Phlegmonous gastritis is a rare type of endogenous acute gastritis1 with a high mortality rate (approximately 50%).2,3 It is usually caused by a pyogenic bacterium and one of the most frequent complications is emphysematous gastritis. Some of the risk factors for presenting with phlegmonous gastritis are: immunodeficiencies, gastric carcinoma, prior therapeutic endoscopic procedure, oncologic diseases, malnutrition, and the chronic use of glucocorticoids.4 However, approximately 50% of the patients described in the medical literature that develop phlegmonous gastritis are healthy subjects.3 The infection mechanism can be through the hematogenic or lymphatic routes or through direct spread from another contaminated zone.5 Diagnosis is made through endoscopy or endoscopic ultrasound. Clinical symptoms are severe abdominal pain, inability to tolerate food, fever, and chills.4,6 At present, treatment ranging from antibiotic therapy to gastric resection is controversial.3,4
A 35-year-old man had a past history of non-atrophic gastritis after endoscopy and associated with Helicobacter pylori, having received eradication treatment one year before. He sought medical attention at our institution for intense epigastric pain of 5-day progression, accompanied by an inability to tolerate food; in the last 24 h he presented with melena and coffee ground vomitus, prompting his arrival at the emergency department. During the medical interview he stated he had eaten spoiled food 8 days prior to his symptom onset.
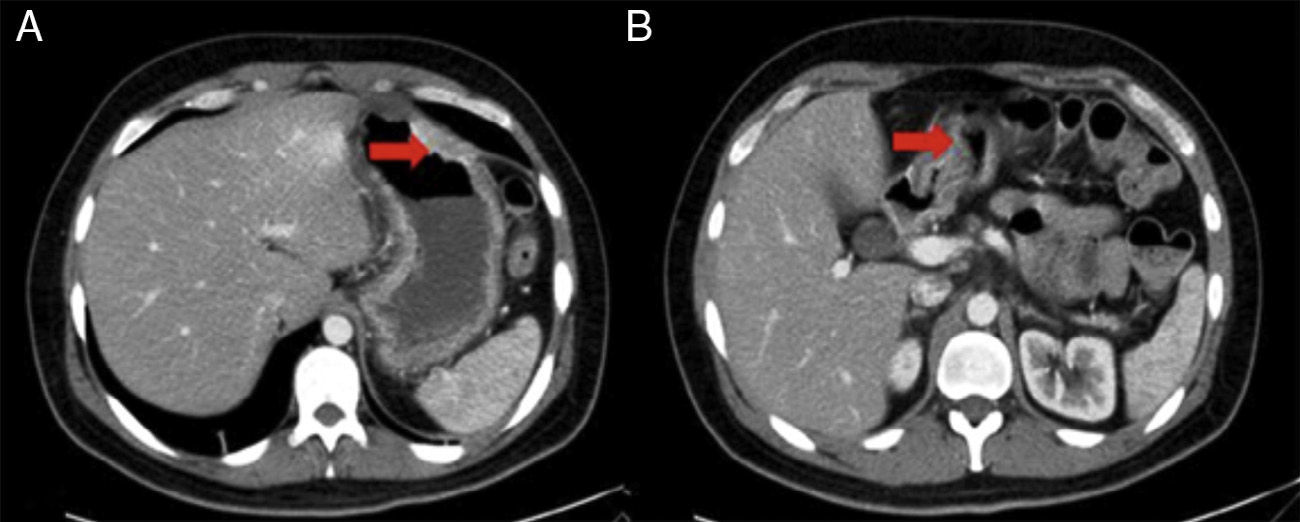
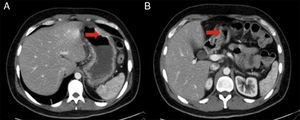
Physical examination revealed diaphoresis, tachycardia, low-grade fever, and normal blood pressure. Superficial palpation produced epigastric pain and no organomegaly. Laboratory work-up showed leukocytes 23.6 x 103μl; neutrophils 21 x 103μl; lymphocytes 0.9%; Hb 12.7 g/dl; MCV 78.5 fl; and C-reactive protein 21.61 mg/dl; the rest of the tests were within normal parameters. Abdominal computed axial tomography (CAT) scan (fig. 1) interpretation stated «important thickening of the gastric wall and duodenum of not-yet-determined etiology, reactive morphology of retroperitoneal and mesenteric lymph nodes, and pancreatic atrophy».
