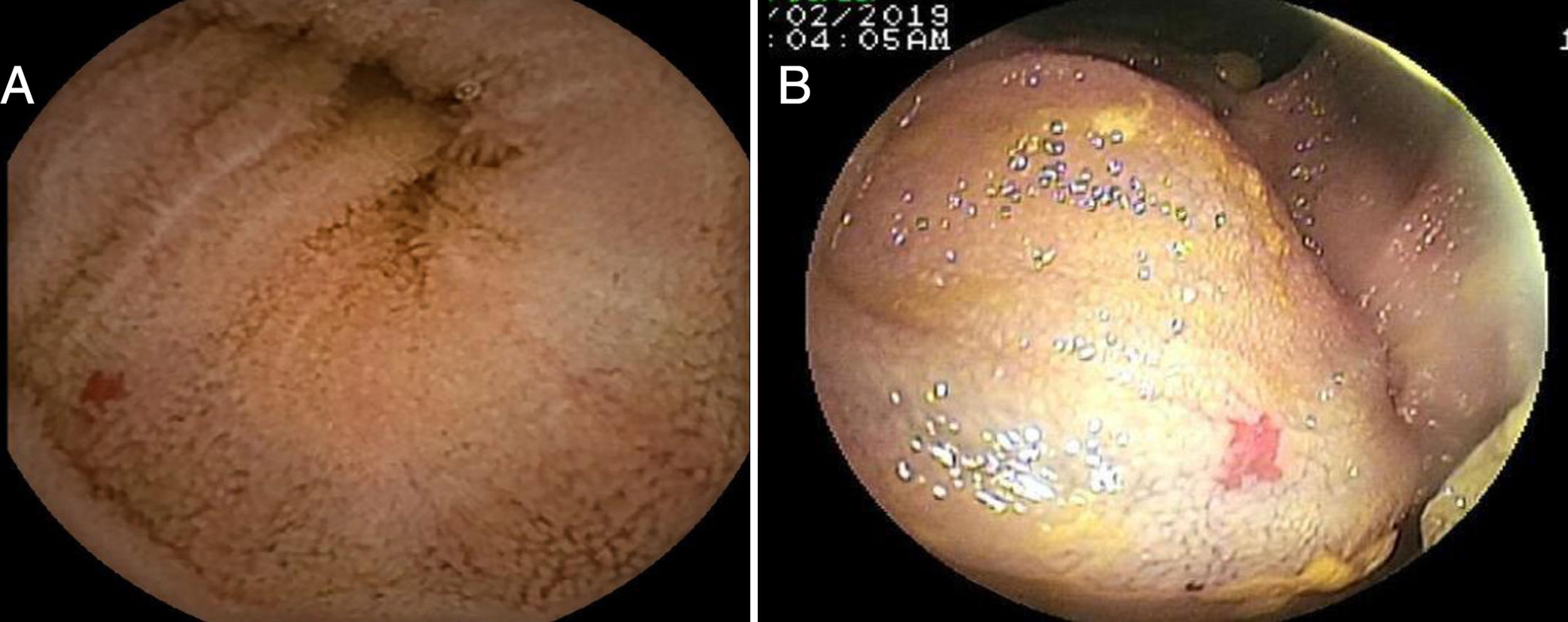
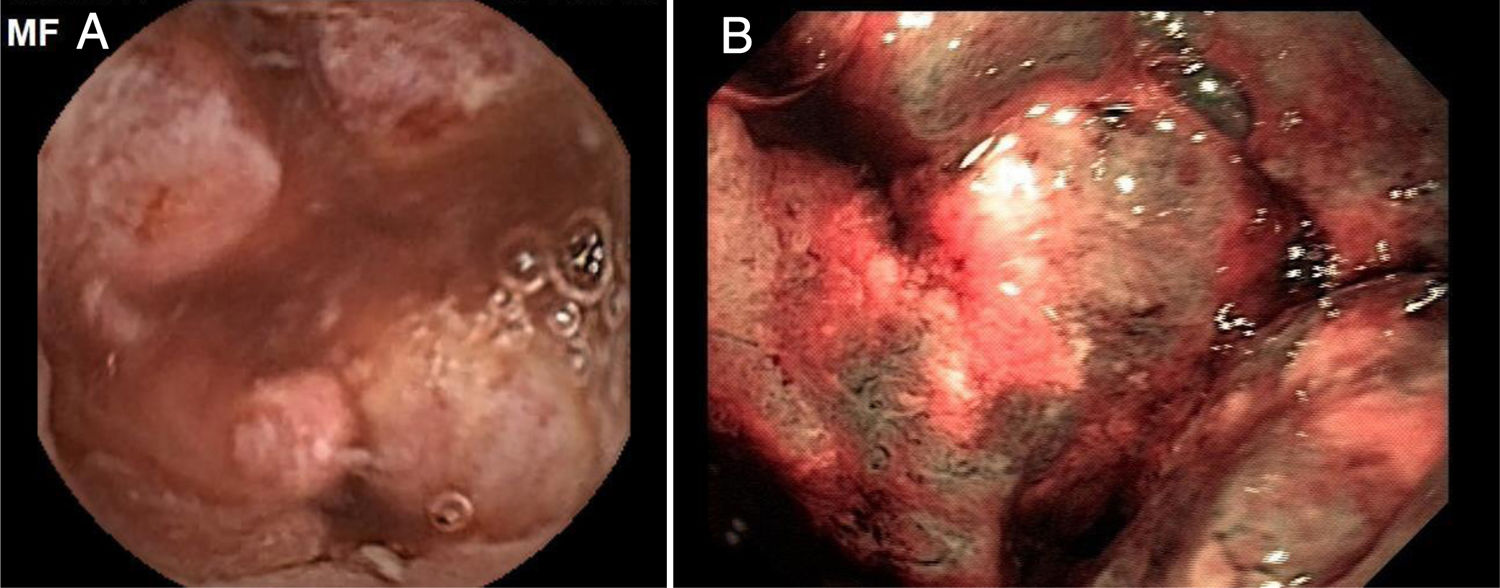
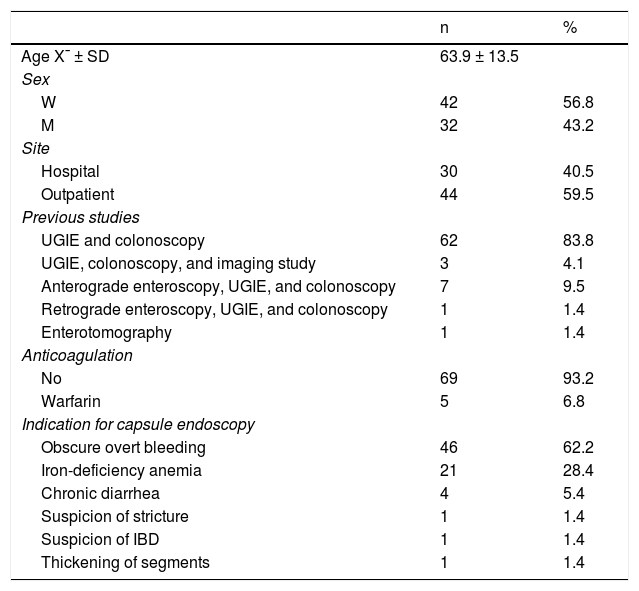
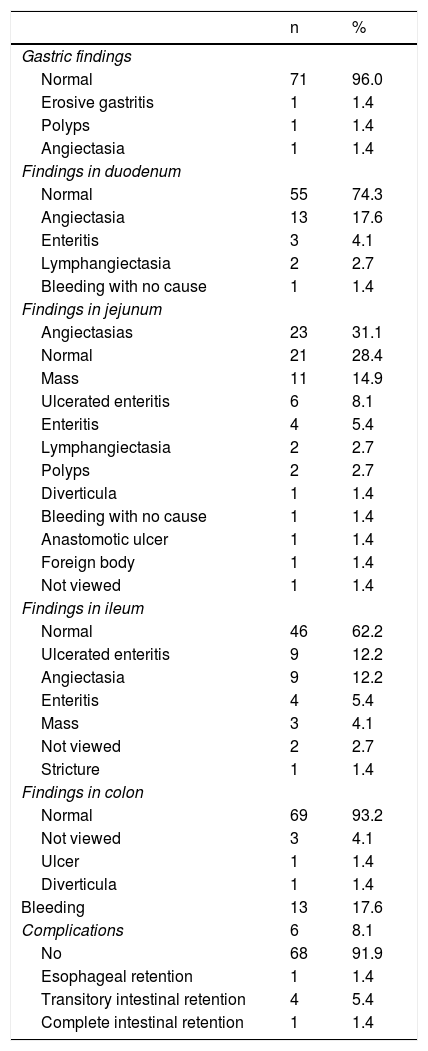
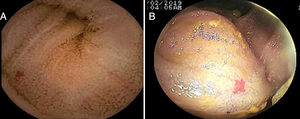
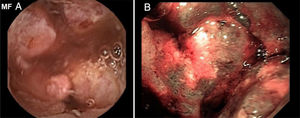
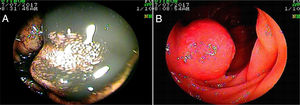
Mean patient age was 63.9 ± 13.5 years, with a predominance of women (n = 42) (56.8%). The main indications for VCE were obscure overt gastrointestinal bleeding in 46 (62.2%) patients and iron-deficiency anemia in 21 (28.4%) patients (Table 1). Angiectasias presented mainly in the jejunum in 23 (31.1%) patients (Fig. 1), followed by the duodenum in 13 (17.6%) patients, the