Neuroendocrine tumors are of great scientific interest, given that they are difficult to diagnose and treat. Despite being relatively rare (<1/100,000 individuals, 1-2% of the gastrointestinal neoplasias) and indolent, their potential malignancy must not be forgotten. An increase in the number of diagnosed tumors has been observed in recent years. The aim of the present study was to update a published case series of 19 patients suspected of presenting with pancreatic neuroendocrine tumor with 51 current cases, to study and compare the new results with those of the previous case series, as well as with other recent publications from Spain, the United States, China, and India.
Materials and methodsA retrospective, multicenter case series was conducted on 70 patients (19 cases published in 2011), whose data has been collected over a period of 23 years. The variables analyzed were: age, sex, symptomatology, tumor size, location, metastasis, final diagnosis, and surgery, among others.
ResultsMean patient age was 55 years and 60% of the patients were men. Disease location was the pancreatic head in 28.5% of the patients and the tail in 27.1%, mean tumor size was 3.9cm (0.2-10cm), 71.4% of the patients had non-functioning tumors, 32.8% had metastases (100% to the liver), 74.2% of the patients were operated on, and actuarial survival was 75%.
ConclusionsDifferences were observed between the previously published case series and the current results. There was an increase in incidentalomas and non-functioning tumors, but no variation in the overall survival rate. The differences with other case series (age, sex, and tumor location) were dependent on the country where the cases were compiled. The increase in tumors could be related to a higher number of diagnoses made through imaging studies and to the greater sensitivity of the devices employed.
Los tumores neuroendocrinos generan un gran interés científico, dada la dificultad de su diagnóstico y tratamiento. A pesar de ser relativamente raros (<1/100,000 individuos, 1-2% de las neoplasias digestivas) e indolentes, no se debe olvidar que, en última instancia, son potencialmente malignos. En los últimos años, se ha observado un incremento en el número de tumores diagnosticados.
El objetivo del estudio fue la actualización de una serie publicada de 19 casos con sospecha de tumor neuroendocrino pancreático, con otros 51 casos actuales, para el estudio y comparación de los nuevos resultados tanto con la serie anterior como con otras publicadas recientemente en España, Estados Unidos, China e India.
Material y métodosSerie retrospectiva, multicéntrica de 70 pacientes (19 publicados en 2011) recopilados a lo largo de 23 años. Variables analizadas: edad, sexo, sintomatología, tamaño, localización, metástasis, diagnóstico final y cirugía, entre otras.
ResultadosEdad media de 55 años; 60% hombres; localización: cabeza (28.5%) y cola (27.1%); tamaño medio 3,9cm (0,2-10cm); no funcionantes el 71.4%; metástasis en el 32.8% (100% hepáticas), operados el 74.2% y supervivencia actuarial del 75%.
ConclusionesSe observan diferencias entre la serie anteriormente publicada y los resultados actuales, con un incremento del porcentaje de incidentalomas y de tumores no funcionantes, pero sin variación en la tasa de supervivencia global. Con otras series actuales, las diferencias (edad, sexo y localización tumoral) dependen del país donde se han recopilado los casos. Este incremento podría relacionarse con el aumento del diagnóstico por la imagen y con la mayor la sensibilidad de los dispositivos.
The first description of pancreatic neuroendocrine tumor (PNET) was made by Wilder et al. in 1927 in a clinical case published in JAMA.1 They are rare tumors with an estimated detection rate < 1/100,000 individuals2, corresponding to 1-2% of the pancreatic neoplasias.3 However, they are of great scientific interest due to the difficulty of their diagnosis and treatment. The majority of those tumors are asymptomatic and are incidental findings in at least 0.5-1.5% of autopsies.2
Most PNETs are sporadic, but they can be associated with hereditary syndromes, including multiple neuroendocrine neoplasia type 1 (MEN1), von Hippel-Lindau syndrome, neurofibromatosis type 1, and tuberous sclerosis.2
PNETs are divided into 2 large groups: functioning tumors that are associated with a syndrome secondary to the ectopic secretion of biologically active substances, and non-functioning tumors (NF-PNETs)4 that are not associated with a syndrome and make up 50-75% of all PNETs.1
PNETs tend to be indolent, but their potential malignancy should not be forgotten. They are slow-growing and have a better prognosis than ductal tumors of the pancreas, even when they present with metastasis. The best treatment is surgery, when the tumor is resectable.5
Overall 5-year survival rates vary at 30% for NF-PNETs and 97% for benign insulinomas, a type of functioning PNET.6 An increase in incidence has been detected in recent years, like that occurring in the United States over the past 2 decades, that is most likely due to the incidental detection of asymptomatic tumors in imaging and endoscopic studies performed when searching for other diseases.4
The aim of the present study was to update a previously published case series on 19 cases suspected of having PNET7 with 51 other current cases, to study and compare the new results with that previous study and similar recently published studies conducted in Spain,8–10 the United States,11 Japan,12 China,11,13 and India.14
Materials and methodsA retrospective, multicenter study was conducted that included 70 patients with PNET that was detected either incidentally or during the evaluation of abdominal pain and diarrhea (19 of which were published in a previous case series). The data were collected over a period of 23 years (from 1994 to 2017).
To conduct the study, the medical records of the patients were reviewed and the established protocols for accessing data and managing clinical records were followed. The research team was accredited by the Department of Information Security to gain access to the clinical history data through username and password.
The variables analyzed were: age, sex, symptomatology, imaging methods employed, number of tumors, location, presence of metastasis, surgery as treatment, postoperative complications, complementary treatments, survival, and definitive diagnosis.
