SpyGlass Discover (Boston Scientific, Natick, Massachusetts, USA) is a new version of digital single-operator cholangioscopes that has recently been developed. The SpyGlass Discover cholangioscope is especially designed to achieve direct visualization of the biliary tract through percutaneous access. It has an optimum efficacy-safety profile due to its easy maneuverability, compared with other models, but its routine use in the West, principally in Latin America, is still limited because of its low level of availability and high cost.1–6
A 74-year-old man underwent a laparoscopic cholecystectomy 4 years earlier due to gallbladder polyps. He presented with a duodenal ulcer that strictured 80% of the intestinal lumen, impeding the passage of the endoscope. For the past 2 weeks, the patient had colicky abdominal pain in the right hypochondrium, associated with fever, jaundice, nausea, and vomiting, as well as a cholestatic liver profile and leukocytosis. Magnetic resonance cholangiography was performed that revealed a 17 mm-dilated common bile duct with a filiform segment and multiple stones inside it, of which at least 5 measured 14 × 10 mm in diameter.
The patient was admitted to the hospital with the diagnosis of acute cholangitis and an endoscopic retrograde cholangiopancreatography procedure was immediately attempted, but failed because the major papilla was inaccessible due to the stricture caused by the duodenal ulcer. The decision was made to carry out percutaneous biliary drainage, through interventional radiology. The procedure required ultrasound equipment with a 3.5 MHz convex transducer and a C-arm with a rotating anode, along with a 22 Go Chiba needle, 4 Fr micropuncture set, 5 Fr × 30 cm catheter, 0.035″ hydrophilic guidewire, and 0.38″ Teflon guidewire. The procedure was started with local anesthesia and the direction of the 22 Go Chiba needle was guided by ultrasound. Fluoroscopy was then carried out for the percutaneous cholangiography and for tracing the access to the biliary tree. A 0.018’’ micro-guidewire was initially introduced through the Chiba needle until approaching access to the bile duct. Once there, it was substituted by a micropuncture set with a 0.035″ guidewire that ensured access to the bile duct, improving the infectious process.
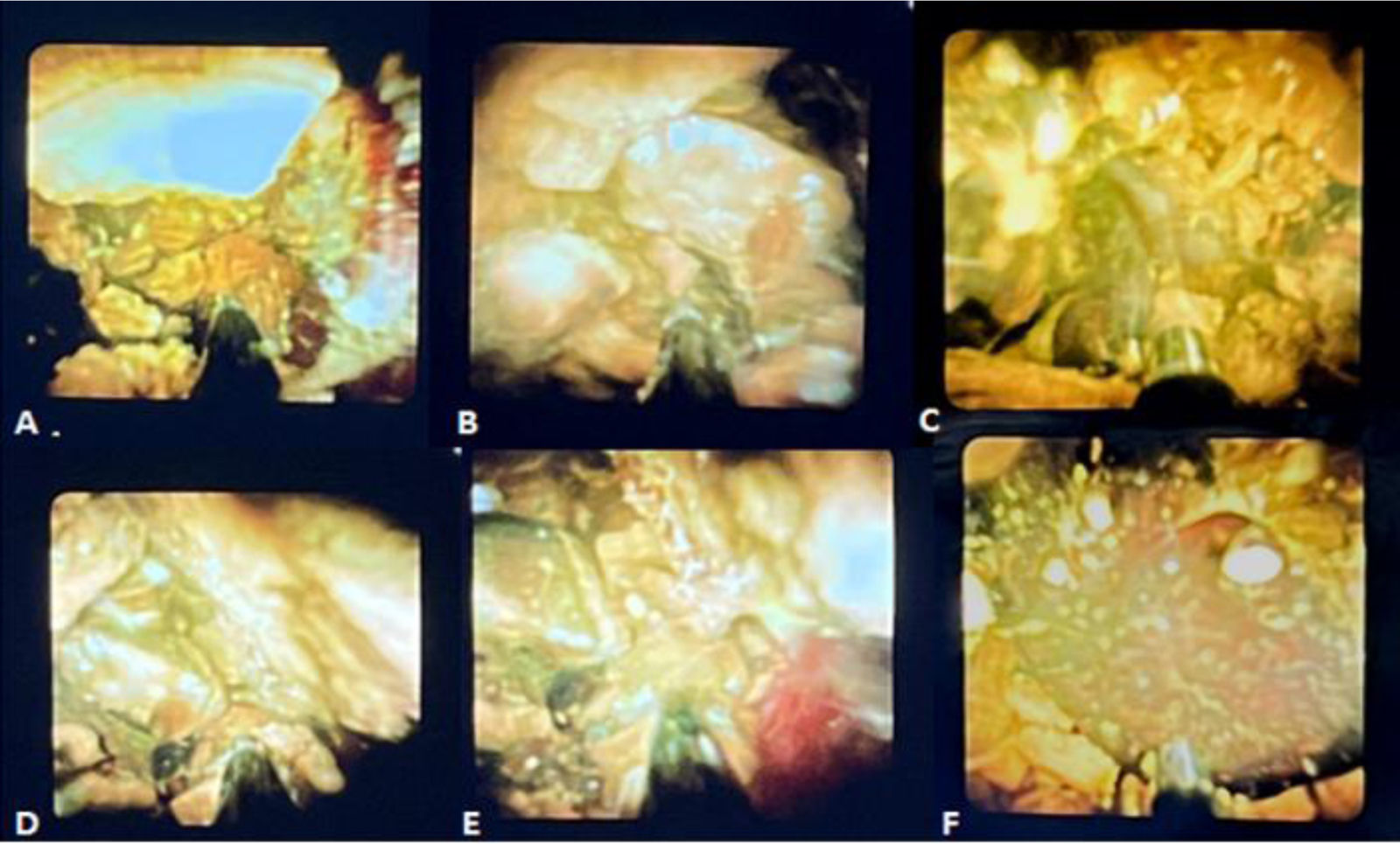
