The present review of noncaloric sweeteners (NCSs) by the Asociación Mexicana de Gastroenterología was carried out to analyze and answer some of the most frequent questions and concerns about NCS consumption in patients with gastrointestinal disorders, through a thorough review of the medical literature. A group of gastroenterologists and experts on nutrition, toxicology, microbiology, and endocrinology reviewed and analyzed the published literature on the topic. The working group formulated conclusions, based on the scientific evidence published, to give an opinion with respect to NCS ingestion. Current evidence does not confirm the carcinogenic potential of NCSs. However, the studies analyzed showed that saccharin could have a proinflammatory effect and that polyols can cause gastrointestinal symptoms and manifestations, depending on the dose and type of compound. The ingestion of xylitol, erythritol, sucralose, aspartame, acesulfame K, and saccharin could increase the secretion of the gastrointestinal hormones that regulate intestinal motility, and stevia and its derivatives could have a favorable effect on the percentage of liver fat. Caution should be taken in recommending aspartame consumption in patients with chronic liver disease because it reduces the ratio of branched-chain amino acids to aromatic amino acids. In addition, NCS ingestion could modify the composition of the intestinal microbiota, having an effect on gastrointestinal symptoms and manifestations. It is important to continue conducting causality studies on humans to be able to establish recommendations on NSC consumption.
Esta revisión de la Asociación Mexicana de Gastroenterología sobre edulcorantes no calóricos (ENC) se realizó con el fin de analizar y responder a través de una amplia revisión bibliográfica, algunas de las preguntas y preocupaciones más frecuentes sobre la ingestión de ENC en pacientes con alteraciones gastrointestinales. Un grupo de gastroenterólogos, expertos en nutrición, toxicología, microbiología y endocrinología, revisó y analizó la literatura publicada en este tópico. El grupo de trabajo generó conclusiones basadas en la evidencia científica publicada para emitir una opinión respecto a su ingestión. En este sentido, la evidencia existente hasta el día de hoy no confirma el potencial carcinogénico de los ENC; sin embargo, los estudios evaluados mostraron que la sacarina podría tener un efecto proinflamatorio y los polioles pueden causar síntomas y manifestaciones gastrointestinales dependiendo del tipo y dosis del compuesto. La ingestión de xilitol, eritritol, sucralosa, aspartame, acesulfame K y sacarina podrían incrementar la secreción de hormonas gastrointestinales reguladoras de la motilidad intestinal. Los glucósidos de esteviol podrían tener un efecto favorable en el porcentaje de grasa hepática. Se debe tener precaución en la recomendación de la ingestión de aspartame en pacientes con hepatopatía crónica debido a que disminuye la relación entre aminoácidos de cadena ramificada y aromáticos. Además, la ingestión de ENC podría modificar la composición de la microbiota intestinal y esto tener efectos sobre los síntomas y manifestaciones gastrointestinales. Es importante que se continúe realizando estudios de causalidad en humanos para poder establecer recomendaciones sobre la ingestión de ENC.
Carbohydrates (CHOs) are an important and necessary part of a healthy diet. According to the Food and Agriculture Organization,1 CHOs should make up from 45 to 60% of the daily energy intake in adults and in children above one year of age. Monosaccharides and disaccharides, commonly known as sugars, are an important part of the CHOs available in foods. Their dietary requirements are chiefly met by the consumption of fruits, fruit juices, vegetables, milk and dairy products, cereals, and foods containing added sugars and starch hydrolysates.2 The consumption of foods with a high CHO content has increased worldwide and they have been associated with metabolic syndrome (MS), cardiovascular diseases, and type 2 diabetes mellitus (DM). Thus, new ingredients, such as the noncaloric sweeteners (NCSs), have emerged to provide foods with the same sweet taste of those sugars, but without the effects attributed to them. Currently, NCSs are widely used in the formulation of foods and beverages, mainly for their low or null calorie content, their low cost, and their sweetness intensity, compared with sucrose (table sugar) or other caloric sweeteners.3,4 NCSs were introduced into the food industry more than 100 years ago and have gained in popularity due to the perceived health benefits of weight loss and improved blood glucose concentrations.5
Different NCSs are regularly used in the food industry worldwide, depending on each country’s regulations. The US Food and Drug Administration (FDA) has approved six NCSs: saccharin, aspartame, neotame, acesulfame-K, sucralose, and advantame. In addition, the two substances, stevia and luo han guo, have been determined to be generally recognized as safe (GRAS).6 Each of those sweeteners has a different intensity, chemical formula, and metabolism associated with gastrointestinal symptoms and manifestations, as shown in Table 1.3,6
The main NCSs on the market, sweetness intensity, sources, and characteristics of interest in the production of gastrointestinal symptoms and manifestations.
| Sweeteners | Main characteristics |
|---|---|
| Saccharin | It has a bitter or metallic taste, is used in sodium or calcium salt forms, and is 200 to 700-times sweeter than sucrose. |
| Aspartame | It is a methyl ester of the aspartic acid and phenylalanine dipeptide, which under very acid or alkaline conditions can be converted into methanol by hydrolysis, and it is 20-times sweeter than sucrose. |
| Neotame | It is a derivative of the aspartic acid and phenylalanine dipeptide compound and is 7,000 to 13,000-times sweeter than sucrose. |
| Acesulfame-K | It is a crystalline potassium salt, 200-times sweeter than sucrose. |
| Sucralose | From 11 to 27% is absorbed, with the rest remaining in the gastrointestinal tract until expelled in feces, and it is 600-times sweeter than sucrose. |
| Advantame | It is 20,000-times sweeter than sucrose. |
| Stevia | It is a heat-stable sweetener of natural origin and 200 to 400-times sweeter than sucrose. |
| Luo han guo | It is 100 to 250-times sweeter than sucrose. |
| Polyols (xylitol, mannitol sorbitol, maltitol) | They are four-carbon sugar alcohols, a product of fermentation of glucose and sucrose, low-digestible, with a high osmotic potential in the intestinal lumen, and they are 60 to 80% sweeter than sucrose. |
To be approved by the FDA, each NCS must undergo testing to establish the amount that is acceptable and safe for consumption, known as the acceptable daily intake (ADI) (Table 2). The ADI is established through toxicity testing on different animal species at different life stages and in several generations and by determining the absorption, digestion, metabolism, and excretory capacity in humans.6,7
Acceptable daily intake of each sweetener approved by the FDA.
| Sweetener | ADI |
|---|---|
| Saccharin | 5mg/ kg of body weight |
| Cyclamate | 7mg/ kg of body weight |
| Aspartame | 50mg/ kg of body weight |
| Sucralose | 15mg/ kg of body weight |
| Acesulfame-K | 15mg/ kg of body weight |
| Stevia | 4mg/ kg of body weight |
ADI: acceptable daily intake; FDA: food and drug administration.
