Neuroendocrine tumors (NETs) are a rare neoplasia that derives from neuroendocrine cells of the pancreas, the gastrointestinal tract, and the bronchopulmonary tree.1 Thirty to 45% of the cases develop metastases, mainly to the liver.2 Liver transplantation (LT) is the treatment of choice in patients that are not candidates for resection and in patients whose tumors cause uncontrollable hormonal symptoms.3,4 Currently, there are criteria for considering LT in patients with NETs that metastasize to the liver, with a 90% 5-year survival rate.5
The aim of the present article was to report on the survival of a patient that underwent LT due to NET with metastasis to the liver, at the 7-year follow-up.
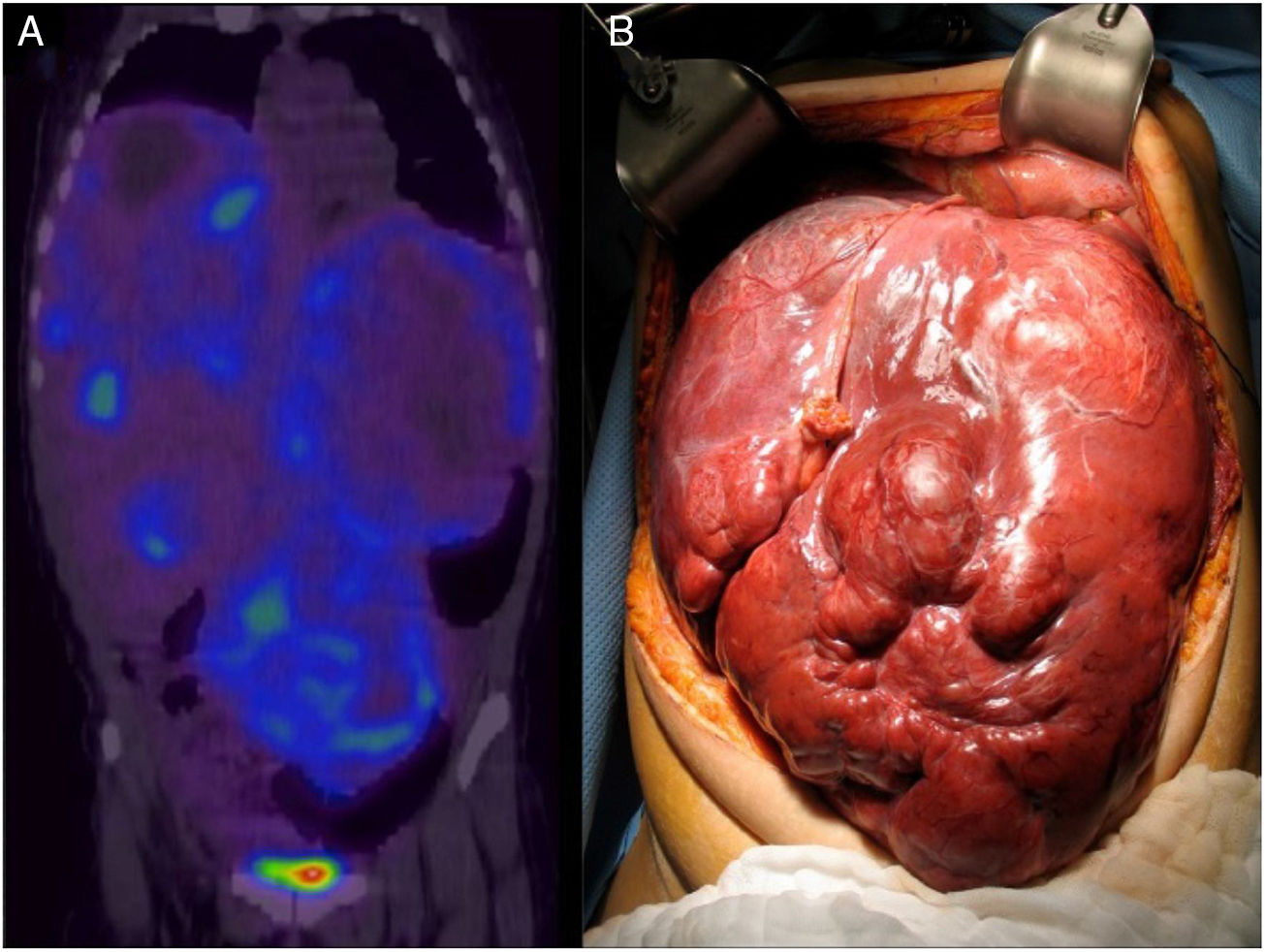
A 26-year-old woman presented with clinical symptoms of 2-year progression characterized by postprandial pain in the epigastrium that progressed to bloating, early satiety, dyspnea, and weight loss of 20kg. A computed tomography (CT) scan and magnetic resonance imaging of the abdomen revealed multiple confluent lesions in the liver, some with cystic degeneration, that occupied more than 80% of the hepatic mass. Serum chromogranin A was reported at 114 ng/dl (1.9-15 ng/ml) and 5-hydroxyindoleacetic acid in 24-h urine was 2.9mg (< 6mg/24h). The liver biopsy report demonstrated a well-differentiated NET (chromogranin+, synaptophysin+, CD56+, HEPAR 1 negative, and Ki-67 < 2%). Enteroscopy and Tc-99-octreotide SPECT/CT could not identify the primary lesion due to the massive tumor involvement in the liver (fig. 1). The Transplantation Committee of our hospital decided to make an exception of the patient's MELD score of 22, given her youth and very poor quality of life, and the fact that the tumor had a low grade of malignancy, the disease had not progressed in 6 months, and no metastases to other sites had been identified.