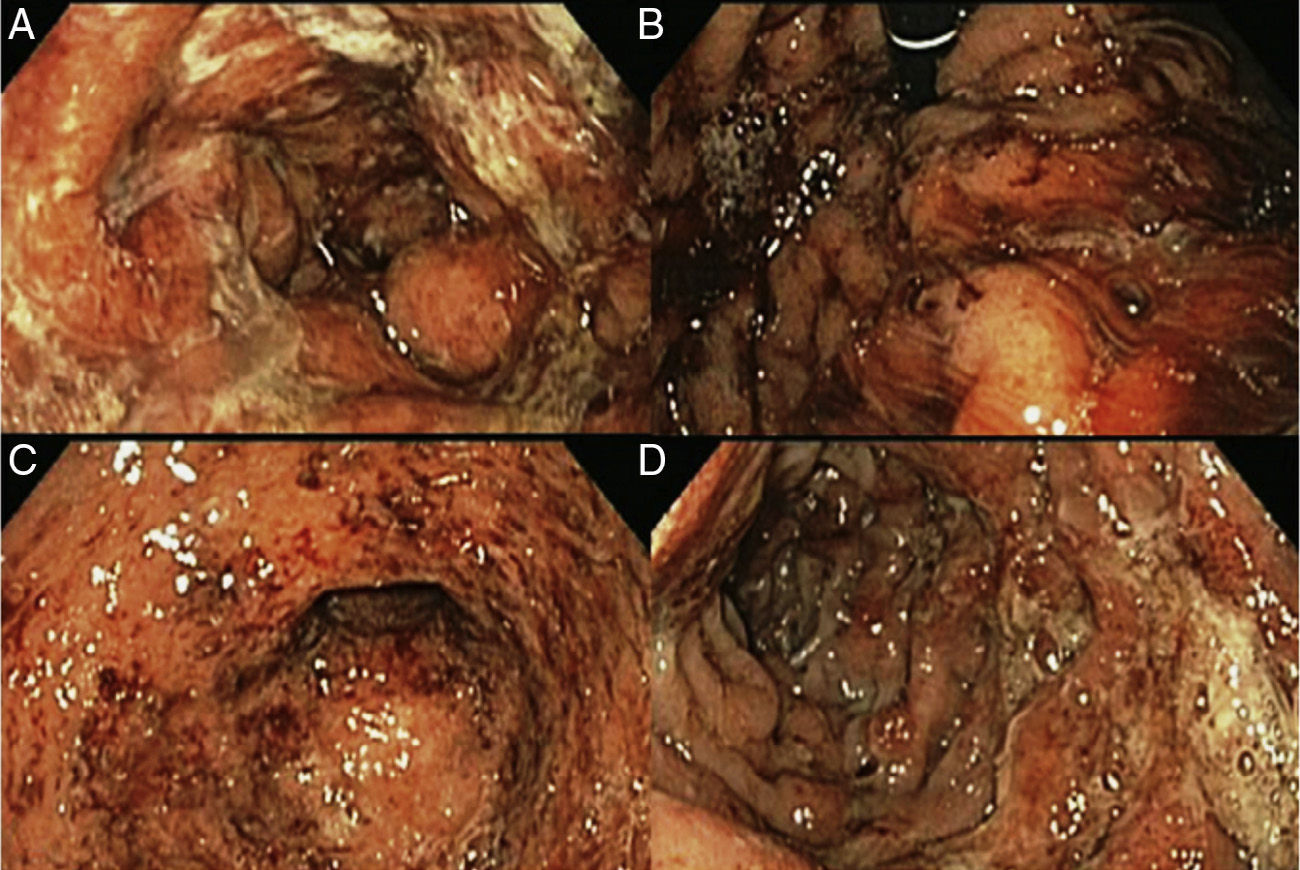
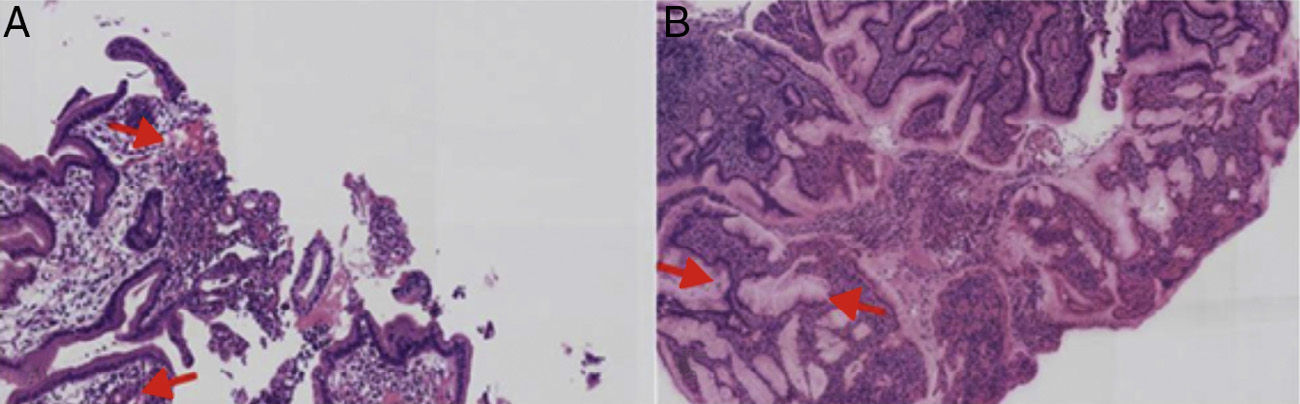
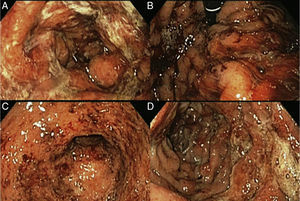
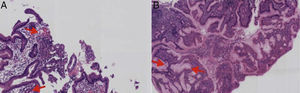
Upper gastrointestinal endoscopy was performed (fig. 2) identifying a normal esophagus and abundant hematic and mucopurulent matter in the gastric chamber, aspirating 200 cc. The gastric mucosa and folds presented with generalized edema and erythema, multiple erosions covered with mucopurulent exudate, and oozing hemorrhage. There were friable, edematous mucosa and folds that were covered with mucopurulent exudate and blood flecks extending up to the third portion of the duodenum. A gastric exudate aspirate and gastric and duodenal biopsies were taken for culture and histopathologic study. The culture had Gram-positive cocci growth consistent with beta-hemolytic streptococci. The pathology was described as «acute, intense, and active inflammation with the formation of microabscesses and a diffuse inflammatory infiltrate with a predominance of polymorphonuclear cells, multifocal ulceration of the epithelium, and foveolar hyperplasia consistent with acute phlegmonous gastritis». Immunohistochemistry was negative for: cytomegalovirus, Helicobacter pylori, mycotic microorganisms, and microbacteria (fig. 3).
A) Gastric mucosa with edema, erythema, and mucopurulent exudate. B) Retrograde view of the stomach showing oozing hemorrhage. C) Open edematous pylorus covered with mucopurulent exudate and blood flecks. D) Duodenum with friable edematous folds and mucosa, with erosions covered with mucopurulent exudate and blood flecks.