There has been an increased intake of the abovementioned NCSs in the population with DM and in healthy persons as a result of lifestyle changes worldwide, especially in the countries that consume a Western diet. Therefore, they are more frequently found in foods and beverages that are not necessarily labeled as low-calorie or sweetened with an NCS.8 In the United States, a study was recently published that showed that 15% of the production volume of foods and beverages between 2005 and 2009 contained NCSs, and that figure has been gradually rising.9
Nevertheless, some studies have related NCS ingestion to an increased risk for obesity, MS, and type 2 DM. However, given the complexity of NCS evaluation in humans, there are few randomized clinical trials assessing their effect on health, and the methodology of those studies has been questioned.5,10–18
Specifically, the studies on the gut microbiota have been the most controversial, given its diversity that is dependent on different host conditions and their interaction with diet. Thus, the majority of measurements have been made through the association of the microbiome (genome of the microbiota), the presence of NCSs in the colon, and the possible clinical manifestations.19,20 In addition, the NCSs that are not completely absorbed in the small bowel could cause alterations in the gut microbiota, modifying the bacterial balance,21 which could then trigger changes in bowel habit and motility, producing increased gastrointestinal manifestations in patients with a gastrointestinal disease.22
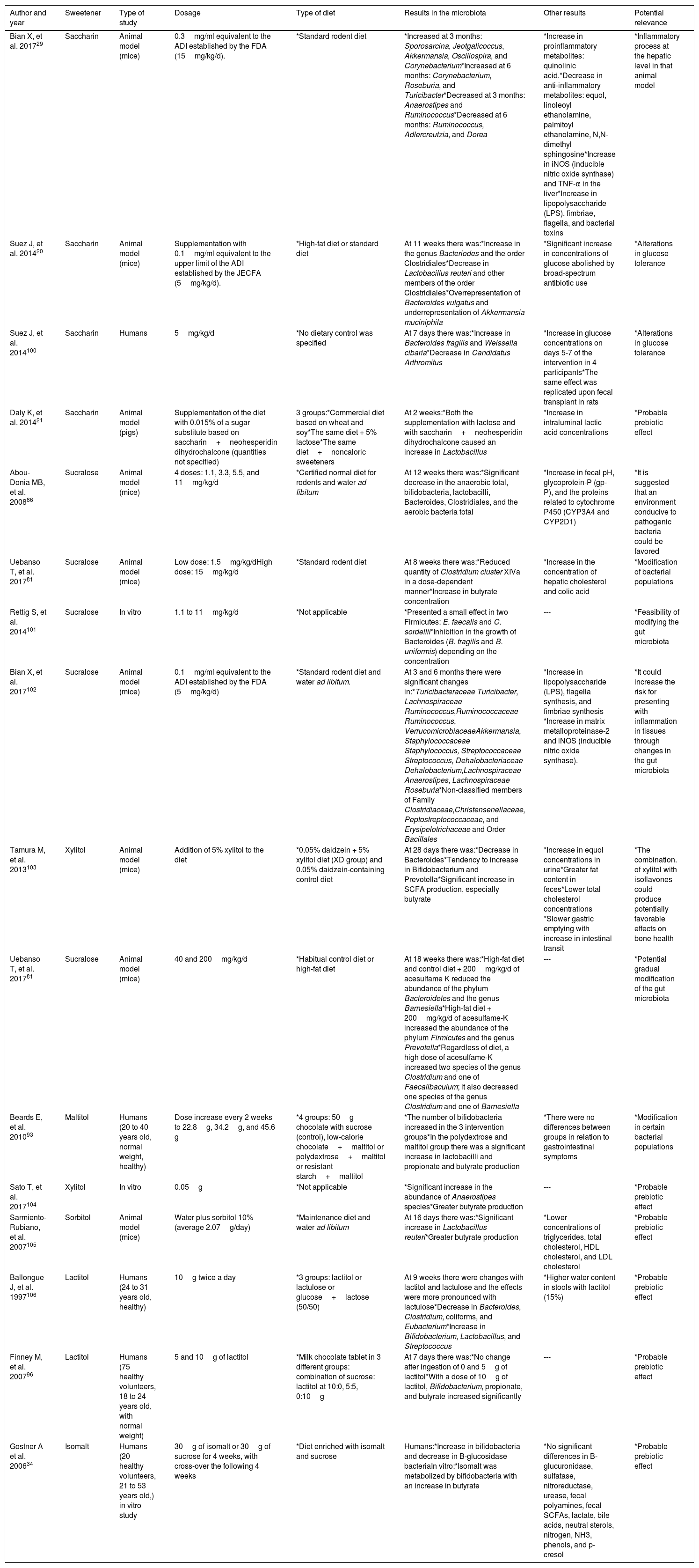
Therefore, the aim of the present work was to provide an expert opinion, with respect to the effect of NCS ingestion on gastrointestinal health, based on published scientific evidence, through a thorough review of the existing literature (Table 3).
Systematization of the bibliography used for the study.