In April of 2011, deceased donor LT was performed utilizing the technique of total exclusion of the vena cava. During the transplant, the primary tumor, measuring 1cm in diameter, was identified at the antimesenteric border of the small bowel. It was resected and an end-to-end enteroanastomosis was performed. The histopathologic report of the specimen corroborated the diagnosis (fig. 2). At seven years since the procedure, the patient has adequate quality of life, her liver function tests are normal, and abdominal CT has shown no disease activity.
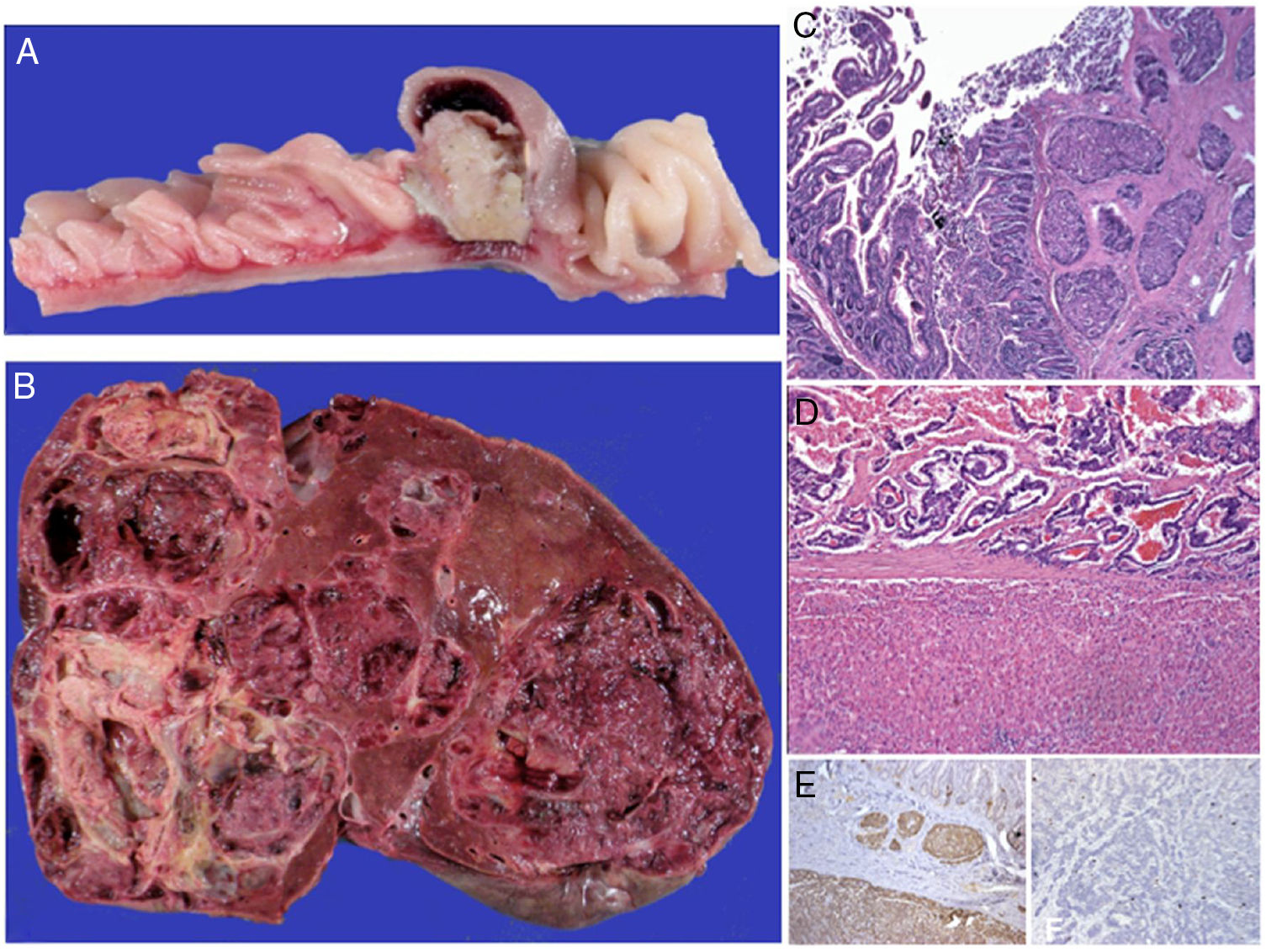
Macroscopic aspect of the neuroendocrine carcinoma of the jejunum (A) and the almost total substitution of the hepatic parenchyma by metastases (B). In both organs the lesion is well-differentiated, with classic carcinoid patterns (C) and sclerosis of the neighboring stroma (C and D). Reactivity for the endocrine differentiation markers was diffuse and intense (chromogranin A in image E), with focal nuclear expression for the MIB-1 cellular proliferation antibody (Ki-67 < 2%).
NETs are slow-growing tumors, enabling some patients to be candidates for LT with good results. Mazzaferro et al. conducted a study in which they compared patients with metastatic NET that were candidates for LT and that met the Milan-NET criteria (age < 60 years, well-differentiated NET, Ki-67 < 5%, stable disease for at least 6 months, “R0” tumor resection with portal drainage, metastasis < 50% of total liver volume, and no extrahepatic disease) with a group with metastatic NET that did not undergo LT. The LT recipients had a 5 and 10-year survival rate of 97 and 51%, respectively, compared with a corresponding 88 and 22% in the patients that did not undergo transplantation (p < 0.001).6 That study clearly demonstrated the benefit of LT in NETs that metastasize to the liver, especially in relation to the follow-up at 10 years. However, some selection criteria for LT continue to be subjects of debate.
In a systematic review of the literature, Fan et al. stated that the risk factors for poor survival or early recurrence were: age > 50 years, symptomatic tumors, primary NET in the pancreas, high level of Ki-67 (> 5%), more than 50% involvement of the liver, and poorly differentiated tumors.7