ileum in 9 (12.2%) patients, and gastric angiectasias in one (1.4%) patient. The tumor was located in the jejunum in 11 (14.9%) patients and in the ileum in 3 (4.1%) patients (Figs. 2 and 3). The majority of the inflammatory lesions were in the ileum, with a total of 13 patients (17.6%). VCE complications presented in 6 (8.1%) patients (Table 2).
General characteristics, management site, previous studies, anticoagulation use, and indication for VCE.
| n | % | |
|---|---|---|
| Age X¯ ± SD | 63.9 ± 13.5 | |
| Sex | ||
| W | 42 | 56.8 |
| M | 32 | 43.2 |
| Site | ||
| Hospital | 30 | 40.5 |
| Outpatient | 44 | 59.5 |
| Previous studies | ||
| UGIE and colonoscopy | 62 | 83.8 |
| UGIE, colonoscopy, and imaging study | 3 | 4.1 |
| Anterograde enteroscopy, UGIE, and colonoscopy | 7 | 9.5 |
| Retrograde enteroscopy, UGIE, and colonoscopy | 1 | 1.4 |
| Enterotomography | 1 | 1.4 |
| Anticoagulation | ||
| No | 69 | 93.2 |
| Warfarin | 5 | 6.8 |
| Indication for capsule endoscopy | ||
| Obscure overt bleeding | 46 | 62.2 |
| Iron-deficiency anemia | 21 | 28.4 |
| Chronic diarrhea | 4 | 5.4 |
| Suspicion of stricture | 1 | 1.4 |
| Suspicion of IBD | 1 | 1.4 |
| Thickening of segments | 1 | 1.4 |
IBD: inflammatory bowel disease; SD: standard deviation; UGIE: upper gastrointestinal endoscopy; VCE: video capsule endoscopy.
