Eosinophilic esophagitis (EoE) is a chronic, immune disorder mediated largely by food antigens. It shares nonspecific symptoms with gastroesophageal reflux disease (GERD). EoE is rarely reported in Mexico, perhaps due to the racial characteristics of the population or because of insufficient diagnostic suspicion.
AimsOur aim was to describe a Mexican cohort with EoE and evaluate the usefulness of the clinical history and endoscopy in the EoE diagnosis, in comparison with GERD patients.
Materials and methodsA retrospective study was carried out on the clinical characteristics and endoscopic and histopathologic findings in patients with EoE, along with a case-control study on patients with GERD. The endoscopic images obtained were interpreted in a blind and randomized manner by 4 gastroenterologists, before and after providing them with information on the characteristic alterations of EoE. The esophageal biopsies were also blinded to 2 pathologists that evaluated their diagnostic correlation. The Fisher's exact test and Mann-Whitney U test were used in the statistical analysis.
ResultsFourteen patients with EoE were included in the study. Ten (71%) of them were men and the mean age of the patients was 35 years. There were more subjects with a personal history of asthma (p=.0023) and food impaction (p=0.04) in the EoE group. The initial evaluation of the endoscopic findings showed 53% correct EoE interpretations and rose to 96% in the second revision (sensitivity 100%, specificity 71%, PPV 65%, NPV 100%).
ConclusionsMexican patients with EoE have similar characteristics to those of patients in western case series. Clinical awareness of the disorder increases endoscopic diagnosis in up to 40% of cases.
La esofagitis eosinofílica (EEo) es un padecimiento crónico e inmunológico mediado por antígenos habitualmente alimentarios. Comparte síntomas inespecíficos con la enfermedad por reflujo gastroesofágico (ERGE). En México es rara vez reportada, tal vez por las características raciales de la población o por una sospecha diagnóstica insuficiente.
ObjetivosDescribir una cohorte mexicana con EEo. Evaluar la utilidad de la historia clínica y de la endoscopia para diagnosticar EEo al compararla con ERGE.
Material y métodosEstudio retrospectivo de las características clínicas, hallazgos por endoscopia e histopatología de pacientes con EEo. Se realizó además un estudio de casos y controles con ERGE. Las imágenes obtenidas por endoscopia fueron interpretadas de forma ciega y aleatorizada por 4 gastroenterólogos, antes y después de otorgarles información sobre las alteraciones características de la EEo. Las biopsias esofágicas también fueron cegadas a 2 patólogos para evaluar su correlación diagnóstica. El análisis estadístico fue elaborado por pruebas exacta de Fisher y U de Mann-Whitney.
ResultadosSe incluyeron 14 pacientes con EEo, 10 (71%) del sexo masculino de 35 años en promedio. En el grupo de EEo hubo más sujetos con historia personal de asma (p=0.0023) e impactación alimentaria (p=0.04). La evaluación inicial de los hallazgos endoscópicos mostró el 53% de interpretaciones acertadas para EEo incrementando al 96% en la segunda revisión (sensibilidad 100%, especificidad 71%, VPP 65%, VPN 100%).
ConclusionesLos pacientes con EEo en México tienen características similares a los de series occidentales. La sospecha clínica incrementa el diagnóstico por endoscopia hasta en un 40% cuando se mejora el conocimiento acerca de la enfermedad.