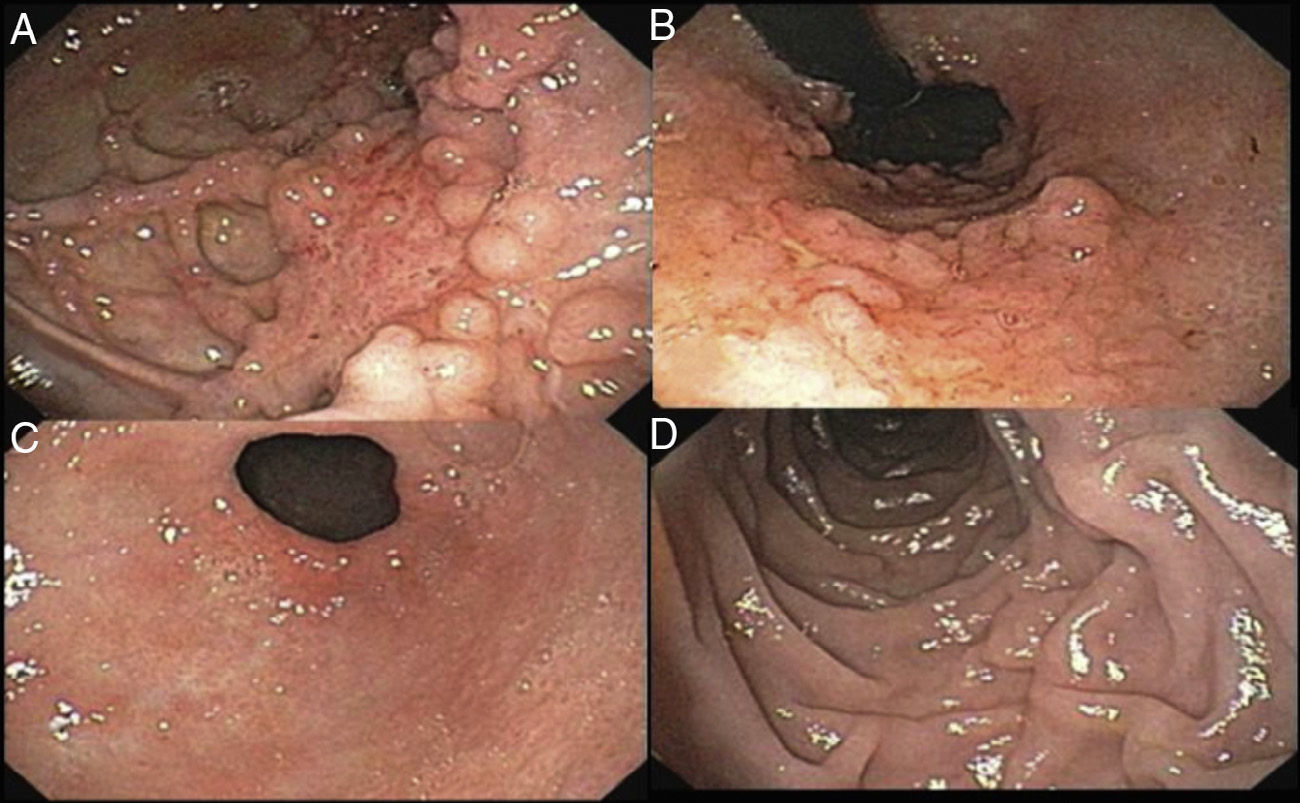
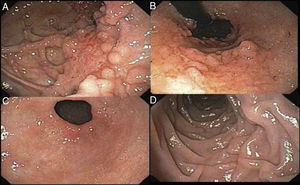
The patient remained in the hospital for 13 days with torpid progression. He was managed with class IV antibiotic therapy, parenteral nutrition, and afterwards with enteral nutrition through a nasojejunal tube. He was released to his home with outpatient treatment. The control endoscopy carried out 2 months later found «altered compliance of the gastric chamber due to scarring; mucosa with a paved and predominantly polypoid pattern on the posterior surface; multiple scars and the formation of false diverticula on the lesser curvature of the gastric body; and the duodenal bulb presented with patchy erythema». There were no further pathologic data (fig. 4).
Our patient was a previously healthy subject with no apparent risk factors, except the prior ingestion of spoiled food. He presented with the classic clinical symptoms described in the literature, but the differential diagnosis was extensive. In our case, the CAT scan enabled the suspicion of this pathology, and the differential diagnoses were excluded. The early endoscopic study together with the Gram staining and direct cultures of the gastric mucosa made it possible to confirm the diagnosis.6 Although treatment is controversial, conservative management with parenteral antibiotic therapy and nutritional support is effective when there is opportune diagnosis, reserving surgery for the presentation of complications.2
Financial disclosureNo financial support was received in relation to this study/article.
Conflict of interestThe authors declare that there is no conflict of interest.
The authors wish to thank Mónica García Gutiérrez MD of the Pathology Department of the Hospital ABC for providing the images of Acute Phlegmonous Gastritis.
Please cite this article as: Cortes-Barenque F, Salceda-Otero JC, Angulo-Molina D, Lozoya-González D. Gastritis flemonosa aguda. Revista de Gastroenterología de México. 2014;79:299–301.