Two weeks later, the permeability of the access site was confirmed, utilizing a 12 Fr and 14 Fr introducer as the working channel, performing a percutaneous cholangiography that revealed a 17 mm saccular dilation in the proximal third of the common bile duct and a strictured segment that measured 10 mm in length. It contained multiple faceted-like stones, the largest of which had a 16 mm diameter. Lastly, the SpyGlass Discover (SpyGlass Direct Visualization System; Boston Scientific, Natick, Massachusetts, USA) cholangioscope was introduced through a percutaneous guidewire, with continuous irrigation with sterile water for visualization, showing the presence of multiple stones in the middle and distal third of the common bile duct. Electrohydraulic lithotripsy was performed at a frequency of 10 Hz and medium power, achieving the fragmentation of the stones (Fig. 1A–F).
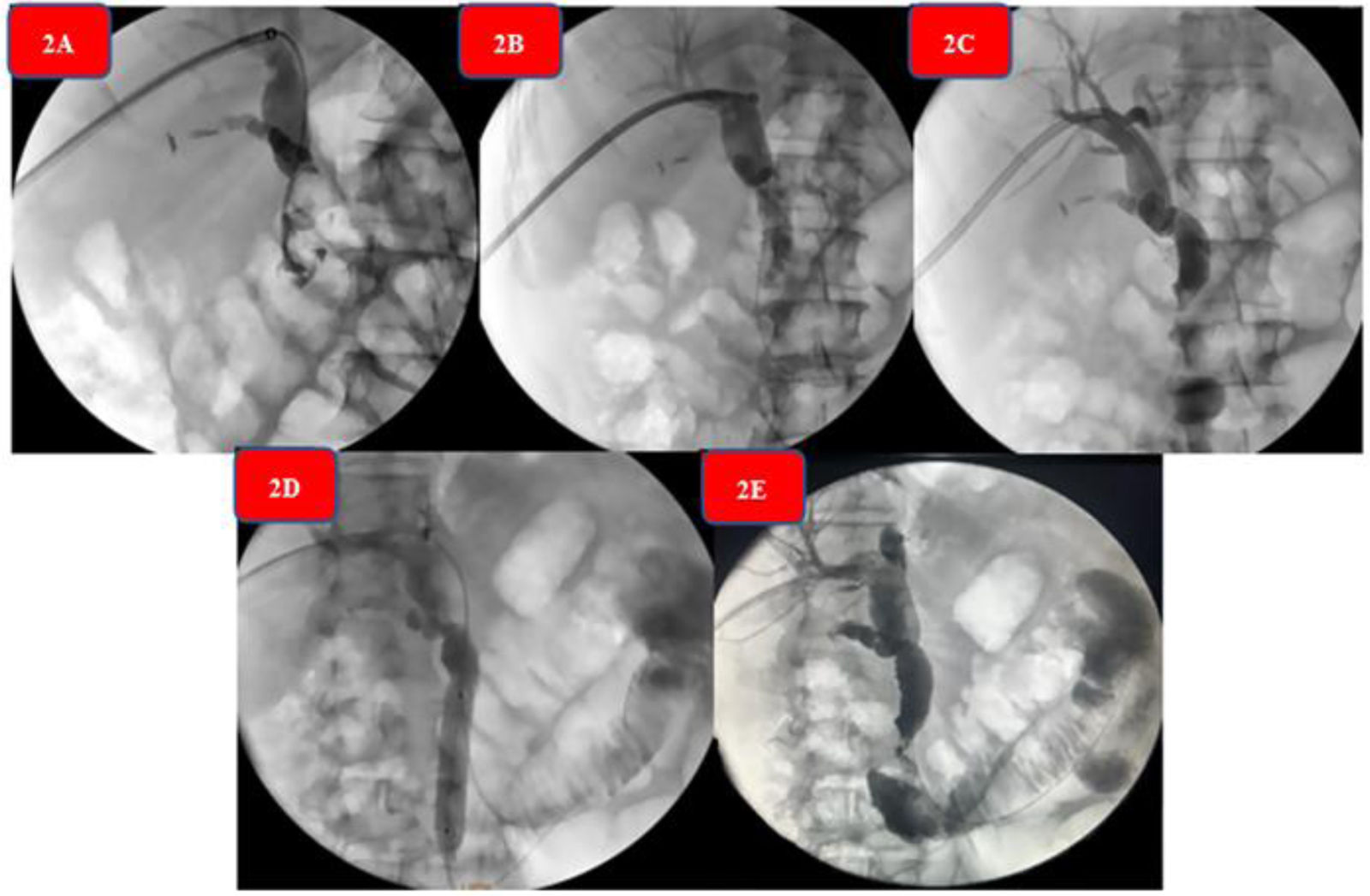
The equipment then advanced to the distal third of the common bile duct, where a concentric stricture with an inflammatory appearance was seen. Thus, the cholangioscope was withdrawn and hydropneumatic dilation was begun, utilizing a 12 mm (3ATM) - 13.5 mm (4.5ATM) - 15 mm (Amara, Endomed) esophageal balloon dilator, achieving passage of contrast medium into the duodenum. Finally, the stone fragments were expelled into the duodenum, under continuous irrigation and propulsion with a 16 mm stone extraction balloon catheter (MTW, Endomed). Control cholangiography showed adequate passage of the contrast substance throughout the bile duct, with no negative defects inside it. Prophylactic antibiotic was administered, and patient progression was favorable. Fig. 2A–E shows the sequence of the anterograde percutaneous cholangiography through fluoroscopy during the entire procedure.
Control follow-up with a liver panel and percutaneous cholangiography was carried out at one month. There were no images of stones inside the common bile duct and so the percutaneous drain was removed.
The strength of our study is that it reports the first scientific evidence of the use of the SpyGlass Discover (Boston Scientific, Natick, Massachusetts, USA) in Peru, in the successful management of difficult-to-treat choledocholithiasis in a patient with altered anatomy. The SpyGlass Discover System (Boston Scientific, Natick, Massachusetts, USA) is a new cholangioscope model that has a specific configuration designed for percutaneous or intraoperative approaches.
