Intragastric balloon therapy is a temporary, minimally invasive method for inducing weight loss. Any balloon with a volume of 400 ml induces satiety and delayed gastric emptying. The aim of the present study was to demonstrate its safety and efficacy.
Materials and methodsWe analyzed the safety and effectiveness of balloon placement in relation to weight loss in 128 patients. Subgroups were compared through the Student’s t test, ANOVA test, and the chi-square test, depending on the type of variable and the number of groups evaluated. Statistical significance was set at a p ≤ 0.05 to evaluate weight loss.
ResultsMean weight loss after balloon therapy was 10.7 kg, with only 2 cases of complications due to oral feeding intolerance. Mean length of time with the balloon was 8 months. There were no complications after 6 months, but no further weight loss either (p = 0.540). Final weight loss versus initial weight loss was significant (p = 0.000). There was greater weight loss in the group that attended the bimonthly control appointments than in the group that did not: 12.7 kg vs. 7.26 kg (p = 0.000).
ConclusionsThe rates of the adverse events of intolerance and complications were very low in our study, compared with results in the literature (1.6% vs. 4.7% and 0% vs. 3%, respectively). Intragastric balloon placement had better results when the patients attended the control appointments. Intragastric balloon therapy is a safe and efficacious method for reducing weight in patients with overweight and obesity.
El balón intragástrico (BIG) es una terapia de mínima invasión temporal para inducir pérdida de peso. Cualquier balón con un volumen de 400 ml induce saciedad y retraso del vaciamiento gástrico. El objetivo de este estudio es demostrar su seguridad y eficacia.
Material y MétodosAnalizamos la efectividad y seguridad en la pérdida de peso en 128 pacientes con balón. Se compararon subgrupos mediante la prueba t de student, ANOVA y Chi 2 dependido del tipo de variables y numero de grupos evaluados tomando una significancia estadística <0.05 para evaluar la pérdida de peso.
ResultadosLa pérdida de peso promedio después de la terapia con balón fue de 10.7 kg con únicamente 2 casos de complicaciones por intolerancia a la vía oral. La permanencia promedio fue de 8 meses, sin haber complicaciones después de los 6 meses, pero tampoco mayor pérdida de peso (p = 0.540), La pérdida de peso final vs inicial fue significativa (p = 0.000), en el grupo que acudía a una cita bimestral de control perdieron mas peso que aquellos que no tuvieron cita; 12.7 kg vs 7.26 kg (p = 0.000).
ConclusionesComparando estos resultados con la literatura, la tasa de eventos adversos como la intolerancia y complicaciones es muy baja en nuestro trabajo (1.6% vs 4.7%) y (0% vs 3%) respectivamente. La colocación del balón intragástrico tuvo mejores resultados cuando el paciente acudió a su cita de control. El balón intragástrico es un método seguro y eficaz para disminuir el peso de pacientes con sobrepeso y obesidad.