VCE findings by anatomic section, bleeding, and complications.
| n | % | |
|---|---|---|
| Gastric findings | ||
| Normal | 71 | 96.0 |
| Erosive gastritis | 1 | 1.4 |
| Polyps | 1 | 1.4 |
| Angiectasia | 1 | 1.4 |
| Findings in duodenum | ||
| Normal | 55 | 74.3 |
| Angiectasia | 13 | 17.6 |
| Enteritis | 3 | 4.1 |
| Lymphangiectasia | 2 | 2.7 |
| Bleeding with no cause | 1 | 1.4 |
| Findings in jejunum | ||
| Angiectasias | 23 | 31.1 |
| Normal | 21 | 28.4 |
| Mass | 11 | 14.9 |
| Ulcerated enteritis | 6 | 8.1 |
| Enteritis | 4 | 5.4 |
| Lymphangiectasia | 2 | 2.7 |
| Polyps | 2 | 2.7 |
| Diverticula | 1 | 1.4 |
| Bleeding with no cause | 1 | 1.4 |
| Anastomotic ulcer | 1 | 1.4 |
| Foreign body | 1 | 1.4 |
| Not viewed | 1 | 1.4 |
| Findings in ileum | ||
| Normal | 46 | 62.2 |
| Ulcerated enteritis | 9 | 12.2 |
| Angiectasia | 9 | 12.2 |
| Enteritis | 4 | 5.4 |
| Mass | 3 | 4.1 |
| Not viewed | 2 | 2.7 |
| Stricture | 1 | 1.4 |
| Findings in colon | ||
| Normal | 69 | 93.2 |
| Not viewed | 3 | 4.1 |
| Ulcer | 1 | 1.4 |
| Diverticula | 1 | 1.4 |
| Bleeding | 13 | 17.6 |
| Complications | 6 | 8.1 |
| No | 68 | 91.9 |
| Esophageal retention | 1 | 1.4 |
| Transitory intestinal retention | 4 | 5.4 |
| Complete intestinal retention | 1 | 1.4 |
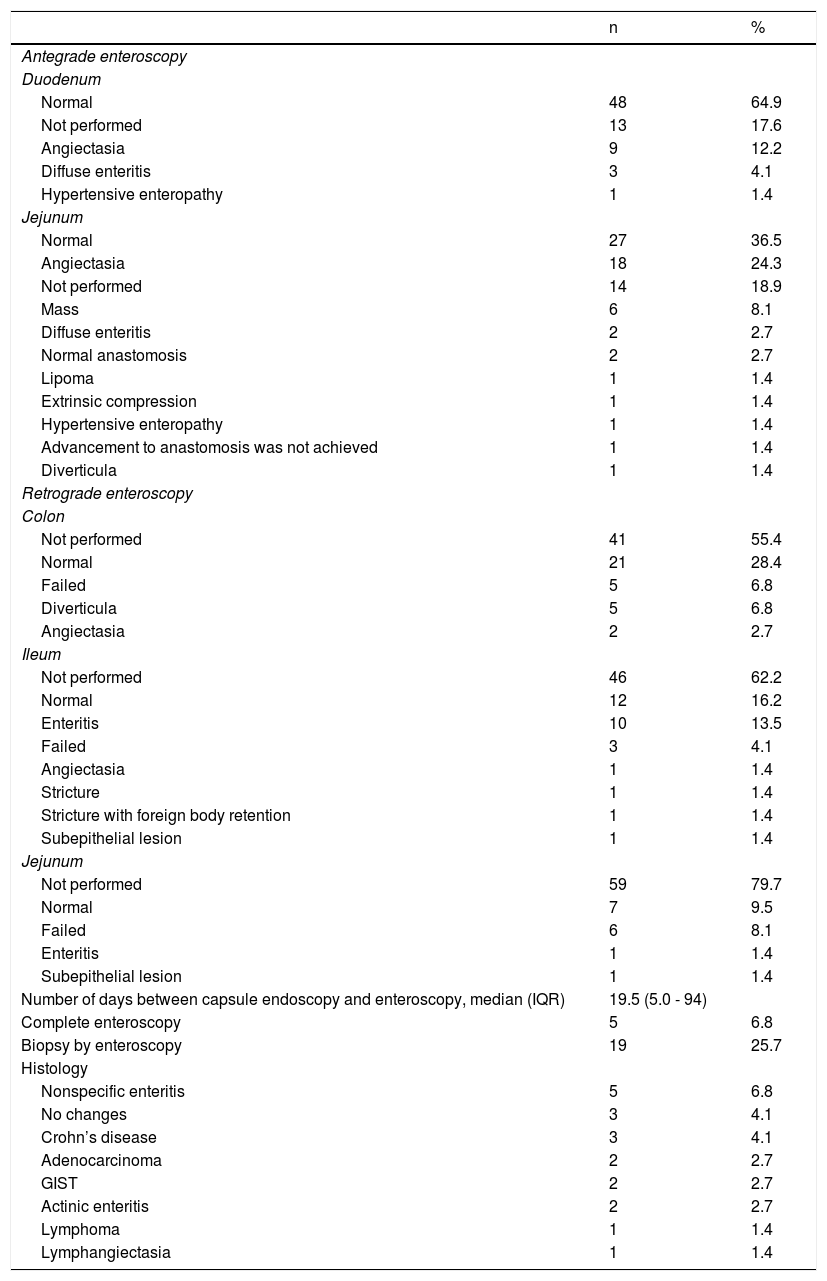
Sixty-three (85.13%) antegrade enteroscopies and 34 (45.94%) retrograde enteroscopies with single or double balloon were performed (some patients required both routes), confirming a total of 43 lesions with the following findings: angiectasias were identified mainly in the jejunum in 18 (24.3%) patients, followed by tumors in 6 (8.1%) patients; angiectasias were identified in the duodenum in 9 (12.2%) patients, followed by inflammatory lesions in 4 (5.5%) patients, and