| Author and year | Sweetener | Type of study | Dosage | Type of diet | Results in the microbiota | Other results | Potential relevance |
|---|---|---|---|---|---|---|---|
| Bian X, et al. 201729 | Saccharin | Animal model (mice) | 0.3mg/ml equivalent to the ADI established by the FDA (15mg/kg/d). | *Standard rodent diet | *Increased at 3 months: Sporosarcina, Jeotgalicoccus, Akkermansia, Oscillospira, and Corynebacterium*Increased at 6 months: Corynebacterium, Roseburia, and Turicibacter*Decreased at 3 months: Anaerostipes and Ruminococcus*Decreased at 6 months: Ruminococcus, Adlercreutzia, and Dorea | *Increase in proinflammatory metabolites: quinolinic acid.*Decrease in anti-inflammatory metabolites: equol, linoleoyl ethanolamine, palmitoyl ethanolamine, N,N-dimethyl sphingosine*Increase in iNOS (inducible nitric oxide synthase) and TNF-α in the liver*Increase in lipopolysaccharide (LPS), fimbriae, flagella, and bacterial toxins | *Inflammatory process at the hepatic level in that animal model |
| Suez J, et al. 201420 | Saccharin | Animal model (mice) | Supplementation with 0.1mg/ml equivalent to the upper limit of the ADI established by the JECFA (5mg/kg/d). | *High-fat diet or standard diet | At 11 weeks there was:*Increase in the genus Bacteriodes and the order Clostridiales*Decrease in Lactobacillus reuteri and other members of the order Clostridiales*Overrepresentation of Bacteroides vulgatus and underrepresentation of Akkermansia muciniphila | *Significant increase in concentrations of glucose abolished by broad-spectrum antibiotic use | *Alterations in glucose tolerance |
| Suez J, et al. 2014100 | Saccharin | Humans | 5mg/kg/d | *No dietary control was specified | At 7 days there was:*Increase in Bacteroides fragilis and Weissella cibaria*Decrease in Candidatus Arthromitus | *Increase in glucose concentrations on days 5-7 of the intervention in 4 participants*The same effect was replicated upon fecal transplant in rats | *Alterations in glucose tolerance |
| Daly K, et al. 201421 | Saccharin | Animal model (pigs) | Supplementation of the diet with 0.015% of a sugar substitute based on saccharin+neohesperidin dihydrochalcone (quantities not specified) | 3 groups:*Commercial diet based on wheat and soy*The same diet + 5% lactose*The same diet+noncaloric sweeteners | At 2 weeks:*Both the supplementation with lactose and with saccharin+neohesperidin dihydrochalcone caused an increase in Lactobacillus | *Increase in intraluminal lactic acid concentrations | *Probable prebiotic effect |
| Abou-Donia MB, et al. 200886 | Sucralose | Animal model (mice) | 4 doses: 1.1, 3.3, 5.5, and 11mg/kg/d | *Certified normal diet for rodents and water ad libitum | At 12 weeks there was:*Significant decrease in the anaerobic total, bifidobacteria, lactobacilli, Bacteroides, Clostridiales, and the aerobic bacteria total | *Increase in fecal pH, glycoprotein-P (gp-P), and the proteins related to cytochrome P450 (CYP3A4 and CYP2D1) | *It is suggested that an environment conducive to pathogenic bacteria could be favored |
| Uebanso T, et al. 201781 | Sucralose | Animal model (mice) | Low dose: 1.5mg/kg/dHigh dose: 15mg/kg/d | *Standard rodent diet | At 8 weeks there was:*Reduced quantity of Clostridium cluster XIVa in a dose-dependent manner*Increase in butyrate concentration | *Increase in the concentration of hepatic cholesterol and colic acid | *Modification of bacterial populations |
| Rettig S, et al. 2014101 | Sucralose | In vitro | 1.1 to 11mg/kg/d | *Not applicable | *Presented a small effect in two Firmicutes: E. faecalis and C. sordellii*Inhibition in the growth of Bacteroides (B. fragilis and B. uniformis) depending on the concentration | --- | *Feasibility of modifying the gut microbiota |
| Bian X, et al. 2017102 | Sucralose | Animal model (mice) | 0.1mg/ml equivalent to the ADI established by the FDA (5mg/kg/d) | *Standard rodent diet and water ad libitum. | At 3 and 6 months there were significant changes in:*Turicibacteraceae Turicibacter, Lachnospiraceae Ruminococcus,Ruminococcaceae Ruminococcus, VerrucomicrobiaceaeAkkermansia, Staphylococcaceae Staphylococcus, Streptococcaceae Streptococcus, Dehalobacteriaceae Dehalobacterium,Lachnospiraceae Anaerostipes, Lachnospiraceae Roseburia*Non-classified members of Family Clostridiaceae,Christensenellaceae, Peptostreptococcaceae, and Erysipelotrichaceae and Order Bacillales | *Increase in lipopolysaccharide (LPS), flagella synthesis, and fimbriae synthesis *Increase in matrix metalloproteinase-2 and iNOS (inducible nitric oxide synthase). | *It could increase the risk for presenting with inflammation in tissues through changes in the gut microbiota |
| Tamura M, et al. 2013103 | Xylitol | Animal model (mice) | Addition of 5% xylitol to the diet | *0.05% daidzein + 5% xylitol diet (XD group) and 0.05% daidzein-containing control diet | At 28 days there was:*Decrease in Bacteroides*Tendency to increase in Bifidobacterium and Prevotella*Significant increase in SCFA production, especially butyrate | *Increase in equol concentrations in urine*Greater fat content in feces*Lower total cholesterol concentrations *Slower gastric emptying with increase in intestinal transit | *The combination. of xylitol with isoflavones could produce potentially favorable effects on bone health |
| Uebanso T, et al. 201781 | Sucralose | Animal model (mice) | 40 and 200mg/kg/d | *Habitual control diet or high-fat diet | At 18 weeks there was:*High-fat diet and control diet + 200mg/kg/d of acesulfame K reduced the abundance of the phylum Bacteroidetes and the genus Barnesiella*High-fat diet + 200mg/kg/d of acesulfame-K increased the abundance of the phylum Firmicutes and the genus Prevotella*Regardless of diet, a high dose of acesulfame-K increased two species of the genus Clostridium and one of Faecalibaculum; it also decreased one species of the genus Clostridium and one of Barnesiella | --- | *Potential gradual modification of the gut microbiota |
| Beards E, et al. 201093 | Maltitol | Humans (20 to 40 years old, normal weight, healthy) | Dose increase every 2 weeks to 22.8g, 34.2g, and 45.6 g | *4 groups: 50g chocolate with sucrose (control), low-calorie chocolate+maltitol or polydextrose+maltitol or resistant starch+maltitol | *The number of bifidobacteria increased in the 3 intervention groups*In the polydextrose and maltitol group there was a significant increase in lactobacilli and propionate and butyrate production | *There were no differences between groups in relation to gastrointestinal symptoms | *Modification in certain bacterial populations |
| Sato T, et al. 2017104 | Xylitol | In vitro | 0.05g | *Not applicable | *Significant increase in the abundance of Anaerostipes species*Greater butyrate production | --- | *Probable prebiotic effect |
| Sarmiento-Rubiano, et al. 2007105 | Sorbitol | Animal model (mice) | Water plus sorbitol 10% (average 2.07g/day) | *Maintenance diet and water ad libitum | At 16 days there was:*Significant increase in Lactobacillus reuteri*Greater butyrate production | *Lower concentrations of triglycerides, total cholesterol, HDL cholesterol, and LDL cholesterol | *Probable prebiotic effect |
| Ballongue J, et al. 1997106 | Lactitol | Humans (24 to 31 years old, healthy) | 10g twice a day | *3 groups: lactitol or lactulose or glucose+lactose (50/50) | At 9 weeks there were changes with lactitol and lactulose and the effects were more pronounced with lactulose*Decrease in Bacteroides, Clostridium, coliforms, and Eubacterium*Increase in Bifidobacterium, Lactobacillus, and Streptococcus | *Higher water content in stools with lactitol (15%) | *Probable prebiotic effect |
| Finney M, et al. 200796 | Lactitol | Humans (75 healthy volunteers, 18 to 24 years old, with normal weight) | 5 and 10g of lactitol | *Milk chocolate tablet in 3 different groups: combination of sucrose: lactitol at 10:0, 5:5, 0:10g | At 7 days there was:*No change after ingestion of 0 and 5g of lactitol*With a dose of 10g of lactitol, Bifidobacterium, propionate, and butyrate increased significantly | --- | *Probable prebiotic effect |
| Gostner A et al. 200634 | Isomalt | Humans (20 healthy volunteers, 21 to 53 years old,) in vitro study | 30g of isomalt or 30g of sucrose for 4 weeks, with cross-over the following 4 weeks | *Diet enriched with isomalt and sucrose | Humans:*Increase in bifidobacteria and decrease in B-glucosidase bacteriaIn vitro:*Isomalt was metabolized by bifidobacteria with an increase in butyrate | *No significant differences in B-glucuronidase, sulfatase, nitroreductase, urease, fecal polyamines, fecal SCFAs, lactate, bile acids, neutral sterols, nitrogen, NH3, phenols, and p-cresol | *Probable prebiotic effect |
ADI: acceptable daily intake; FDA (Food and Drug Administration; HDL: high density lipoprotein; JECFA: Joint FAO/WHO Committee Report on Food Additives; LDL: low density lipoprotein; SCFA: short chain fatty acid.