Obesity increases the risk for premature death, as well as the association of numerous diseases. The worldwide prevalence of obesity reflects the fact that there is no appropriate standardized treatment for each grade of obesity.1–4 More than 1.4 billion adults across the globe present with overweight or obesity. A 5-10% weight loss can contribute to preventing or delaying the appearance of diabetes and other comorbidities associated with obesity.5–10 Intragastric balloon placement is a temporary, minimally invasive therapy for inducing weight loss. Any balloon with a volume of 400 ml induces satiety and contributes to delayed gastric emptying.11–13
The procedure is indicated for the following individuals: patients with overweight, presenting with a body mass index (BMI) of 27 kg/m2 (the European reference) or 30 kg/m2 (the United States reference), in whom other previous weight loss therapies have failed; patients with a BMI of 30-35 kg/m2, and so are not candidates for bariatric surgery; patients with a BMI of 40 kg/m2 or those with a BMI of 35 kg/m2 and one or two obesity-related comorbidities that reject bariatric surgery because of the risks involved. A balloon can be placed in patients with severe obesity (BMI > 50 kg/m2) as an initial weight loss measure, prior to bariatric surgery, to improve clinical conditions and reduce the risks associated with anesthesia.14,15
The Orbera balloon (a registered trademark of Apollo Endosurgery, Inc.) is the most widely used worldwide. It is made of silicon and has a capacity of 400-700 ml. In the United States, it is used in patients with a BMI of 30 to 40 kg/m2 that have not achieved adequate weight loss through diet and exercise.16–19 In a 2015 meta-analysis, mean weight loss was 12.3%, 13.16%, and 11.27% at 3, 6, and 12 months after its placement. At 6 months, patients had lost 32.1% of their excess weight and reduced their BMI by a mean of 5.7 kg/m2.20 In a study on 143 patients with Orbera balloon placement, the incidence of diabetes, hyperlipidemia, hypertriglyceridemia, and hypercholesterolemia was shown to have decreased, one year after its removal.21
In another study on 119 patients with Orbera balloon placement, levels of hemoglobin A1C, fasting glucose, cholesterol, triglycerides, and C-reactive protein had decreased and quality of life questionnaire scores had improved, at 6 months after its removal.22 Long-term weight loss with the balloon was greater, when compared with medications, but less favorable, when compared with bariatric surgery. The percentages of excess weight loss with bypass, gastric sleeve, and gastric band surgeries were 70, 60, and 50%, respectively, at 2 years. There is an expected maximum weight loss of 25 to 38% with the Orbera balloon.23–26 The majority of complications associated with the balloon procedure occur during removal and they include esophageal tear, pneumonia, and esophageal bleeding or perforation.26–30 Weight gain after undergoing a weight loss therapy is a common problem in patients with overweight and obesity. Fifty percent of the weight lost during balloon therapy can be maintained through adequate diet and exercise up to one year after balloon removal.28
Some patients request repeat balloon placement, which can be performed one month after the removal of the previous balloon. Repeat placement can be as effective as the prior therapy.29–33 In patients with severe obesity, the balloon can be used in conjunction with the surgical approach. Preoperative weight loss improves postoperative results and reduces the risk for intraoperative complications.34,35 In an Italian study by Genco et al.,30 in which 2,515 intragastric balloons were placed, they reported a positioning failure rate of 0.08% and a total complication rate, including obstruction and perforation, of 2.8%, over a 6-month period. Weight loss was measured through BMI, with a mean reduction of 4.9 ± 12.7 kg/m2. Balloon placement was concluded to be safe and effective for reducing up to a mean of 10 kg of weight. The aim of the present study was to evaluate the safety and efficacy of intragastric balloon placement at a referral center, as well as broadening the knowledge about intragastric balloon therapy, given that there are few publications with large case series on the topic in Mexico.
Materials and methodsA total of 138 reports of patients that underwent intragastric balloon placement were retrieved from the endoscopy archive, along with an obesity follow-up log on those patients from 2015 to 2017. The variables of sex, age, initial BMI, final BMI, initial weight, final weight, total weight loss, height, control appointments, length of time with the balloon, and complications were considered. Ten patients with incomplete records or absence of the variables of interest were excluded. A descriptive analysis was performed through measures of central tendency for the quantitative variables and frequencies for the qualitative variables. The inferential analysis was carried out initially using a univariate model through a t test for related variables (a paired t test), comparing weight loss, the percentage of excess weight, and BMI before and after intragastric balloon placement. The Student’s t test was then used for comparing the means between two groups for the unrelated quantitative variables and the ANOVA test for those with more than two groups, such as in the BMI interpretation. The chi-square test was used for the qualitative variables, to reach statistical significance at a p ≤ 0.05 with a 95% confidence interval (95% CI). The SPSS software version 23 (SPSS Inc. Chicago, IL) was used for both the descriptive and inferential analyses.