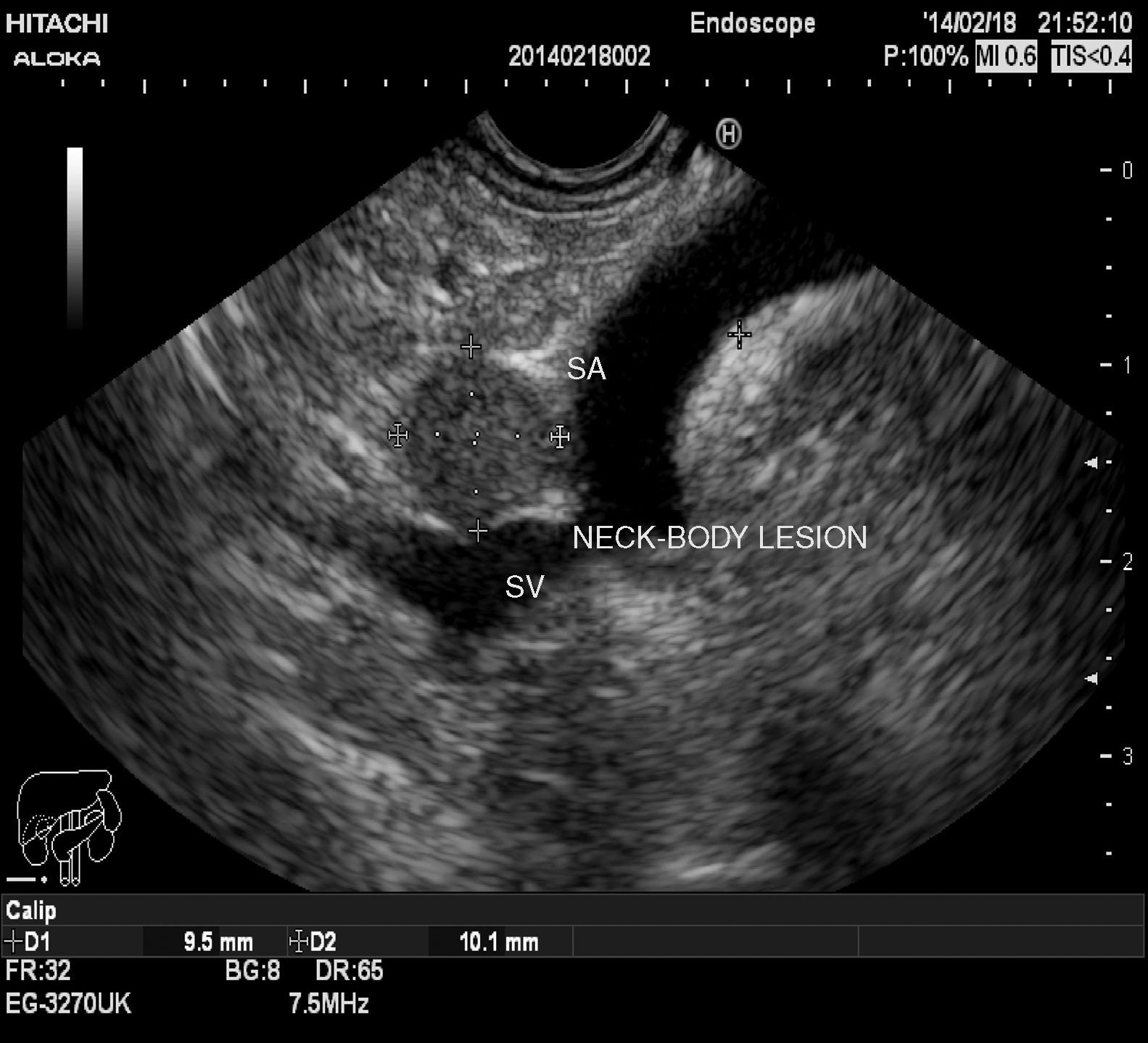
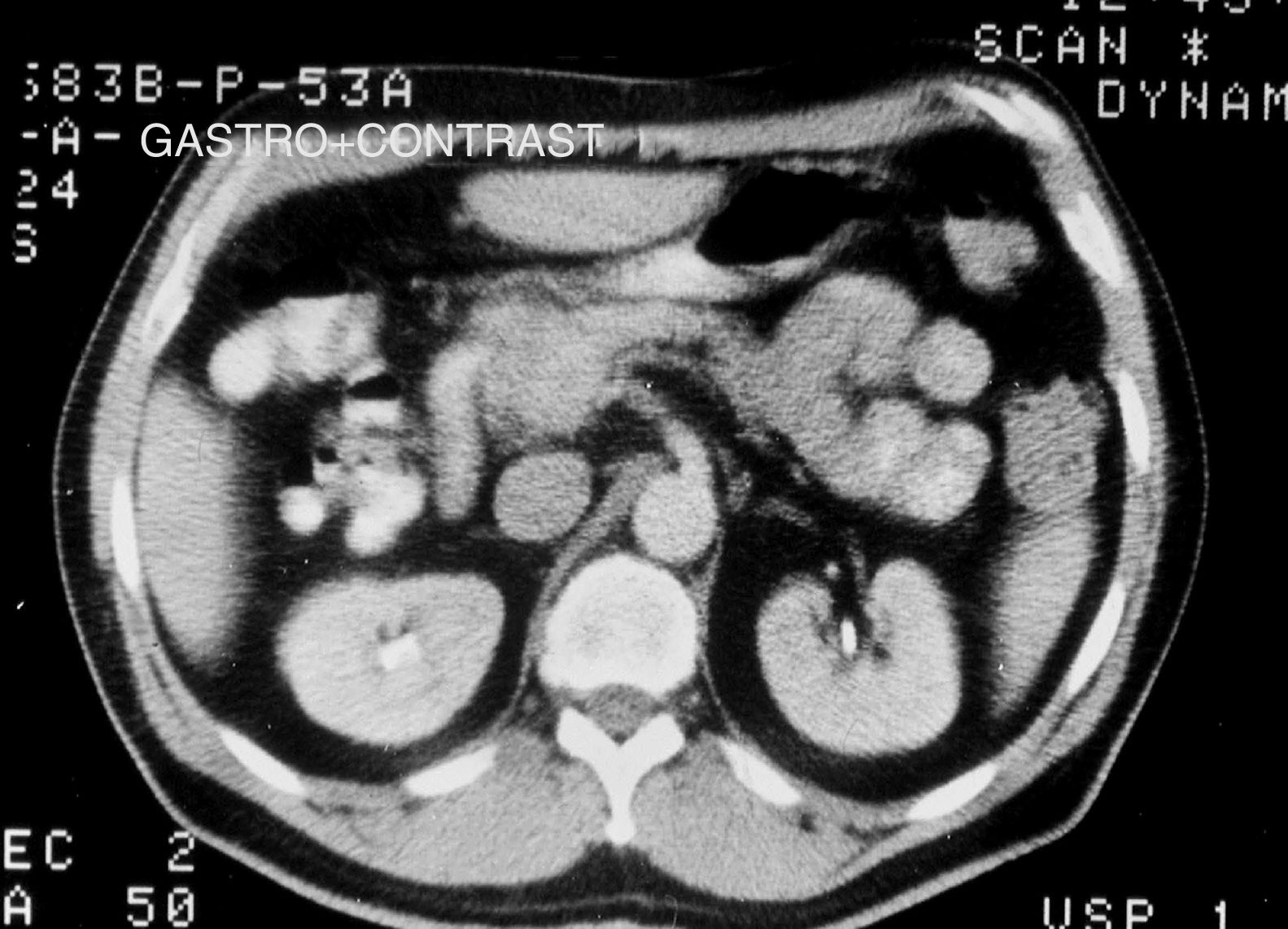
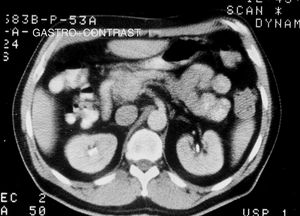
Tumors were detected and diagnosed through imaging techniques and laboratory tests. The gold standard for making the diagnosis was pathologic anatomy. Of the imaging studies used, conventional abdominal ultrasound (US), endoscopic ultrasound (EUS), and computed axial tomography (CAT) (figs. 1 and 2) were the studies through which the majority of incidental tumors were detected. Abdominal CAT, magnetic resonance imaging (MRI), and endoscopic ultrasound-guided fine needle aspiration (EUS-FNA) were utilized for the definitive diagnosis.