In an analysis of 213 patients in the European Liver Transplant Registry, the main risk factors were multivisceral resection or multivisceral transplantation, poorly differentiated tumors, and hepatomegaly. If patients were selected without those factors, the 5-year survival rate was from 60 to 80%.8
Mazaferro et al. arbitrarily chose 6 months as the ideal waiting period before LT, assuming that patients with liver metastases that have a longer follow-up before the transplant will have better progression. That hypothesis has been confirmed in retrospective studies. Currently, the majority of centers do not consider LT in asymptomatic patients or in those with stable disease, but once the disease progresses or becomes refractory to medical treatment, LT can be performed in selected cases. Therefore, rather than there being a specific waiting period, TH should be carried out after a time of disease stability, but before disease progression.9
In our patient, despite the fact that she presented with 2 risk factors (unresected primary tumor and massive liver involvement), the decision was made for the patient to undergo LT, 6 months after follow-up and under treatment with octreotide. At present, the patient's survival is over 7 years and she is disease-free. To the best of our knowledge, the present report is the first on LT due to NET with metastasis to the liver in Mexico.
Ethical disclosuresProtection of human and animal subjects. The authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of data. The authors declare that they have followed the protocols of their work center regarding the publication of patient data and have maintained patient anonymity throughout.
Right to privacy and informed consent. Informed consent was not required for the publication of the present article because no personal data identifying the patient were published.
Financial disclosureNo specific grants were received from public sector agencies, the business sector, or non-profit organizations in relation to this study.
Conflict of interestThe authors declare that there is no conflict of interest.
Please cite this article as: Vilatobá M, Hurtado-Gómez S, García-Juárez I, Huitzil-Melendez D, Gamboa-Domínguez A. Trasplante hepático en tumor neuroendocrino metastásico, primer reporte en México. Revista de Gastroenterología de México. 2019;84:414–416.