Ethical considerationsThe authors declare that no human or animal experiments were carried out in the present study, nor was informed consent from the patients required, given that no personal information was disclosed. This study was conducted following the principles of the Declaration of Helsinki and authorized by the research and ethics committee of the Hospital Central Militar.
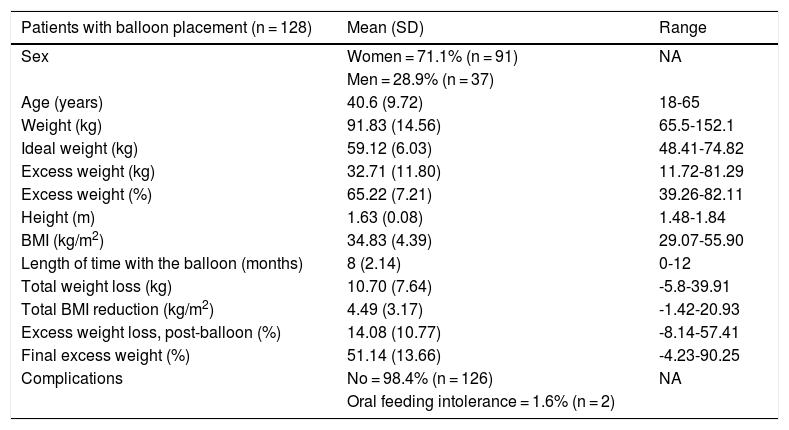
ResultsRegarding the 128 patients with balloon placement, the following means for each variable were reported: initial BMI 34.83 kg/m2, final BMI 30.33 kg/m2, initial weight 91.83 kg, final weight 81.13 kg, total weight loss 10.70 kg, initial excess weight 65.22%, final excess weight 51.14%, height 1.63 m, length of time with the balloon 8.23 months, and age 40.6 years, as shown in Table 1.
Demographic characteristics and study variables.
| Patients with balloon placement (n = 128) | Mean (SD) | Range |
|---|---|---|
| Sex | Women = 71.1% (n = 91) | NA |
| Men = 28.9% (n = 37) | ||
| Age (years) | 40.6 (9.72) | 18-65 |
| Weight (kg) | 91.83 (14.56) | 65.5-152.1 |
| Ideal weight (kg) | 59.12 (6.03) | 48.41-74.82 |
| Excess weight (kg) | 32.71 (11.80) | 11.72-81.29 |
| Excess weight (%) | 65.22 (7.21) | 39.26-82.11 |
| Height (m) | 1.63 (0.08) | 1.48-1.84 |
| BMI (kg/m2) | 34.83 (4.39) | 29.07-55.90 |
| Length of time with the balloon (months) | 8 (2.14) | 0-12 |
| Total weight loss (kg) | 10.70 (7.64) | -5.8-39.91 |
| Total BMI reduction (kg/m2) | 4.49 (3.17) | -1.42-20.93 |
| Excess weight loss, post-balloon (%) | 14.08 (10.77) | -8.14-57.41 |
| Final excess weight (%) | 51.14 (13.66) | -4.23-90.25 |
| Complications | No = 98.4% (n = 126) | NA |
| Oral feeding intolerance = 1.6% (n = 2) |
BMI: body mass index; SD: standard deviation.