EUS-FNA was performed in the cases of non-functioning tumors or those in which there was doubt about the tumor. The 22-G needle was used in the majority of the cases, with 1-3 passes, and a mean of 2 biopsies per tumor.
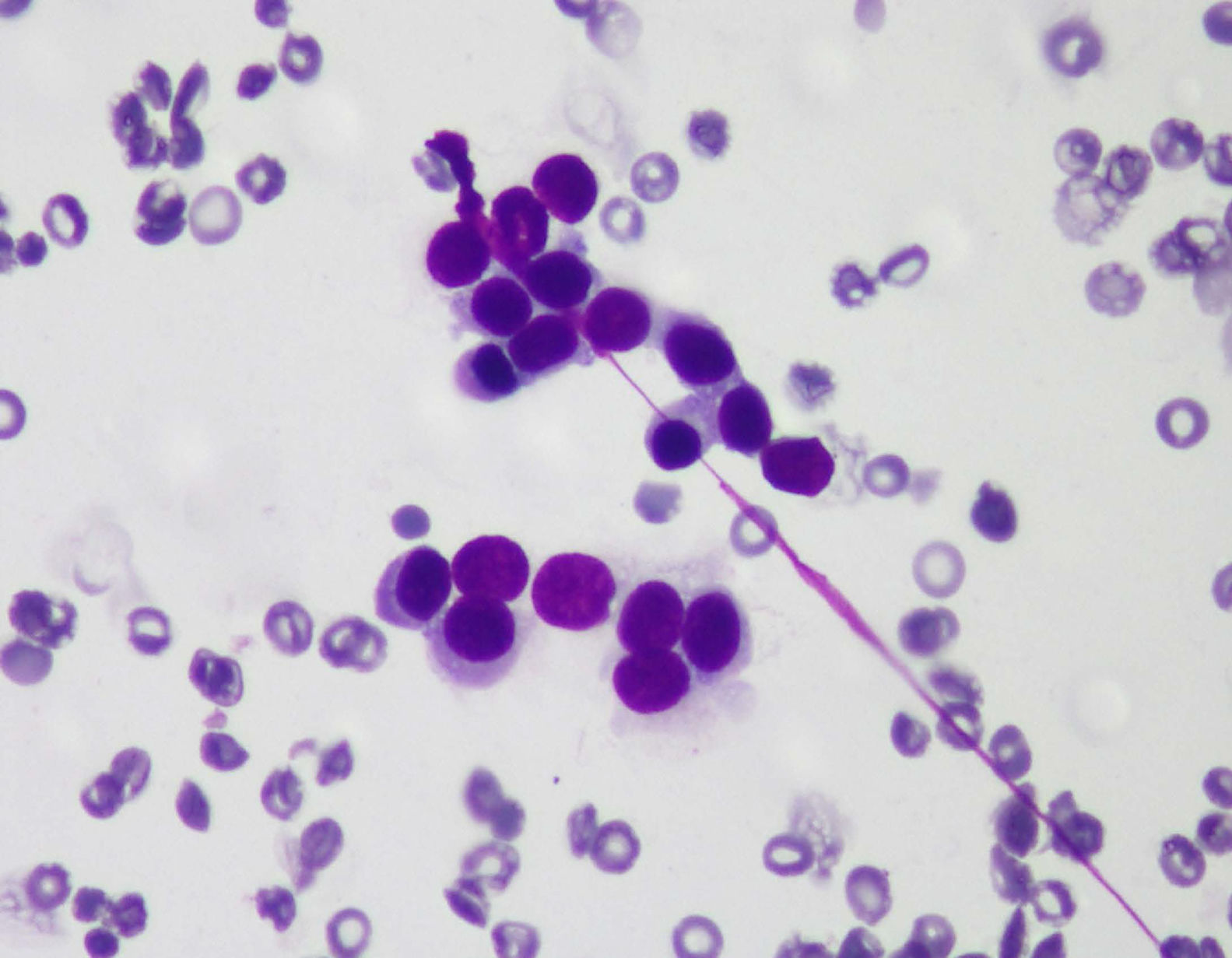
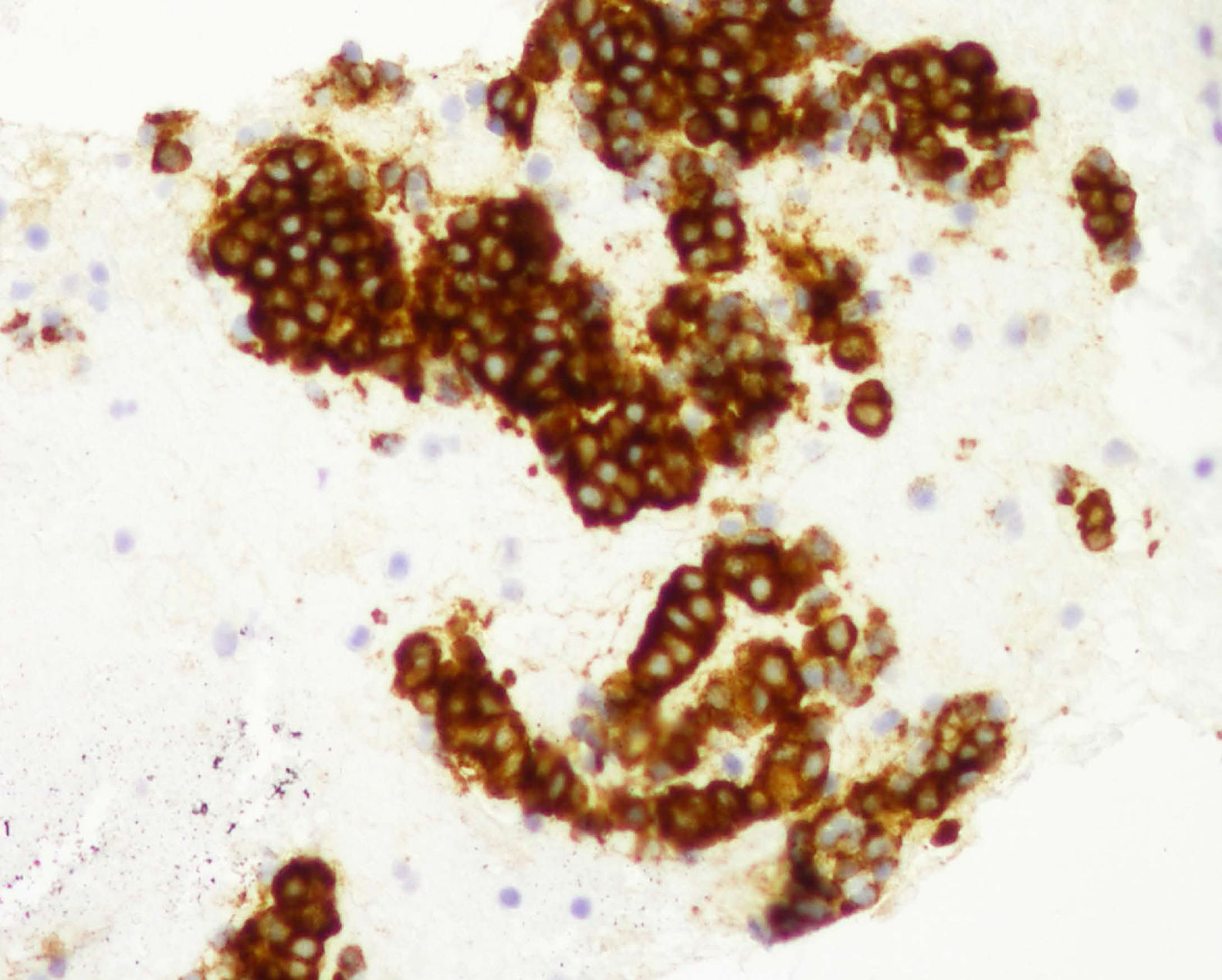
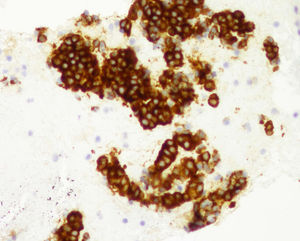
Cytology and immunocytochemistry studies were performed on the samples obtained through EUS-FNA, carrying out histologic staining for chromogranin A (fig. 3), neurospecific enolase, gastrin, insulin, vasoactive intestinal peptide, pancreatic polypeptide, and serotonin. The ki-67 index (K2 Leica Microsystems, Barcelona, Spain) was evaluated to obtain tumor grade (fig. 4). The number of tumor cells with nuclear staining was > 500 cells in each section (X400). The 2010 WHO guidelines were followed for the ki-67 analysis, and were based on ki-67 index value and the mitotic count, classifying the tumors into well-defined PNET, or grade 1 (G1) and grade 2 (G2) tumors, with a solid, trabecular pattern or a glandular pattern, and poorly-defined PNET, or grade 3 (G3), which facilitated making the decisions for the best treatment and follow-up for the patient.