Eosinophilic esophagitis (EoE) is a chronic inflammatory immune disorder that is mediated largely by food antigens.1 This disease was first described in 1978 by the physicians, Landres and Kuster, at the Scripps Clinic in San Diego, California,2 in the biopsies of a patient diagnosed with achalasia. It was recognized as a clinicopathologic entity after the report by Dr. Tom DeMeester of the University of Southern California in 1993.3 Since then, it has been reported in adults, and particularly in children, in the United States and Europe.4 EoE was initially considered a consequence of gastroesophageal reflux disease (GERD), but today it is recognized as an immune disorder that in some cases is potentiated by GERD.5 Diagnosis is suspected when there are intermittent esophageal symptoms that are initially dysfunctional (globus, dysphagia, retrosternal pain) and then structural (persistent dysphagia, retrosternal pain, or food obstruction).6 In both cases, endoscopy enables the identification of subtle, but instructive, mucosal alterations, such as white exudates, edema with linear furrows, wall thinning resembling “crepe paper”, multiple rings or “trachealization”, and/or stricture with circumferential stellar inflammatory exudates.7
Biopsies of the middle third of the esophagus show eosinophilic infiltrate with a minimum density of 15 eosinophils per high power field, degranulation, and eosinophilic microabscesses.8 There are no pathognomonic signs or symptoms and so an integrated diagnosis must be made. Furthermore, there are other causes of esophageal eosinophilic infiltration without EoE, as well as patients with EoE and no endoscopic alterations.7 Ten patients with EoE have been reported in two recent case series in Mexico,9,10 and there have also been reports of isolated cases.11,12
AimThe aim of this study was to describe the clinical characteristics, through endoscopy and histopathology, of Mexican adult patients with EoE, and to identify the predictive elements of the differential diagnosis and compare them with patients presenting with GERD. An additional goal was to evaluate the diagnostic accuracy of esophageal endoscopy in a private practice medical community.
MethodsThe work of 2 studies is presented herein.
- 1.
Clinical differential diagnosis (study 1): A retrospective study was conducted on patients diagnosed with EoE seen within the time frame of September 2006 and June 2015 at a private medical unit in Mexico City and a university hospital in Veracruz. These patients were included in a prospective register for the evaluation of their clinical, endoscopic, and histopathologic characteristics in relation to proton pump inhibitor (PPI) therapy duration and response, specific diet, and/or topical steroids (fluticasone or budesonide).