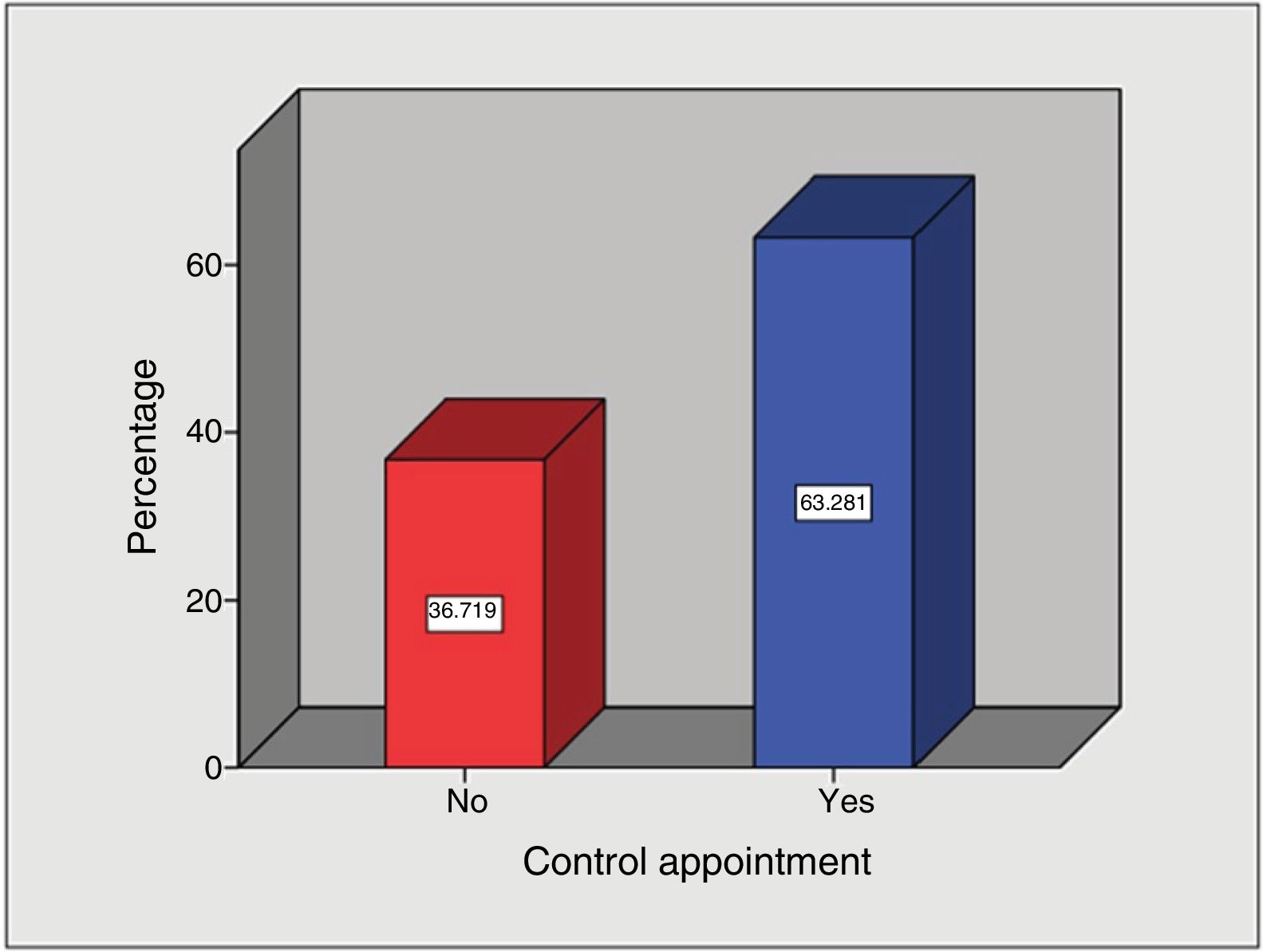
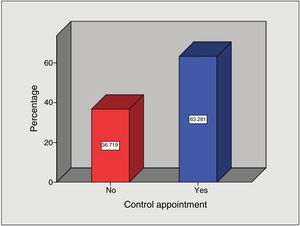
The analysis of weight control follow-up visit attendance showed that of the patient total, the majority of patients (n = 81) attended their bimonthly appointments (63.3%) and 47 patients did not (36.7%) (Fig. 1).
In relation to the total of balloons placed, there were only 2 cases (1.6%) of complications due to oral feeding intolerance during the first 14 days after placement. No severe complications were reported during balloon placement or removal. The majority of the patients that underwent intragastric balloon placement were women (71.1%) and 28.9% were men (Table 2).