found via the antegrade route. Retrograde enteroscopy identified angiectasias in the colon in 5 (6.8%) patients and diverticula in 2 (2.7%) patients; enteritis was viewed in the ileum in 10 (13.5%) patients, stricture in 2 (2.7%) patients, angiectasias and subepithelial lesion in one (1.4%) patient; and enteritis and subepithelial lesion in the jejunum in one (1.4%) patient (Table 3).
Antegrade and retrograde single and double balloon enteroscopy findings, number of days between VCE and enteroscopy, biopsy, and histologic findings.
| n | % | |
|---|---|---|
| Antegrade enteroscopy | ||
| Duodenum | ||
| Normal | 48 | 64.9 |
| Not performed | 13 | 17.6 |
| Angiectasia | 9 | 12.2 |
| Diffuse enteritis | 3 | 4.1 |
| Hypertensive enteropathy | 1 | 1.4 |
| Jejunum | ||
| Normal | 27 | 36.5 |
| Angiectasia | 18 | 24.3 |
| Not performed | 14 | 18.9 |
| Mass | 6 | 8.1 |
| Diffuse enteritis | 2 | 2.7 |
| Normal anastomosis | 2 | 2.7 |
| Lipoma | 1 | 1.4 |
| Extrinsic compression | 1 | 1.4 |
| Hypertensive enteropathy | 1 | 1.4 |
| Advancement to anastomosis was not achieved | 1 | 1.4 |
| Diverticula | 1 | 1.4 |
| Retrograde enteroscopy | ||
| Colon | ||
| Not performed | 41 | 55.4 |
| Normal | 21 | 28.4 |
| Failed | 5 | 6.8 |
| Diverticula | 5 | 6.8 |
| Angiectasia | 2 | 2.7 |
| Ileum | ||
| Not performed | 46 | 62.2 |
| Normal | 12 | 16.2 |
| Enteritis | 10 | 13.5 |
| Failed | 3 | 4.1 |
| Angiectasia | 1 | 1.4 |
| Stricture | 1 | 1.4 |
| Stricture with foreign body retention | 1 | 1.4 |
| Subepithelial lesion | 1 | 1.4 |
| Jejunum | ||
| Not performed | 59 | 79.7 |
| Normal | 7 | 9.5 |
| Failed | 6 | 8.1 |
| Enteritis | 1 | 1.4 |
| Subepithelial lesion | 1 | 1.4 |
| Number of days between capsule endoscopy and enteroscopy, median (IQR) | 19.5 (5.0 - 94) | |