- 2.
Endoscopic challenge (study 2): A case-control study was conducted on 9 of the above patients. The cases were paired by age and sex with 18 GERD patients, 9 of whom had Barrett's esophagus with no esophagitis. The main clinical manifestations of the two diseases were compared, along with the endoscopic and histopathologic findings, and diagnostic accuracy was then established.
In the two studies, EoE diagnosis was made according to the consensus published in 2011,13 which states that it should include:
- 1.
Symptoms of esophageal dysfunction (globus, dysphagia, retrosternal pain, and food obstruction).
- 2.
Eosinophilic infiltration with a density of more than 15 eosinophils per high power field in the absence of any other differential diagnosis, mainly GERD, excluded due to the lack of treatment response with a PPI for 8 weeks or a 24-h esophageal pH study.
- 3.
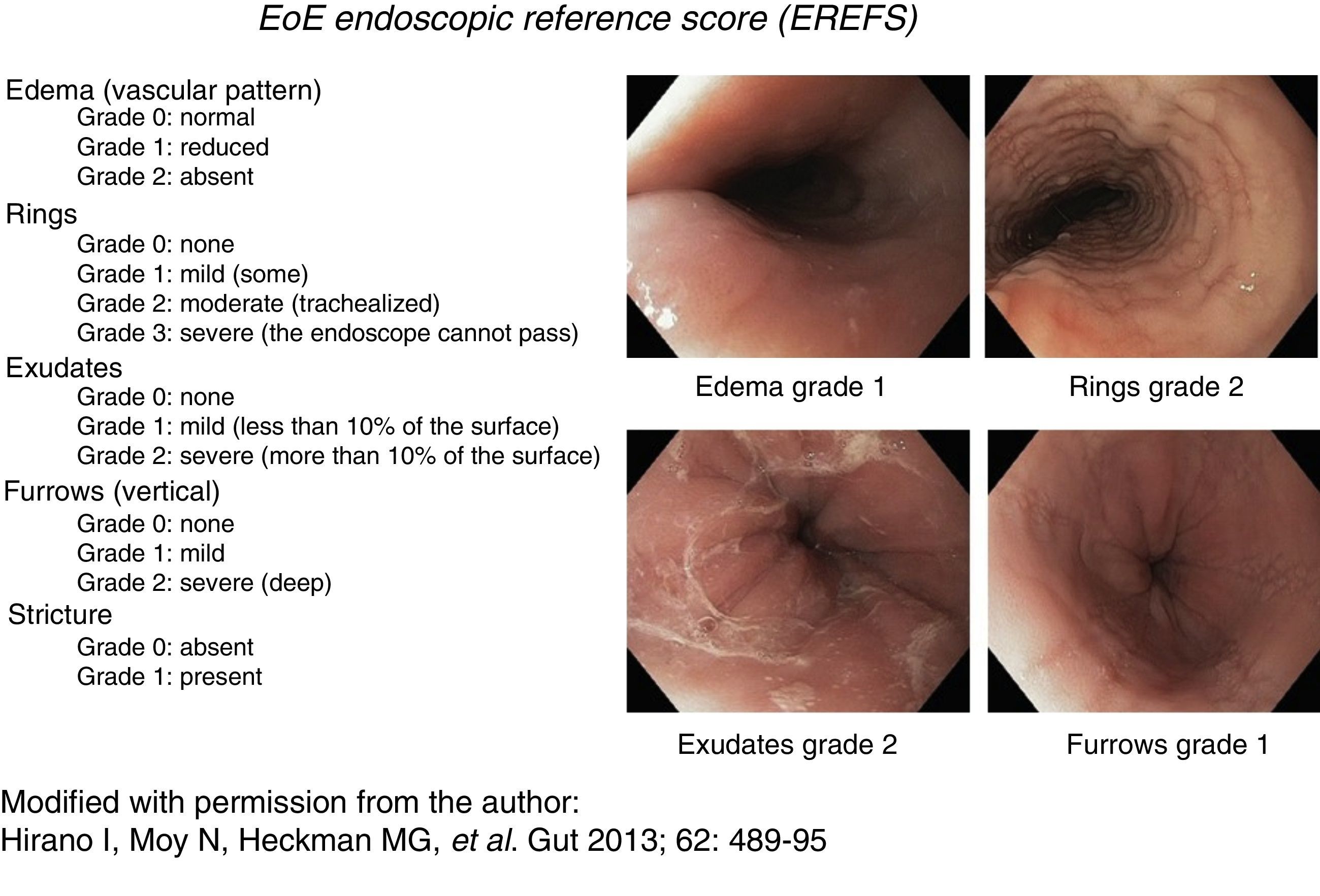
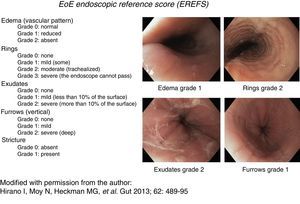
Consistent endoscopic findings such as: inflammatory exudates (punctate, in plaques, or resembling flakes), mucosal edema with linear furrows, reduced esophageal diameter or inflammatory-exudative stricture, easily torn mucosa (called “crepe paper”), and “trachealization” or “feline esophagus”. These findings were used as diagnostic support and were classified and evaluated according to the scale described by Hirano et al.,14,15 currently known as the EREFS reference score (fig. 1). This scale has a minimum value of 0 and a maximum of 10 and assesses 5 characteristics: Edema, Rings, Exudates, Furrows, and Stricture.
Figure 1.Endoscopic reference score for eosinophilic esophagitis with examples. Modified with permission from the author, from Hirano et al., 14.
(0.35MB).
Clinical data were obtained from the case records and correspond to those of the initial consultation, before the first endoscopy. The demographic characteristics, family and personal history of allergic disorders, medication, and paraclinical studies, such as total eosinophil count, serum IgE, C-reactive protein, erythrocyte sedimentation rate, special skin test to study allergies (“atopy patch test”), and 24-h esophageal pH study when deemed necessary, especially in patients being considered for anti-reflux surgery, were all registered. Symptoms for GERD were systematically evaluated using the Carlsson-Dent questionnaire,16 as well as a directed interview for dysphagia, chest pain, and food impaction in relation to their intensity, frequency, and duration, according to the Straumann scale.17 All the patients (EoE and GERD) were endoscopically evaluated with high definition equipment (Olympus EXERA-III, GIF-180H), with a minimum of 8 circumferential esophageal biopsies, 4 in the middle esophagus and 4 in the distal esophagus.