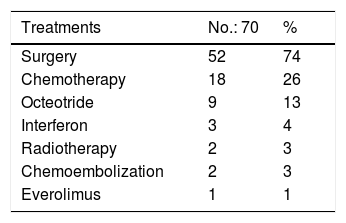
Treatments were laparoscopic or open surgery, chemotherapy, octeotride, interferon, radiotherapy, chemoembolization, or everolimus, according to the type, location, and grade of the tumor, as well as the presence of metastasis.
When the tumor was resectable and the clinical and morphologic characteristics were coherent, according to CAT plus EUS imaging, laparoscopic or open surgery was performed in accordance with the characteristics of each case. Because the present study was retrospective and covered a 23-year period, the first cases were treated through open surgery and all the cases corresponding to the later years were resolved through laparoscopic surgery.
Statistical analysisThe Windows SPSS 11.5 statistical package was utilized to carry out the data analyses. Median, mean, standard deviation, and percentages were calculated through specific formulas.
Ethical principlesThe study was conducted according to the principles of the Declaration of Helsinki (the latest version) and the Good Clinical Practice guidelines.
ResultsA total of 70 cases were studied and the results of the variables analyzed were: mean age at disease presentation, 55 ± 14 years (range: 13-84 years); by sexes: 60% (42/70) men and 40% (28/70) women. Ninety percent (18/20) of the patients with functioning tumors presented with abdominal pain and diarrhea, PNET was suspected, and imaging studies were carried out to locate those tumors. Twenty percent (4/20) of the patients had hypoglycemia and all of the patients that presented with incidentalomas (28/70, 40%) were asymptomatic.
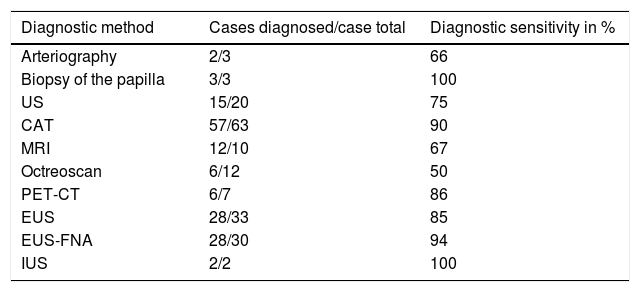
In relation to the imaging techniques, abdominal CAT was employed to locate the tumor in symptomatic patients and it was an incidental finding in the asymptomatic patients. Sixty-three cases (90% of the case series) had abdominal CAT and its sensitivity was 90% (57/63) (fig. 2). EUS-FNA was performed on 43% (30/70) of the patients, with a diagnostic sensitivity of 94% (28/30) and EUS had 85% (28/33) sensitivity (Table 1). EUS-FNA was utilized in the cases in which there was diagnostic doubt (30/70, 43%). Cytology and immunocytochemistry studies were performed on the samples obtained through EUS-FNA and the ki-67 index was evaluated in 80% (24/30) of those cases: diagnostic sensitivity was 83.3% (20/24) and the concordance of the surgical specimen with the ki-67 index was also 83.3% (10/12 specimens).
Diagnosis through the imaging study employed in the 70-patient case series.
| Diagnostic method | Cases diagnosed/case total | Diagnostic sensitivity in % |
|---|---|---|
| Arteriography | 2/3 | 66 |
| Biopsy of the papilla | 3/3 | 100 |
| US | 15/20 | 75 |
| CAT | 57/63 | 90 |
| MRI | 12/10 | 67 |
| Octreoscan | 6/12 | 50 |
| PET-CT | 6/7 | 86 |
| EUS | 28/33 | 85 |
| EUS-FNA | 28/30 | 94 |
| IUS | 2/2 | 100 |
CAT: computed axial tomography; EUS: endoscopic ultrasound; EUS-FNA: endoscopic ultrasound-guided fine needle aspiration; IUS: intraoperative ultrasound;
MRI: magnetic resonance imaging; PET-CT: positron emission tomography-computed tomography; US: abdominal ultrasound.
Mean tumor size was 3.9cm (range 0.2-10). The number of study patients was 70 and the number of tumors was 70. Tumor location was the head of the pancreas in 20 cases (28.5%), the tail in 19 cases (27.1%), the neck-body in 16 cases (22.8%), the uncinate process in 5 cases (7.1%), the gastroduodenal wall in 5 cases (7.1%), and the papilla in 3 cases (4.2%). The tumor could not be located in 2 cases (2.8%), having utilized US, CAT, MRI, and EUS.