| Complete enteroscopy | 5 | 6.8 |
| Biopsy by enteroscopy | 19 | 25.7 |
| Histology | ||
| Nonspecific enteritis | 5 | 6.8 |
| No changes | 3 | 4.1 |
| Crohn’s disease | 3 | 4.1 |
| Adenocarcinoma | 2 | 2.7 |
| GIST | 2 | 2.7 |
| Actinic enteritis | 2 | 2.7 |
| Lymphoma | 1 | 1.4 |
| Lymphangiectasia | 1 | 1.4 |
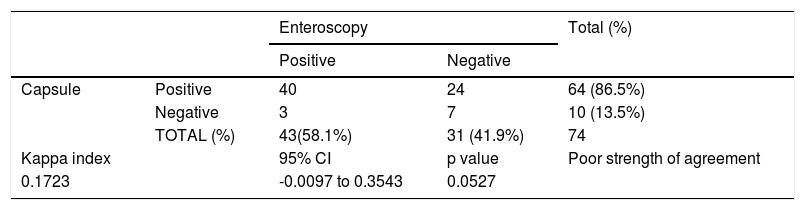
Overall diagnostic yield for the detection of positive findings was greater in VCE than in enteroscopy (86.5% vs. 58.1%, respectively, p = 0.0527), with no statistically significant differences between the two studies (Table 4). VCE detected 64 lesions, with signs of active bleeding in 13 (17.6%). Importantly, there were no signs of angiectasias in the enteroscopies of 14 (18.91%) patients whose VCEs showed them. There were statistically significant differences in the diagnostic yield of VCE, compared with enteroscopy, with respect to angiectasias (41.9% vs. 29.7% respectively, p = 0.0001) (Fig. 1), tumors (18.9% vs. 9.4%, respectively, p = 0.0002) (Figs. 2 and 3), and inflammatory lesions (23% vs. 19%, respectively, p < 0.0001).
Cohen’s Kappa statistic for positive pathologic findings between VCE and enteroscopy.
| Enteroscopy | Total (%) | |||
|---|---|---|---|---|
| Positive | Negative | |||
| Capsule | Positive | 40 | 24 | 64 (86.5%) |
| Negative | 3 | 7 | 10 (13.5%) | |
| TOTAL (%) | 43(58.1%) | 31 (41.9%) | 74 | |
| Kappa index | 95% CI | p value | Poor strength of agreement | |
| 0.1723 | -0.0097 to 0.3543 | 0.0527 | ||
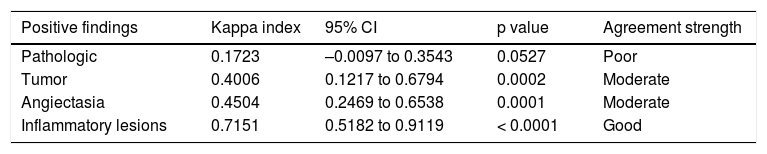
Overall, the kappa index for positive findings between VCE and enteroscopy was poor (Ik = 0.17, 95% CI: –0.0097-0.3543). However, the level of diagnostic agreement of the findings was modified, according to the type of lesion (Table 5).