Like the traditional cholangioscope (Spyglass Direct DS Visualization System), the Spyglass Discover System (Boston Scientific, Natick, Massachusetts, USA) has a minimum diameter of 1.2 mm for the accessory channel, a minimum angular range of 30°, and a distal width of 10.5 Fr (3.5 mm). Its main difference and advantage, compared with the conventional long cholangioscopes (230 cm), is the greater and easier maneuverability it offers the endoscopist. This is due to certain technical characteristics in its structure, such as its working length of 65 cm. However, there is very little scientific evidence supporting its use and the majority of publications are isolated case reports.7–9 Phillpotts et al.10 analyzed 5 cases of percutaneous and intraoperative cholangioscopy, utilizing the SpyGlass Discover (Boston Scientific), in both normal and altered bile ducts, and showed a 100% (5/5) success rate, with no adverse events in any of the cases. Our study reaffirms those results, adding more scientific evidence on the benefits of using the SpyGlass Discover System (Boston Scientific, Natick, Massachusetts, USA), and now in a Latin American population.
In conclusion, our study shows that the use of the SpyGlass Discover System (Boston Scientific, Natick, Massachusetts, USA) in patients with altered anatomy is a reality in Latin America, and that in expert hands, has an optimum efficacy-safety profile. Nevertheless, more studies with a larger patient cohort and longer follow-up period are needed, to consolidate these results in the medium and long terms.
Ethical considerationsThe authors declare that no experiments were conducted on animals or humans during the present study. It describes the case of a patient with difficult-to-treat choledocholithiasis in an altered anatomy that was successfully resolved through SpyGlass Discover System cholangioscopy. The study was conducted in accordance with the Declaration of Helsinki and the authors confirm that it meets all the established norms for scientific research, including the data confidentiality of the patient described herein, as well as his informed consent.
Financial disclosureNo financial support was received from any public or private institution in relation to this case report.
Conflict of interestThe authors declare that there are no conflicts of interest.