Six of the lesions were cystic tumors (6/70, 8.5%) and 23 tumors (23/70, 32.8%) metastasized to the liver.
The most widely-used treatment was laparoscopic surgery or an open procedure, carried out in 74.2% of the patients (52/70). There were postoperative complications in 13.4% of those patients (7/52) that included fistulas, pancreatitis, pseudocysts, and abdominal collections. Enucleation was performed in the insulinomas and no complications were reported. The patients with metastatic disease (23/70, 32.8%) received medical treatment. A total of 25.7% of the patients (18/70) were treated with chemotherapy, 12.8% (9/70) with octeotride, 4.2% (3/70) with interferon, 2.8% (2/70) with radiotherapy, 2.8% (2/70) with chemoembolization (one of those patients before surgery [1/2, 50%] and one after surgery [1/2, 50%]), and 1.4% received everolimus (1/70) (Table 2).
Mean follow-up time was 36 months and survival at the end of the study was 75.7% (53/70). The patients that did not undergo surgery (25.7%, 18/70) had follow-up every six months. The patients that died (24.2%, 17/70) first presented with obstructive jaundice or metastatic disease.
Three groups were defined in the definitive diagnosis:
- -
NF-PNET: 71.4% (50/70) of the cases.
- -
Functioning PNET: 28.5% (20/70) of the cases, 5.7% (4/20) of which were insulinomas, 15.7% (11/20) resulted in Zollinger-Ellison syndrome due to gastrinomas, 2.8% (2/20) were glucagonomas, 1.4% (1/20) were somatostatinoma, and 4.2% (3/20) were carcinoid disease (one patient had a carcinoid-like syndrome). A total of 2.8% (2/70) of the tumors secreted multiple hormones.
- -
MEN1: 11.4% (8/70): 2 cases were non-functioning tumors and 6 cases were functioning tumors (one insulinoma, 4 gastrinomas, one glucagonoma).
Forty percent (28/70) of the tumors were incidentalomas.
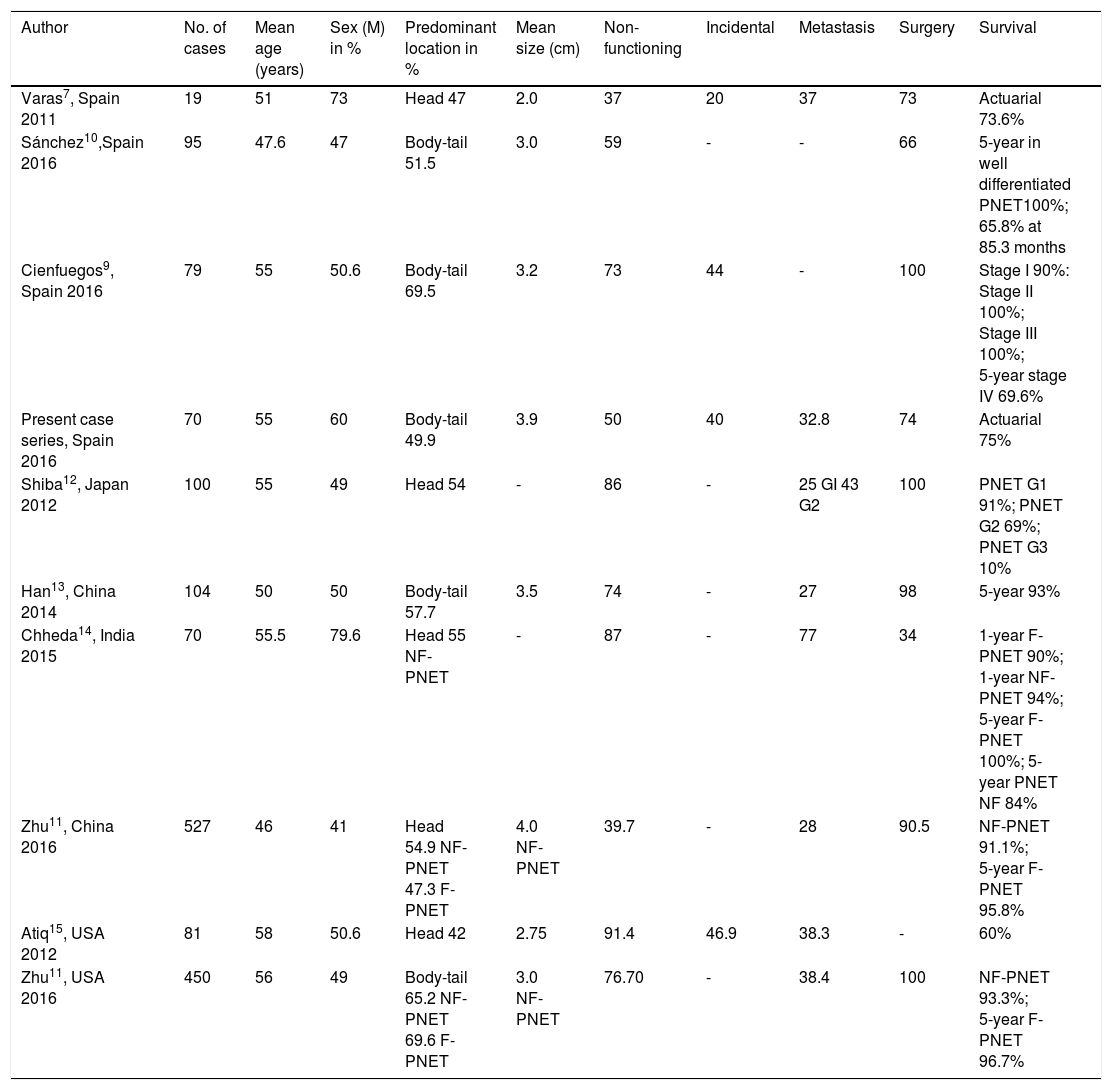
Finally, 9 patients (9/70, 12.8%) had tumors associated with other benign or malignant tumors. Concordance between initial suspicion and diagnostic confirmation of PNET was 87.1% (61/70). Table 3 compares the results from different case series and the data analysis is described in the Discussion section.