Agreement summary of findings between capsule endoscopy and enteroscopy.
| Positive findings | Kappa index | 95% CI | p value | Agreement strength |
|---|---|---|---|---|
| Pathologic | 0.1723 | –0.0097 to 0.3543 | 0.0527 | Poor |
| Tumor | 0.4006 | 0.1217 to 0.6794 | 0.0002 | Moderate |
| Angiectasia | 0.4504 | 0.2469 to 0.6538 | 0.0001 | Moderate |
| Inflammatory lesions | 0.7151 | 0.5182 to 0.9119 | < 0.0001 | Good |
The present study found a similar diagnostic yield between VCE and antegrade and/or retrograde, single or double balloon enteroscopy performed after the VCE, on patients suspected of having small bowel bleeding that had negative upper gastrointestinal endoscopy and colonoscopy (86.5% vs. 58.1% respectively, p = 0.0527). In addition, overall agreement was low, regarding the same findings in the two modalities, with respect to lesions and anatomic site (k = 0.17). However, diagnostic agreement was modified, according to the type of lesion, obtaining a higher frequency of agreement in inflammatory lesions (k = 0.71), angiectasias (k = 0.45), and tumor (k = 0.40).
Due to its location and length, the small bowel used to be considered the Pandora’s box of the gastrointestinal tract for the study of pathologies at that level. The diagnostic approach to the middle intestine improved when the Food and Drug Administration (FDA) approved its examination through enteroscopy in the year 2000 and VCE in 2001. Of those two diagnostic methods, VCE is the method of choice for the study of the small bowel, because of its technical and operative characteristics, being less invasive and less expensive. It indicates the access route if a later invention is required. It is limited by the fact that biopsies cannot be taken, and interventions cannot be performed,5,6,8 which is where enteroscopy becomes essential as a diagnostic and therapeutic method.9–11 The majority of reports describe a similar yield for the two techniques. In the study by Tian Min et al.,12 VCE followed by enteroscopy was performed on 62 patients, finding an overall detection rate of intestinal lesions of 70.9% (44/62), with no significant differences between the 2 methods (p = 0.6739). Likewise, there were no statistically significant differences in the diagnostic yield between the two techniques in the present study.
In a systematic review by Liao Z et al.13 that included 227 retrospective and prospective studies, with a total of 22,840 VCEs for evaluating small bowel lesions, the most frequent indication was occult gastrointestinal bleeding (66%). The diagnostic yield was 59.4% for lesions, in general, and 60.5% for occult bleeding. Angiectasias were identified in 50% of the cases. The complete retention rate was 1.4%, the same as the rate in the present study (n = 1, 1.4%). The diagnostic yield of that study was higher for positive pathologic findings (86.5%) and the main indication was obscure overt gastrointestinal bleeding.
Fukumoto et al.14 evaluated 76 patients suspected of having small bowel pathology. Total enteroscopy was achieved in 77.6% of the patients with VCE and 56.6% with double balloon enteroscopy. Lesions were detected in 42/76 patients with VCE and 46/76 patients with enteroscopy, with no statistically significant differences and a moderate agreement rate (kappa index = 0.57). Differences were observed between VCE and enteroscopy in 16 patients and no lesions were detected through enteroscopy in 6 patients. Different analyses have shown that VCE is limited for detecting certain submucosal lesions due to the lack of air insufflation, especially in the proximal intestine. In the present study, 64/74 lesions were detected through VCE and 43/74 lesions were detected through enteroscopy, with no statistically significant differences, but with poor agreement (0.17, 95% CI: –0.0097-0.3543). No lesions were detected through enteroscopy in 24 patients, in whom lesions were identified in the previous VCE. In a meta-analysis and subgroup analysis of positive findings, lesions were detected in 72.2% of the VCEs but were identified in only 27.8% of the double balloon enteroscopies.15 Unlike that reported by other authors, in our study, the diagnostic yield for tumor was greater with VCE than with enteroscopy (18.9% vs. 9.4%, respectively, p = 0.0002), with moderate agreement (0.40, 95% CI: 0.1217-0.6794). The poor agreement in the interpretation of positive pathologic findings was due to the fact that we determined the agreement between the lesions identified with the same findings and at the same anatomic site in both the VCE and enteroscopy. That was the importance of our study, which differs from previous analyses. A large portion of the literature does not take those relations into account, which could be due to the poor agreement between the two procedures.