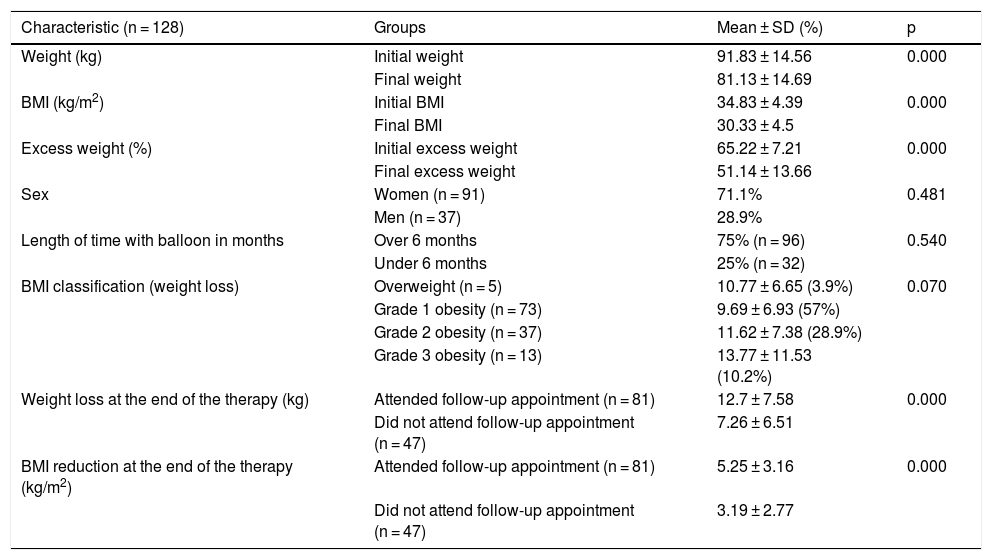
Subgroup results of balloon therapy.
| Characteristic (n = 128) | Groups | Mean ± SD (%) | p |
|---|---|---|---|
| Weight (kg) | Initial weight | 91.83 ± 14.56 | 0.000 |
| Final weight | 81.13 ± 14.69 | ||
| BMI (kg/m2) | Initial BMI | 34.83 ± 4.39 | 0.000 |
| Final BMI | 30.33 ± 4.5 | ||
| Excess weight (%) | Initial excess weight | 65.22 ± 7.21 | 0.000 |
| Final excess weight | 51.14 ± 13.66 | ||
| Sex | Women (n = 91) | 71.1% | 0.481 |
| Men (n = 37) | 28.9% | ||
| Length of time with balloon in months | Over 6 months | 75% (n = 96) | 0.540 |
| Under 6 months | 25% (n = 32) | ||
| BMI classification (weight loss) | Overweight (n = 5) | 10.77 ± 6.65 (3.9%) | 0.070 |
| Grade 1 obesity (n = 73) | 9.69 ± 6.93 (57%) | ||