Case series comparison with the present case series.
| Author | No. of cases | Mean age (years) | Sex (M) in % | Predominant location in % | Mean size (cm) | Non- functioning | Incidental | Metastasis | Surgery | Survival |
|---|---|---|---|---|---|---|---|---|---|---|
| Varas7, Spain 2011 | 19 | 51 | 73 | Head 47 | 2.0 | 37 | 20 | 37 | 73 | Actuarial 73.6% |
| Sánchez10,Spain 2016 | 95 | 47.6 | 47 | Body-tail 51.5 | 3.0 | 59 | - | - | 66 | 5-year in well differentiated PNET100%; 65.8% at 85.3 months |
| Cienfuegos9, Spain 2016 | 79 | 55 | 50.6 | Body-tail 69.5 | 3.2 | 73 | 44 | - | 100 | Stage I 90%: Stage II 100%; Stage III 100%; 5-year stage IV 69.6% |
| Present case series, Spain 2016 | 70 | 55 | 60 | Body-tail 49.9 | 3.9 | 50 | 40 | 32.8 | 74 | Actuarial 75% |
| Shiba12, Japan 2012 | 100 | 55 | 49 | Head 54 | - | 86 | - | 25 GI 43 G2 | 100 | PNET G1 91%; PNET G2 69%; PNET G3 10% |
| Han13, China 2014 | 104 | 50 | 50 | Body-tail 57.7 | 3.5 | 74 | - | 27 | 98 | 5-year 93% |
| Chheda14, India 2015 | 70 | 55.5 | 79.6 | Head 55 NF-PNET | - | 87 | - | 77 | 34 | 1-year F-PNET 90%; 1-year NF-PNET 94%; 5-year F-PNET 100%; 5-year PNET NF 84% |
| Zhu11, China 2016 | 527 | 46 | 41 | Head 54.9 NF-PNET 47.3 F-PNET | 4.0 NF-PNET | 39.7 | - | 28 | 90.5 | NF-PNET 91.1%; 5-year F-PNET 95.8% |
| Atiq15, USA 2012 | 81 | 58 | 50.6 | Head 42 | 2.75 | 91.4 | 46.9 | 38.3 | - | 60% |
| Zhu11, USA 2016 | 450 | 56 | 49 | Body-tail 65.2 NF-PNET 69.6 F-PNET | 3.0 NF-PNET | 76.70 | - | 38.4 | 100 | NF-PNET 93.3%; 5-year F-PNET 96.7% |
M: male; F-PNET: functioning pancreatic neuroendocrine tumor; NF-PNET: non-functioning pancreatic neuroendocrine tumor.
The present case series analyzed included 19 patients that were reported on in a case series published in 2011.7 The idea of updating the previous case series with new patients came about due to the increase in PNET diagnoses observed in recent years. The intention was to evaluate whether there was any change in the more recently diagnosed tumors that would make the results vary. Fifty-one (71.43%) of the 70 patients included in the present case series were diagnosed within the past 8 years, representing 34.8% of the 23-year data collection period. The increase observed in the PNETs coincides with that reported in published studies.8
That increase could be favored by the expanding availability of imaging techniques, such as ultrasound, commonly used as examination methods in medical consultations, as well as the greater use of imaging studies and endoscopy as screening methods for other diseases. PNET is often detected as an incidental finding in studies such as those. Its definitive diagnosis should be made through more complex imaging techniques, such as abdominal CAT or MRI. Uncertain cases require cytologic or immunocytochemical studies for confirmation and grading, and the sample should be obtained through EUS-FNA.
Upon comparing our present case series with the one published in 2011, we found that the percentage of incidentaloma doubled: it was 20% in the first series and 40% in the present series. We also observed an increase in the percentage of non-functioning tumors diagnosed (from 37% in the 2011 case series to 71.4% in the present series). Those data support the explanation that the increase in the number of tumors diagnosed is due to the increase in incidental findings, the majority of which are asymptomatic, non-functioning tumors. What was not observed, was an increase in the actuarial survival rate. The percentages were very similar in both case series: 74 and 75.7%, respectively. Comparing our study with other case series also published in Spain, the study by Cienfuegos et al.9 showed an even higher percentage of incidentalomas (44%) than our study, and the variables of age and predominant sex concurred with our results. Mean tumor size was somewhat smaller in their study (3.2 vs. 3.9cm) and there was also a greater predominance of tumors located in the neck-tail of the pancreas (69.5 vs. 49.9). However, when we compared both case series with the 2016 case series by Sánchez et al.,10 the results did not coincide. In that case series, the mean age was lower (47 vs. 55 and 55 years, respectively), there was a slight difference by sexes (47% men), and a lower percentage of non-functioning tumors (59 vs. 73 and 71%), but primary tumor location in the body-tail was similar to the results in our series (51.5 vs. 69.5 and 49.9%) (Table 3).