A 2011 meta-analysis that included 10 studies, with a total of 651 patients, reported that the diagnostic yield for VCE and double balloon enteroscopy was 62% and 56%, respectively, with no difference in identifying the cause of bleeding.16 The main indication for enteroscopy was overt intestinal bleeding, with an overall yield of pathologic findings in the small intestine of 65.21%.17 In our case series, the diagnostic yield was similar for the 2 procedures (86.5% vs. 58.1%, respectively, p = 0.0527), with higher values than others reported. Nevertheless, diagnostic yield is modified by different factors. Therefore, inconclusive or negative VCEs are indications for performing complementary enteroscopy.18 In the present study, in the 10 patients with negative VCE, lesions were confirmed through enteroscopy in 3 (30%) patients (VCE false negatives). Likewise, the enteroscopy false negatives in 24 (37.5%) patients could be related to the interval of time between the two procedures and the difficulty in performing complete enteroscopies, which were only achieved in 5 (6.8%) patients.
In a retrospective cohort of 418 patients that underwent VCE, Shiani et al.19 evaluated 95 patients with positive findings in VCE that then underwent diagnostic and therapeutic single balloon enteroscopy. There was strong agreement for active bleeding and blood clots, moderate agreement for vascular lesions, and poor agreement for ulcers. There was no correlation for masses and polyps, unlike the present study that showed moderate agreement for tumors.
Marmo et al.20 conducted a prospective, multicenter study on 193 patients and reported good agreement for vascular lesions (0.72, 95% CI: 0.59-0.84) and inflammatory lesions (0.78, 95% CI: 0.58-0.99]) and no agreement for polyps or tumors. Similarly, our study showed that agreement varied, depending on the type of lesion. It was good for inflammatory lesions, but moderate for tumors. Our results concur with reports that the two methods are specific and complementary for the detection of tumors in the small intestine.21
Systematic reviews including 12,823 procedures, describe total enteroscopy, defined as the examination of the entire small bowel, whether by antegrade or retrograde access routes or a combination of the two, in 44% of the patients. One of the limitations for its performance is the technical difficulty involved and the duration of the examination, which can take up to 4 hours.22 The performance rate of complete single balloon enteroscopy has been reported at 26.1%,23 which is lower, compared with the double balloon procedure.24,25 There is quite a difference with the present study, in which total enteroscopy was achieved in 6.8% of the patients, with the limitation of not distinguishing between the single or double balloon procedures. Reports in the literature show no difference in the diagnostic yield, the therapeutic yield, or the failed enteroscopy rate between single and double balloon enteroscopy.26,27
Other limitations of the present study, apart from those already mentioned, were its retrospective design, interobserver variation, and the fact that it was conducted at a referral hospital, meaning there could be a reference bias with an increase in diagnostic yield. In addition, a Pillcam SB3 and SB2 subgroup analysis was not carried out, given that, according to the literature, the SB3 detects a higher number of P1 lesions, without modifying the diagnostic yield of the P2 lesions that were selected as significant, given their high potential for bleeding.28
ConclusionsThe present study showed that the overall yield was similar between VCE and single and double balloon enteroscopy for detecting small bowel lesions, with a poor overall agreement for positive findings. The main factor that could modify those results was the type of lesion, with good agreement for inflammatory lesions and moderate agreement for angiectasias and tumors.
Financial disclosureThe present study was self-funded.
Conflict of interestThe authors declare that there is no conflict of interest.
Please cite this article as: Cañadas Garrido R, Rincón Sánchez RA, Costa Barney VA, Roa Ballestas PA, Espinosa Martínez CA, Pinzón Arenas DF, et al. Concordancia diagnóstica entre la videocápsula endoscópica y enteroscopia mono y de doble balón en la hemorragia de intestino delgado en un hospital de alta complejidad en Bogota, Colombia. Revista de Gastroenterología de México. 2021;86:51–58.