| Grade 2 obesity (n = 37) | 11.62 ± 7.38 (28.9%) | ||
| Grade 3 obesity (n = 13) | 13.77 ± 11.53 (10.2%) | ||
| Weight loss at the end of the therapy (kg) | Attended follow-up appointment (n = 81) | 12.7 ± 7.58 | 0.000 |
| Did not attend follow-up appointment (n = 47) | 7.26 ± 6.51 | ||
| BMI reduction at the end of the therapy (kg/m2) | Attended follow-up appointment (n = 81) | 5.25 ± 3.16 | 0.000 |
| Did not attend follow-up appointment (n = 47) | 3.19 ± 2.77 |
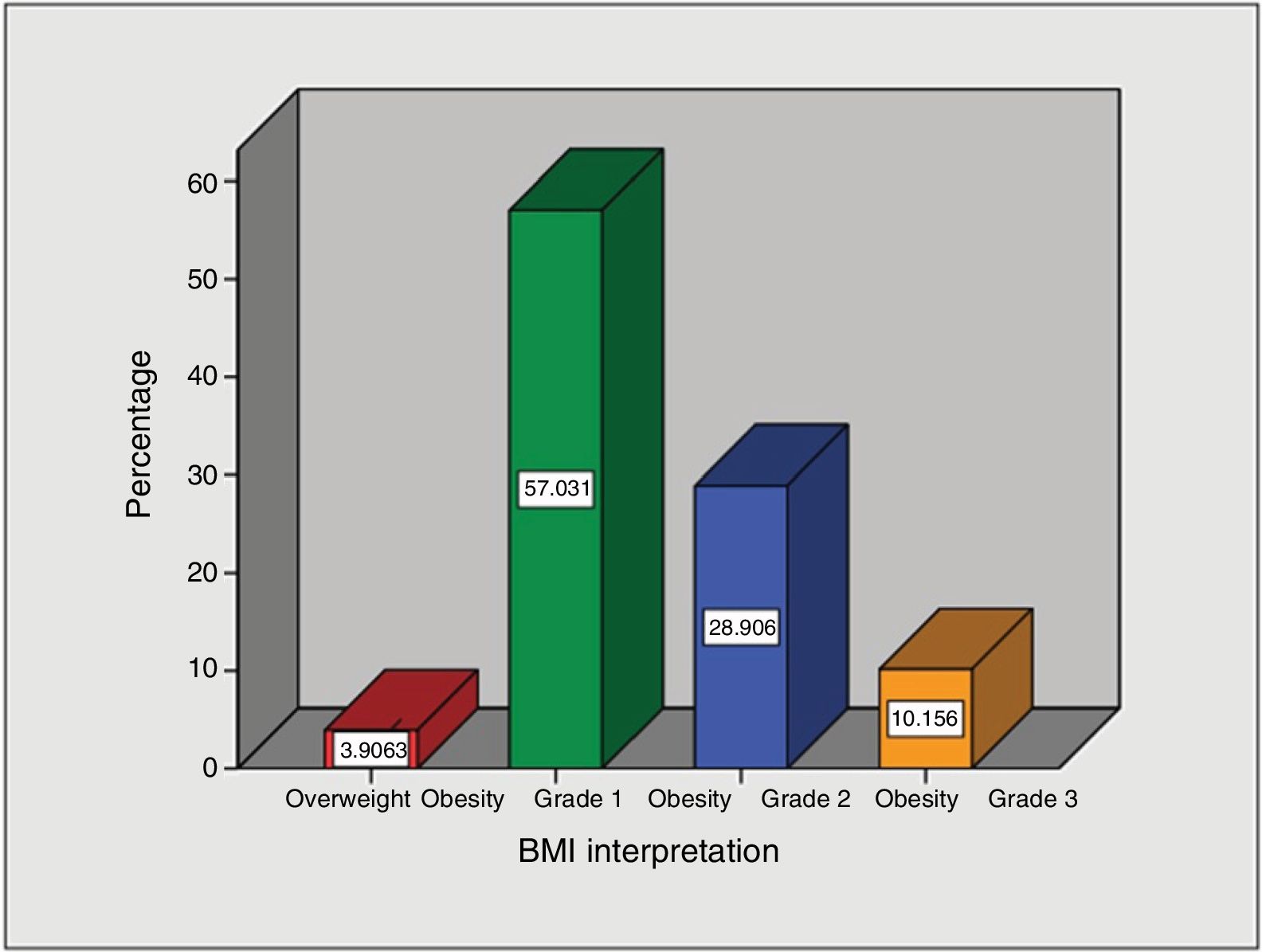
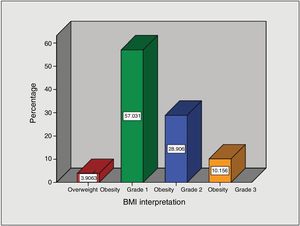
Regarding BMI interpretation, most of the intragastric balloons were placed in patients with grade 1 obesity (57%), followed by grade 2 obesity (28.9%), grade 3 obesity (10.2%), and finally, overweight (3.9%) (Fig. 2).