Two general coordinators from the areas of gastroenterology and nutrition (RVF and NBH) were designated and experts in gastroenterology, nutrition, toxicology, microbiology, and endocrinology were invited to participate in the review. A thorough search of the literature was carried out utilizing the following databases: The Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE (PubMed), EMBASE (Ovid), LILACS, CINAHL, BioMed Central, and the World Health Organization International Clinical Trials Registry Platform (ICTRP). The search included articles published within the time frame of 1969 to 2018. The search criteria consisted of the following terms: «artificial sweeteners» «non-caloric sweeteners» combined with the terms: «stevia», «sucralose», «aspartame», «acesulfame-k», «saccharin», «inflammation», «polyols», «microbiota», «carcinogenesis», «treatment», «gastrointestinal symptoms», «cirrhosis», «cancer», «therapy», «NAFLD», «NASH», «review», «guidelines» and their Spanish equivalents. The complete bibliography was distributed to all the members of the review team. The coordinators then formulated questions that were submitted to a first electronic evaluation to assess their drafting, content, and clinical relevance. Four teams composed of 4 or 5 collaborators each had the task of answering 5 questions, based on the evaluated scientific evidence. Each of the evidence-based answers was openly presented, discussed, analyzed, and agreed upon by all the participants. Once there was a consensus on the response and opinion, a summary was drafted for its incorporation into the final document.
ResultsGeneral characteristics of the noncaloric sweetenersNCS absorption, metabolism, and excretion varies, depending on the compound and its quantity (Table 1).
Acesulfame-K is a derivative of hydrophilic organic acid that is almost completely absorbed in the small bowel by microvilli action, to then enter the systemic circulation and be distributed to the tissues of the entire body. Ninety-nine percent of the nonabsorbable portion is excreted in urine and less than 1% in feces, without being metabolized.23
Aspartame is a chemical bond of two amino acids (phenylalanine and aspartic acid). Its hydrolysis and absorption take place in the gastrointestinal tract through the action of esterases and peptidases. Its products that are absorbable in the intestinal mucosa are methanol (10%), aspartic acid (50%), and phenylalanine (40%), which take different metabolic pathways, depending on their composition.24
Saccharin is an o-benzoic acid sulfimide, 85 to 95% of which is absorbed in the small bowel. It binds reversibly to plasma proteins and is distributed to all the organs. The nonabsorbable portion is eliminated in urine and feces, without being metabolized.24
Steviol glycosides, also known as “stevia”, are obtained from a plant (Stevia rebaudiana Bertoni) whose leaves produce extracts of two of the sweet glycosides, stevioside and rebaudioside. They are slowly absorbed into the portal circulation and can undergo glucuronidation in the liver and be re-transported by bile into the intestine.25
Cyclamate is the sodium salt or calcium salt of cyclamic acid (cyclohexylsulfamic acid). Approximately 40% is absorbed by the intestine and eliminated in urine without being metabolized. Thirty percent of the non-absorbed portion is metabolized by the microbiota (Enterococcus) into cyclohexylamine, cyclohexanol, and cyclohexanone.26,27
Noncaloric sweeteners and their relation to the inflammatory gastrointestinal processThere are no studies on humans about the effect of NCSs and the inflammatory process. The evidence is limited and comes from cell lines or studies on animal models.28
A study conducted on a murine model showed that saccharin administered for 6 months induced increased expression of certain inflammation factors, such as the inducible nitric oxide synthetase (iNOS) enzyme and tumor necrosis factor-alpha (TNF-α) in male rat livers (C57BL/6J). Said study suggested that the mRNA expression of the genes that encode those factors could be indirectly associated with the alteration of the gut microbiota and its metabolic functions.29
On the other hand, steviol glycoside and its related compounds have antioxidant and anti-inflammatory effects. Stevioside has been shown to significantly suppress the release of TNF-α and interleukin (IL)-1ß, induced by lipopolysaccharides and the mild suppression of nitric oxide in THP-1 monocytes (a leukemia-derived cell line), without having a direct toxic effect.30 Likewise, different studies have shown that high concentrations of stevioside and steviol reduced the viability of human colon cancer cell lines. Specifically, steviol increased chloride secretion and attenuated the IL-8 production stimulated by TNF-α.31 Likewise, stevioside dose-dependently inhibited the expression of TNF-α, IL-6, and IL-1β in RAW264 cells (murine macrophagic monocytes) stimulated by lipopolysaccharides and exerted an anti-inflammatory property by inhibiting NF-κβ activation, the mitogen-activated protein kinase signaling, and the release of proinflammatory cytokines.32 In other studies, stevioside and steviol showed no cytotoxicity in cell cultures and both compounds had mediating effects on the inflammatory process through the potential release of TNF-α, IL-1β, and IL-6. At present, the findings suggest that stevioside could be a therapeutic agent in inflammatory diseases, and that steviol and isosteviol, together with rebaudioside A, could have therapeutic benefits because they have been associated with antihyperglycemic, antihypertensive, anti-inflammatory, antitumor, antidiarrheic, diuretic, and immunomodulatory beneficial actions.33
In conclusion, there is scant experimental evidence on a potential direct proinflammatory effect of saccharin. To the contrary, there is evidence, albeit no clinical studies, with respect to the anti-inflammatory activity of steviol and its derivatives.