Upon comparing our case series with those conducted in the United States, we found that mean age was similar (56 and 58 years, respectively), but neither sex predominated, and primary tumor location was different, with respect to each case series. In studies conducted in China and Japan,11–13 mean age was similar to the results in the case series conducted in Spain and the United States, predominance by sex was slight, as in the case series from the United States,11,15 and the predominant location was the head of the pancreas. Those results suggest that PNETs in the Asian countries could be different.11 Comparing our case series with that of Chheda et al.14 (India, in 2015), there was a marked predominance of men (79.6%), mean patient age at disease appearance was 55.5 years, which was similar to our results (mean 55 years), the predominant tumor location was the head of the pancreas (56%), which was similar to the percentages in the Chinese series, 87% of the patients had non-functioning tumors, and there was an elevated presence of metastasis (77%), higher than all the other case series evaluated. In addition, the number of patients that underwent surgery in the Indian case series (34%) was much lower than the figures of the rest of the case series (all above 70%). However, the 5-year survival rate in the case series by Chheda et al. was high (84-100%, according to tumor type) and very similar to survival in the rest of the case series (Table 3). A weakness of our study was not having 5-year overall survival data.
In our case series, mean tumor size (3.9cm) was similar to the size of NF-PNETs in the studies from China and the United States (mean tumor size of 4cm and 3cm, respectively).11 In the case series published in 2011,7 in which the percentage of non-functioning tumors was lower (37%), the mean tumor size (2.0cm) was below that of our present case series, in which the NF-PNETs predominated (71%).
Detection rates through US or abdominal CAT of PNETs vary among authors: in the study by Rösch16 they ranged from 30-50%. Abdominal CAT was very accurate for PNET detection, with a sensitivity > 80% and a capacity to visualize tumors as small as 4mm. However, sensitivity decreases in tumors < 2cm, compared with larger ones. In cases of symptomatic non-functioning tumors that measure > 3cm at diagnosis, CAT can reach 100% sensitivity.17 When a PNET cannot be located through CAT, EUS has a mean detection rate of 90% (range 77-100%).18 According to several published studies, EUS is superior to multidetector CAT,19 something we did not find in our study, in which abdominal CAT was more sensitive (90%) than EUS (85%) (Table 1).
Our group published a retrospective review of 55 patients suspected of having NET that underwent radial or sectorial EUS. Forty-two tumors were detected in 40 cases, EUS-FNA was performed in 26 cases, and there was histologic confirmation in 9 cases, with 100% sensitivity.20 EUS-FNA diagnostic sensitivity was 94% in the present case series.
Despite the fact that the majority of PNETs are solid lesions, 6 tumors were cystic (8.5%), with smooth margins and peripheral enhancement in the arterial and portal venous phases on the CT scan. In general, it is difficult to differentiate cystic PNET from another pancreatic cystic lesion and imaging tests have a diagnostic error rate of approximately 43%, according to the case series described by Singhi.21
In regard to metastasis, 32.8% (23/70) of the tumors presented with metastasis at diagnosis, similar to the 37% (7/19 cases) found in the 2011 case series.7 Fischer and Cherenfant reported an incidence of carcinoma and systemic and lymph node metastases between 13 and 25%, respectively, in incidental tumors < 2cm.22,23 Some authors believe there is a close relation between tumor size and risk for metastasis in functioning PNETs that increases in tumors > 2cm.24,25 In an epidemiologic study conducted in the United States and published in 2008, the figures were 32 to 73% and the cases were metastatic at diagnosis.5 Liver metastases are the most frequent (98%), and accounted for 100% of the metastases in our case series. Other less affected sites include: bone (20%), retroperitoneum (12%), and abdominal cavity (11%).26 According to the literature, most PNETs present with metastasis (60%) or locally advanced disease (21%).27
Surgical resection is the treatment of choice, even for incidentalomas. It should be performed by teams with experience in pancreatic surgery. However, the exception is small NF-PNETs (<2cm), in which the morbidity and mortality of the patient must be considered when contemplating surgical intervention.28 The role of routine surgery is controversial in patients with MEN1 and Zollinger-Ellison syndrome, because tumors in those patients hardly ever remit without extensive resection (Whipple operation).2 In advanced stage disease (IV), surgical resection should be combined with curative resection (R0) of the metastases, as long as it is possible in the multimodal treatment context, such as chemoembolization and chemotherapy.3
From the current PNET case series, it can be concluded that there are population variations in relation to sex and location, but predominant tumor type and size coincide. All the series have elevated and very similar overall survival figures. With respect to our previously published case series, there was an increase in the incidentally detected PNETs, in the percentage of non-functioning tumors, and in mean tumor size, which could be related to the increased use of imaging studies for clinical diagnosis and the greater sensitivity provided by the current imaging technology. However, no increase in actuarial survival rates was observed, but more prospective and comparative studies are required.
Ethical disclosureProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that no patient data appear in this article.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Financial disclosureNo financial support was received in relation to this article.
Conflict of interestThe authors declare that there is no conflict of interest.
The authors wish to thank the following physicians: Gornals J., Loras C., Alastrué A., Díez Caballero A., Artigas V., Sanfeliu E., Romera N., Cruz M., Cerdá P., and Montenegro A, for their contributions in relation to the patients studied.
Please cite this article as: Varas-Lorenzo MJ, Cugat E, Capdevila J, Sánchez-Vizcaíno Mengual E. Detección de tumores neuroendocrinos pancreáticos: 23 años de experiencia. Revista de Gastroenterología de México. 2019;84:18–25.