DiscussionThe majority of the intragastric balloons were placed in active military personnel, with a mean age of 40 years. The procedure was performed in many cases due to physical status requirements of the army. Mean weight loss and final BMIs at our hospital were similar to those reported in the largest series in the medical literature: 11.65% vs 11.27% and 4.5 kg/m2 vs 5.7 kg/m2, respectively.20
Despite the fact that the follow-up system included a programmed appointment for each patient that underwent intragastric balloon placement, a little over one-third of the patients did not attend their weight control appointments, making the partial bimonthly weight loss evaluation difficult to carry out in our study.
When comparing our results with those in the literature, the rates of the adverse events, such as intolerance and complications, were very low: 1.6% vs 4.7% and 0% vs 3%, respectively.26,27 In our patients, grade 1 obesity was the main indication for balloon placement, the same as is reported in the United States, whereas the procedure is accepted in patients with a BMI of 27 kg/m2 in Europe.14, 15
Patients with a BMI > 35 kg/m2 and comorbidities, as well as patients with grade 3 obesity, were also included in the balloon placement protocol, but they were then evaluated and guided by a surgical team to propose a bariatric procedure, given that bariatric surgery has better long-term results.23,24
Our weight reduction and BMI results were very similar to those reported in the largest case series in the medical literature,20 showing that intragastric balloon placement at our hospital is a safe and efficacious adjuvant method in overweight and obesity therapy that includes a nutritional plan. It was also useful in patients programmed to undergo bariatric surgery, by improving their clinical and metabolic conditions before the procedure, thus reducing the risk for postoperative complications.35
Intragastric balloon placement had better results in the patients that attended their weight control appointments. However, a little more than one-third of the patients (36.7%) did not attend them. That could be explained by the fact that their military schedule did not coincide with the programmed appointments or the patients had no symptoms or complaints after the placement procedure. Consequently, in some cases, the balloon remained in place longer than the 6 months recommended as therapy. We found no significant differences in relation to having the balloon longer than 6 months (p = 0.540), or in the BMI classification between subgroups (p = 0.070) (Table 2).
ConclusionsIntragastric balloon placement is a safe and efficacious weight reduction method for patients with overweight and patients with obesity. A good previous evaluation, with respect to patient selection, increases the success of the therapy and reduces its complications.
Adequate follow-up after balloon placement can improve results almost twice as much as not having one.
In patients with obesity > 35 kg/m2 and comorbidities, as well as in patients with obesity > 40 kg/m2 that do not wish to undergo bariatric surgery as treatment, intragastric balloon placement is a good alternative for reducing excess weight and its associated comorbidities in those patients.
More comparative studies between weight reduction therapies with balloon placement and surgical alternatives are needed to evaluate the long-term risk-benefit of each of them.
One of the weaknesses of our study was the loss to long-term follow-up, preventing a weight regain evaluation. Nevertheless, it clearly showed that intragastric balloon therapy achieved the desired goal in the majority of our patients, with a minimum of discomfort or complications.
Financial disclosureThe nature of the present study was purely scientific and received no type of sponsoring or financial support from any laboratory or business enterprise.
Conflict of interestNo laboratory or business enterprise exerted any type of influence on the study to alter its results in their favor and therefore the authors declare that there is no conflict of interest.
Please cite this article as: Almazán-Urbina FE, Santiago-Torres M, Rangel-Cruz E. Colocación de balón intragástrico en el tratamiento del sobrepeso y obesidad: experiencia de un centro de referencia en México. Revista de Gastroenterología de México. 2020;85:410–415.