Polyols and gastrointestinal manifestationsTo analyze the gastrointestinal symptoms and manifestations associated with NCS ingestion, it is important to differentiate the high-intensity sweeteners from those that provide volume and texture. Polyols used as additives or to confer volume, texture, or sweetness to foods, belong to that latter group.34
The polyols erythritol, isomaltol, lactitol, maltitol, polyglycitol, mannitol, sorbitol, xylitol, and tagatose are hydrogenated CHOs present in some fruits, vegetables, mushrooms, and some industrialized foods.35,36 Erythritol is absorbed in the small bowel without being metabolized and so ingestion of 80g per day is tolerated,37 whereas isomaltol is partially absorbed and highly fermentable by the gut microbiota of the colon (approximately 90%).34 Consequently, those low-digestible CHOs can cause diarrhea and other gastrointestinal manifestations, such as flatulence, bloating, and abdominal discomfort.38 Symptoms vary depending on their dose, if they are liquid or solid, if they are consumed together with other foods, and ingestion velocity, among others.39
Wolnerhanssen et al. conducted a double-blind, randomized clinical trial with 10 subjects per group to evaluate the effect of xylitol (50g) and erythritol (75g), compared with placebo (75g of glucose), on the release of glucagon-like protein-1 (GLP-1), cholecystokinin (CCK), and gastric emptying. The results showed that volunteers that consumed erythritol and xylitol had a delay in gastric emptying and increased secretion of GLP-1 and CCK.40
Other studies have tested dosage to make recommendations regarding the tolerability of those NCSs. The doses tested varied from 20g/day (mannitol) to 40g/day (isomaltol, maltitol, and polyglycitol).41 However, some studies have shown that a dose above 20g/day of lactitol caused flatulence and distension. Likewise, sorbitol, at doses of 20-30g/day, caused abdominal pain, and at doses above 50g/day, caused osmotic diarrhea.42
Thus, it can be concluded that polyols can cause gastrointestinal symptoms and manifestations, depending on the type of compound and the dose ingested.
Noncaloric sweeteners and intestinal motilityThere is little direct evidence that relates NCS ingestion to intestinal motility modifications. Results in animal models suggest that certain NCSs can promote the release of GLP-1 or gastroinhibitory peptide, which modify intestinal movement.43,44
In vivo and in vitro studies44,45 have shown that sucralose, aspartame, and acesulfame-K cause enteroendocrine cell stimulation, increasing the release of GLP-1, CCK, and peptide YY (the anorexigenic peptide released by the cells of the colon). One hypothesis is the activation of sweet taste receptors in the mouth. However, similar responses have been seen by directly infusing NCSs into the intestine, due to the presence of sweet taste receptors in the small bowel. NCSs could bind to those sweet taste receptors, stimulating the release of the gastroinhibitory peptide and GLP-1, which modify intestinal movement. Nevertheless, conclusive effects on intestinal motility, or an increase in GLP-1 or insulin, have not been seen in clinical trials,46 nor have studies with aspartame and saccharin shown clinical evidence of gastric emptying disorders.47–49
In contrast, the results of Meyer-Gerspach et al. showed that there was greater appetite stimulation and reduced satiety, after acesulfame-K ingestion, compared with a placebo group that did not show those changes.50 Brown et al. reported that the ingestion of sucralose and acesulfame-K in carbonated beverages, prior to a glucose load, produced a significant increase of GLP-1.51 Those results were reproducible in other populations, in which there was a significant association between the ingestion of those NCSs plus glucose and a greater release of GLP-1.52,53
In conclusion, there are studies on animal models that have shown an increase in gastrointestinal hormone secretion indirectly related to intestinal motility. However, there is not enough information on humans to sustain that NCSs, per se, directly affect intestinal motility. Thus, more studies are needed to establish their role in digestive tract functioning.
Noncaloric sweeteners and cancer in the digestive tractStudies conducted at the beginning of the 1970s on animal models exposed to high doses of saccharin showed carcinogenic effects in the bladder. That triggered a series of clinical studies on humans that evaluated the carcinogenic effect of NCSs, and the results were not reproducible. Likewise, no scientific evidence on humans has currently been published that describes its carcinogenic effect in the bladder.54 Cyclamate was also studied under the same conditions, and at present, no carcinogenic effects on the bladder have been described.55 A case-control study (556 cases of stomach cancer or pancreatic cancer vs. 1,199 controls) on an Italian population, whose aim was to evaluate the risk for cancer from saccharin and aspartame ingestion, showed no statistically significant differences for the development of gastric cancer or pancreatic cancer. The authors concluded that there was no increased risk for neoplasias common in that population.56
In another case-control study that evaluated the role of saccharin and other NCSs in cancers of the esophagus, colon, or rectum, the ingestion of more than 2 packets (24mg of sucralose) or tablets of the NCS per day vs. none, showed no increase in the risk for those cancers. Similar results were found for colon cancer and rectal cancer for consumers of other NCSs, especially aspartame, vs. nonconsumers. The authors concluded that there was no statistically significant evidence to sustain that saccharin or aspartame increased the risk for cancer in humans.57
A 2017 review by Lohner on the possible positive or negative health effects of NCSs on healthy subjects included 372 studies. Fifty-one primary studies evaluated the association between NCS ingestion and the risk for cancer. Of the study total, only four were case-control studies that evaluated the risk for cancer in the digestive tract. One of them assessed the risk for colorectal cancer, showing a significant increase with the use of NCSs. However, two studies evaluated the risk for pancreatic cancer, finding no association with NCSs, and another study evaluated the risk for bile duct cancer, finding no significant associations.58
Regarding hepatocellular carcinoma, one study assessed the safety of cyclamate. Twenty-one macaques were given cyclamate at doses of 100mg/kg/day or 500mg/kg/day, from birth up to 24 years. Three of the 21 macaques presented with cancer, one of which was hepatocellular carcinoma. The study employed doses above the ADI for humans (11mg/kg/day) for a long period of ingestion, but despite the cases of liver cancer observed, the evidence did not conclusively show that cyclamate produced a risk for developing hepatocellular carcinoma. As explained by the authors, the incidence of neoplasias observed in the macaques was normal.26
Importantly, the latest International Agency for Research on Cancer (IARC) monograph on the evaluation of carcinogenic risks to humans stated that NCSs are considered high-priority additives for re-evaluation in future studies, because of great public interest and the fact that evidence on the topic is still inconclusive,59 given that studies on rodents have linked NCS ingestion to the development of tumors.60,61
In conclusion, the majority of the scientific evidence up to the present day does not support the carcinogenic potential of NCSs. Nevertheless, due to the increase in the number of studies conducted on different animal models, more epidemiologic and causality studies on humans are needed for a definitive conclusion.
Noncaloric sweeteners in nonalcoholic fatty liver disease and steatohepatitisWith respect to the effect of NCSs on the pathophysiology and progression of nonalcoholic fatty liver disease (NAFLD) and nonalcoholic steatohepatitis (NASH), no studies have been conducted on humans at present. Nevertheless, in a study by Holvoet et al., they evaluated the effect of stevia-derived compounds on lipid accumulation in the liver in mice that were genetically modified to not produce the leptin hormone (OB/OB). The mice were divided into a control group, a group treated with stevioside (10mg/kg/day), a group treated with rebaudioside A (12mg/kg/day), and a group treated with steviol (5mg/kg/day). There was a decrease in the percentage of liver fat in all the groups treated with stevia-derived compounds. Likewise, improvement in the hepatic metabolism of glucose and protection against oxidative stress were observed with the administration of those compounds.62
In their study, Janssen et al. evaluated the effect of glucose, fructose, and aspartame on lipid metabolism through magnetic resonance spectroscopy (MRS) to determine their effect on the development of hepatic steatosis in Wistar rats. The intervention consisted of a solution of 13% glucose, 13% fructose, or 4% aspartame, for 7 weeks. There was a significant accumulation of liver fats in the glucose and fructose groups, induced by an increase in de novo lipogenesis, whereas aspartame did not significantly affect the fat content in the liver.63
In conclusion, there is little evidence on the effect of NCS ingestion on NAFLD and NASH. However, studies on animal models suggest that aspartame does not appear to be associated with the pathophysiology of NASH, whereas stevia derivatives could have a favorable effect on the percentage of liver fat. More research is needed that establishes causality in clinical trials with patients.
Noncaloric sweeteners during the inflammatory process of cirrhosis of the liver and its complicationsCurrently, there is one study on humans and other studies on animal models that evaluate the effects of different NCSs on liver diseases.
In a randomized crossover clinical trial, Hertelendy et al. evaluated aspartame ingestion in 13 patients with chronic alcoholic liver disease. In phase 1 of the trial, 15mg/kg/weight/day of aspartame was added to their regular diet. After a washout period of two days, phase 2 was begun, with the administration of a placebo. The results showed mean aspartame ingestion of 1,154±278mg per day and a content of approximately 650mg of phenylalanine. There was a significant reduction in the branched-chain amino acid (BCAA)/aromatic amino acid (AAA) ratio in the aspartame group. That alteration was not found in the intervention with placebo. There were no increases in the concentrations of ammonia or episodes of clinical encephalopathy immediately after the interventions. In that sense, the study showed the safety of a single dose of aspartame, but it did not evaluate the long-term effect or the effect of sustained aspartame ingestion, given that the decrease in the BCAA/AAA ratio could make those patients more susceptible to hepatic encephalopathy in the medium term and the long term.64 Therefore, the recommendation of aspartame ingestion in patients with chronic liver disease should be made with caution.
Latha et al. conducted the only study at present on acute liver injury in an animal model of the disease induced by lipopolysaccharides (LPSs) in 32 Wistar rats. The effect of the raw leaf of Stevia rebaudiana (500mg/kg) and stevioside (250mg/kg) was evaluated. There was a decrease in aspartate aminotransferase (AST) and in alanine aminotransferase (ALT) with both compounds. In addition, there was an increase in the antioxidant capacity and a decrease in the proinflammatory cytokines with both agents, but the increase was greater in the stevioside group. The conclusion from those results was that both Stevia rebaudiana and stevioside, unpurified and at very high doses, could have a protective effect on the liver, improving the clinical picture of acute liver injury.65 Nevertheless, that dose was significantly higher than the ADI for highly purified stevia extracts in humans and therefore is not equivalent to the stevia that is marketed and approved.
The evaluation of said studies suggests that stevia compounds could have a favorable effect on acute liver injury, with Stevia Rebaudiana providing a greater benefit. However, not only were the doses employed higher than the expected doses in humans, but the type of compound was also different from the highly purified stevioside concentrations in the commercially available and approved preparations.
Different studies on liver injury have been published that were carried out on animal models, evaluating aspartame ingestion and its effects on liver function.
A study on mice analyzed the effect of chronic administration of aspartame on the glutathione redox state and the liver transsulfuration pathway in 3 groups: a control group, a group treated with aspartame (80mg/kg, prepared in a solution of 0.9% NaCl), and a group treated with aspartame plus N-acetylcysteine (163mg/kg, pH 6.8-7.2), for 90 days. The administration of aspartame significantly increased the concentrations of ALT and AST and there was a marked decrease in the hepatic concentrations of reduced glutathione, oxidized glutathione, and γ-glutamylcysteine, as well as a decrease in the metabolites of the transsulfuration pathway, such as cysteine, S-adenosylmethionine, and S-adenosylhomocysteine. In addition, those effects were attenuated in the rats that received aspartame plus N-acetylcisteine.66
Other studies have evaluated the effect of long-term aspartame ingestion on Wistar rats. In the studies that supplemented doses of 75, 500, and 1,000mg/kg weight, the dose of 75mg/kg significantly reduced antioxidant capacity due to the decrease in glutathione and the glutathione-reductase enzyme. The effect was significantly higher at the dose of 500mg/kg weight. At the dose of 1,000mg/kg weight, there was severe glutathione depletion, as well as a significant decrease in glutathione peroxidase (GPX) and glutathione reductase, a significant increase in the transaminases, alkaline phosphatase, and the gamma glutamyl transpeptidase (GGT), as well as leukocyte infiltration into the liver tissue.67,68
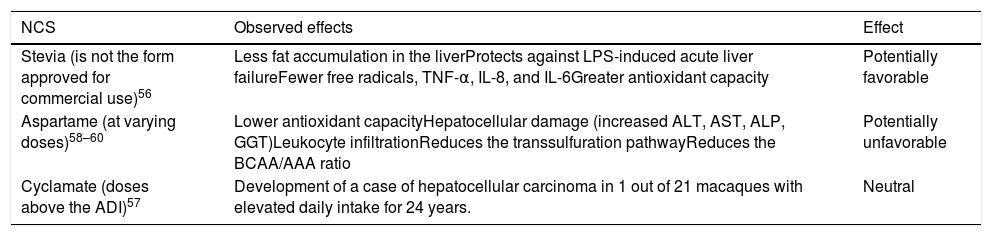
Aspartame is the most widely studied NCS, with consistent results among the different studies conducted on animal models that utilized doses above and below the ADI. Under those conditions, aspartame was consistently observed to reduce the hepatic antioxidant capacity, producing altered liver function tests in animal models. Likewise, even though the results in humans did not show significant clinical manifestations, they did show a decrease in the BCAA/AAA ratio, which could potentially affect patients that are susceptible to hepatic encephalopathy (Table 4).
Effect of different NCSs on the liver. Studies on animals.
| NCS | Observed effects | Effect |
|---|---|---|
| Stevia (is not the form approved for commercial use)56 | Less fat accumulation in the liverProtects against LPS-induced acute liver failureFewer free radicals, TNF-α, IL-8, and IL-6Greater antioxidant capacity | Potentially favorable |
| Aspartame (at varying doses)58–60 | Lower antioxidant capacityHepatocellular damage (increased ALT, AST, ALP, GGT)Leukocyte infiltrationReduces the transsulfuration pathwayReduces the BCAA/AAA ratio | Potentially unfavorable |
| Cyclamate (doses above the ADI)57 | Development of a case of hepatocellular carcinoma in 1 out of 21 macaques with elevated daily intake for 24 years. | Neutral |
ALP: alkaline phosphatase; ALT: alanine aminotransferase; AST: aspartate aminotransferase; BCAA/AAA: branched-chain amino acid/aromatic amino acid; GGT (gamma-glutamyl transpeptidase; IL: interleukin; LPS: lipopolysaccharides; TNF-α: tumor necrosis factor α.
It can thus be concluded that stevia compounds could be partially beneficial in acute liver injury. Aspartame should be recommended with caution in patients with chronic liver disease due to the decrease in the BCAA/AAA ratio, which could potentially affect patients that are susceptible to hepatic encephalopathy.
Noncaloric sweeteners and their interaction with the gut microbiotaThe microbiota of the gastrointestinal tract is composed of 100 trillion microorganisms of at least 1,000 different bacterial species, whose participation is crucial in the physiologic and pathophysiologic processes of the host.69,70 The gut microbiota outnumbers human cells and it contains 100-times more genes than the human genome.71,72
The type and number of gut bacteria are altered by different mechanisms, which include dietary composition73 and the wide range of diets in each region of the world.74
The population of microorganisms is now known to have numerous functions in the intestine, such as promoting the maturation and integrity of the intestinal epithelium, protection against pathogens, immunologic modulation, intestinal immunologic balance, and the prevention of inflammation.71 Its alteration has been linked to different diseases, including food allergies, inflammatory bowel disease, necrotizing enterocolitis, obesity, fatty liver, and colon cancer, among others.72
Studies have evaluated the relation between diet and the gut microbiota, leading to the analysis of the effect of NCS ingestion on the gut microbiota.75 However, there have been limitations regarding measurement techniques, doses employed, diet, and the determination of its metabolites, such as the short chain fatty acids (SCFAs).21 Therefore, the use of methods that better represent bacterial diversity, such as bacterial (microbiome) DNA sequencing, are preferred, to obtain a more complete microbiologic profile.76
Until a few years ago, NCSs were considered metabolically inert, with no apparent physiologic effects. However, some of them undergo numerous changes in the intestine, interacting with the microbiota, thus modifying its metabolites in the different areas of the intestine77 (Table 5).
NCSs and the biologic plausibility of their significant presence in the colon.
| Noncaloric sweeteners | According to their digestion and absorption, greater interaction with the microbiota |
|---|---|
| Acesulfame K | No |
| Aspartame | No |
| Saccharin | Yes |
| Steviol glycosides | Yes |
| Sucralose | Yes |
Adapted from Magnuson et al.24
Each of the sweeteners and its possible interaction with the microbiota are listed below.
Acesulfame-K: A randomized controlled trial on animal models showed that the ingestion of acesulfame-K (37.5mg/kg/day for 4 weeks) triggered significant changes in the gut microbiota. In that study, there was a division by sex, showing an increase in Bacteroides and Anaerostipes in males and a decrease in Lactobacillus, Clostridium, an unassigned Ruminococcaceae genus, and an unassigned Oxalobacteraceae genus in females. It should be mentioned that the concentration employed in that study was higher than the ADI in humans (higher than 15mg/kg/day).78 In a study conducted on humans (a cross-sectional study) that included 31 volunteers, no modifications in the gut microbiota or significant differences in the relative abundance by sexes were shown between acesulfame-K consumers and nonconsumers.79 Nevertheless, in a review by Nettleton et al., there were changes in the microbiota and in the metabolites it produces, after acesulfame-K ingestion, specifically on the SCFAs, pyruvate and butyrate. The authors did not specify the mechanism by which the microbiota was altered. One hypothesis is that it takes place through systemic metabolites derived from the metabolome of specific bacteria that interact with the additive.11,80 However, in a study conducted on mice that included a group treated with a solution of acesulfame-K at 15mg/kg of weight, there were no differences in the abundance of bacteria in feces, including Firmicutes and Bacteroidetes. The authors concluded that there were no significant differences in the microbiota.81
Bian et al. demonstrated an association between acesulfame-K genotoxicity and the inhibition of glucose fermentation by intestinal bacteria. Acesulfame-K also exerts antimicrobial activity because it belongs to the chemical class of sulfonamides. Thus, acesulfame-K could significantly impact the content and microbial diversity of the gut microbiota.78 In elevated doses it modifies the bacterial composition in animal models.
Aspartame: Aspartame is hydrolyzed into phenylalanine, aspartic acid, and methanol. In regular consumption, only 10% of aspartame (by weight) is converted into methanol.82 Some studies have shown that methanol affects the growth of anaerobes and elevated concentrations completely inhibit the growth of Escherichia coli (E. coli), anaerobes, and facultative microorganisms present in the human microbiota.83 A study on animal models (44 male rats) showed that a high-fat diet plus aspartame increased the proportion of Firmicutes and Clostridium cluster CXI. Specifically the addition of aspartame modified the proportion of Firmicutes - Bacteroidetes, due to an increase in Firmicutes, and particularly in the Clostridium cluster CXI.84 Another study on animal models showed that low doses of aspartame modified serum metabolites, such as lysine, serine, glycine, propionate, creatine, 3-hydroxybutyrate, methanol, glycerol, and urea. That same study demonstrated that aspartame metabolism was associated with increased concentrations of propionate (a SCFA). However, in humans, aspartame has not been shown to modify glucose tolerance or insulin concentrations. Likewise, aspartame caused significant changes in the microbiota, such as an increase in Enterobacteriaceae and Clostridium leptum, modulation in the increase in Firmicutes and Bacteroidetes, and the increase in Roseburia ssp. Unlike other studies that utilized doses above the ADI, the effects found in that study were with low doses of aspartame (approximately 5-7mg/kg/day).84
In conclusion, aspartame releases a molecule of methanol that has the effects on the growth of anaerobes and E. coli described above, but the systemic, long-term effect of regular aspartame ingestion is unknown. Nevertheless, given that it can modify the composition of the microbiota, aspartame should be recommended with caution in patients with a chronic liver disease. More studies are needed to establish its implications in the development of metabolic diseases.
Saccharin: Saccharin is one of the most widely used sweeteners by the industry and has been analyzed in the largest number of studies. In a study utilizing adult male C57BL/6 mice conducted by Suez et al., saccharin, sucralose, and aspartame ingestion, compared with water, glucose, or sucrose, produced glucose intolerance in both the thin and obese animals. Through fecal microbiota transplantation of mice that were fed saccharin or glucose into germ-free mice, the researchers showed that the germ-free mice that received the fecal transplant from the group that consumed saccharin developed glucose intolerance, compared with those that received the fecal transplant from the group that was fed glucose. The authors suggest that the effects of the NCSs on metabolic parameters, such as insulin resistance, are partially due to the induced changes in the gut microbiota.20
The same group of researchers analyzed the effect of saccharin on glycemic control in 7 healthy volunteers. Four of the seven subjects showed glucose intolerance at the maximum ADI of saccharin (5mg/kg) in 3 divided daily doses. The authors concluded that the effect of NCSs on the heterogeneity of the human gut microbiota could make some individuals more vulnerable to developing glucose intolerance, after saccharin ingestion.20 That study has been criticized by some authors because in its conclusion sections, Suez et al. state that all sweeteners affect the microbiota, producing glucose intolerance, but their study mainly evaluates saccharin.
In addition, other authors have demonstrated that many of the bacterial genera and species found in rodents are not present in humans.21 In their study, Bian et al. evaluated the effect of saccharin ingestion on a murine model of 8-week-old C57BL/6J mice. The intervention group received 0.3mg/ml of saccharin dissolved in water, for 6 months, whereas the control group received only water. Effects on the microbiota and modifications in different bacterial genera were observed at 3 and 6 months, as well as increased levels of the iNOS and TNFα genes, through the examination of proinflammatory gene expression at the mRNA level.29
In a study on a porcine model by Daly et al., they studied two groups. Both groups were on a standard diet and saccharine was added to the diet of one group. There was a significant increase in the Ruminococcaceae and Veillonellaceae families, and in particular the Lactobacillaceae family, in the group with the diet that included saccharin. However, the study did not quantify food ingestion or state the dose of saccharin.21,85
Sucralose: In their study on mice, Donia et al. reported a significant decrease in anaerobes, bifidobacteria, lactobacilli, and Bacteroides, as well as a reduced abundance of clostridia and the total aerobic bacteria, but the sweetener utilized (Splenda) contained 99% maltodextrins and dextrose and only 1% pure sucralose.86,87 In addition, Uebanso et al. evaluated the effect of sucralose and acesulfame-K on the gut microbiota and metabolism for 8 weeks in three groups of mice (a group with sucralose at 1.5mg/kg of weight, a group with sucralose at 15mg/kg of weight, and a control group). They found no differences in the quantity of fecal bacteria or in the abundance of Firmicutes and Bacteroidetes. However, there was a significant sucralose dose-dependent decrease in the abundance of Clostridium cluster XVIa and butyrate production.81
Stevia: Steviol glycosides are the final absorbable product of bacterial metabolism in the colon.76,88 Those glycosides are not absorbed in the small intestine but are hydrolyzed by gut microbiota bacilli of the Bacteroides group.89,90
There is little information about the effect of those metabolites on the balance of the microbiota. Nevertheless, some studies have shown that complete stevia extracts have antimicrobial properties that can influence the population of the gut microbiota. Those changes have produced alterations in enzyme digestive activity, the production of SCFAs, and the health of the animals.89,91 Other studies have also shown that steviol glycosides inhibit the in vitro growth of Lactobacillus reuteri.92 However, at present, those results cannot be extrapolated to the commercial presentations that contain highly purified steviol glycosides.
Polyols: Different studies have analyzed the effect of polyols on the gut microbiota. However, each polyol contains individual characteristics that can produce different intestinal manifestations associated with its consumption (Table 3).42,87,93–95
Currently, the most widely-studied polyols are isomalt, lactitol, maltitol, xylitol, and erythritol, and studies on both animal models and humans have shown that the part of those NCSs that is nonabsorbable in the small bowel, reaches the colon integrally and is rapidly fermented by the action of the microbiota, producing SCFAs, CO2, CH4, and H2.34 Some polyols, such as lactitol and erythritol, have greater interaction with the gut microbiota that is associated with digestive alterations. In the case of lactitol, the small quantities of hydrogen and SCFAs that it produces are utilized as a source of energy for Bifidobacterium and Lactobacillus spp, which lowers the intestinal pH, as well as the production and absorption of ammonia (NH3).42,96–98 Erythritol inhibits Streptococcus mutans growth42,99 and is poorly fermentable by the gut microbiota, which could give it prebiotic qualities.99
With those results, it can be concluded that NCSs potentially have the capacity to modify the composition of the gut microbiota, which could have an effect on some gastrointestinal manifestations. Nevertheless, it is important for more studies to be conducted to establish their implications in the development of diseases related to those changes.
ConclusionsThere is no clinical evidence of a possible inflammatory effect on the intestine caused by NCSs.
NCSs do not cause gastrointestinal alterations but sweeteners that provide volume and texture, such as polyols, can cause symptoms and digestive disorders, mainly diarrhea and bloating, depending on the type of compound and its dose.
High-intensity NCSs do not directly affect intestinal motility.
No carcinogenic potential of NCSs has been demonstrated.
Studies conducted on animal models suggest that aspartame has no influence on the pathophysiology of NAFLD, whereas stevia derivatives appear to have a favorable effect on the percentage of liver fat. However, more studies are needed to establish that possible association.
In patients with chronic liver disease, aspartame should be recommended with caution in those patients susceptible to developing hepatic encephalopathy, until better quality clinical evidence is available.
Studies on experimental models have shown changes in the composition of the gut microbiota associated with NCS ingestion.
More clinical trials are needed to define whether those changes have an impact on the human gut microbiota and their consequences in relation to digestive health.
Financial disclosureThe Asociación Mexicana de Gastroenterología obtained non-conditional financing for logistic purposes from Heartland Consumer Products México and from the Instituto de Bebidas de Coca-Cola de México. The scientific agenda, discussion, and conclusions stated in the present document were established autonomously and drafted independently by the members of the working group.
Conflict of interestN Bueno-Hernández is a speaker for Carnot, Abbvie, and Takeda. R. Vázquez-Frias is a speaker for and has received research funding from Alexión, BioGaia, Carnot, Mayoly-Spindler, Nestlé, and Sanofi. A.T. Abreu y Abreu is an advisor and speaker for Laboratorios Sanofi, AB Biotics, Takeda, Mayoly-Spindler, Carnot, MD Pharma, Menarini Biocodex, Columbia, and Abott. P. Almeda-Valdés is a speaker for Boehringer Ingelheim, Eli-Lilly, Sanofi, AstraZeneca and an advisor for Boehringer Ingelheim, Sanofi, Abbot, and Servie. R.I. Carmona-Sánchez is an advisor for Asofarma and a speaker for Mayoly-Spindler, Asofarma, and Chinoin. A. Consuelo-Sánchez is an advisor for Laboratorios Sanofi and a speaker for Sanofi and Mead Johnson Nutrition. He provides research support and is a scientific advisor for Alexion Pharmaceuticals Inc. V. Hernández-Rosiles is a speaker for Nestlé. M.E. Icaza-Chávez is a speaker for Asofarma and Takeda. A. Noble-Lugo is a speaker for Asofarma and Takeda. A. Ruiz-Margaín is a speaker for Victus and provides research support for Novartis. M.A. Valdivinos-Díaz is an advisor for Biocodex, Sanofi, Takeda, a speaker for Takeda, Ferrer, Mayoly-Spindler, Columbia, Biocodex, and Menarini, and he has received funding for academic development from Takeda. F.E. Zárate-Mondragon is an advisor and speaker for Mead Johnson. A. Romo-Romo, L.A. Barajas-Nava, and A.J. Espinosa-Flores declare that they have no conflict of interest.
Please cite this article as: Bueno-Hernández N, Vázquez-Frías R, Abreu y Abreu AT, et al. Revisión de la evidencia científica y opinión técnica sobre el consumo de edulcorantes no calóricos en enfermedades gastrointestinales. Revista de Gastroenterología de México. 2019;84:492–510.