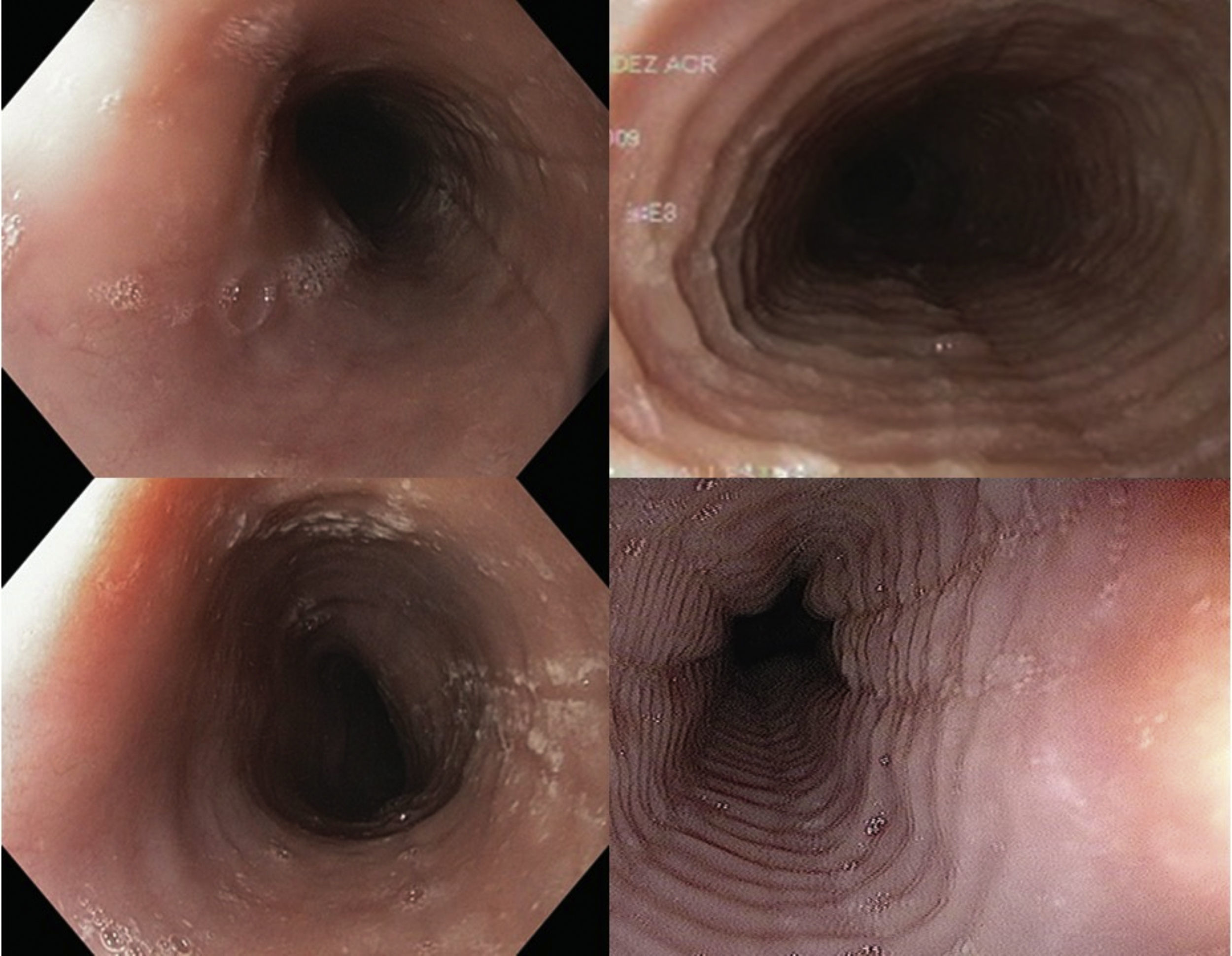
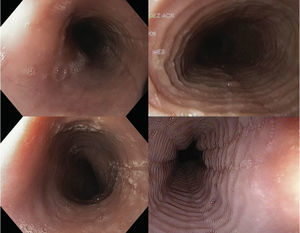
Endoscopic imagesFour digital photographs of the endoscopic findings from the 27 patients included in the case-control study were prepared (fig. 2). These images were mixed, encoded, blinded, and sent with no clinical information to 4 gastroenterologists certified in gastrointestinal endoscopy (ATD, RBA, ELM, MAF) for their diagnostic interpretation (evaluation 1). Once their initial assessment was obtained, the gastroenterologists were given information with 4 illustrative references7,13,18,19 of EoE images to re-evaluate (evaluation 2) the previous images in the same order, blinded, and with no clinical information.
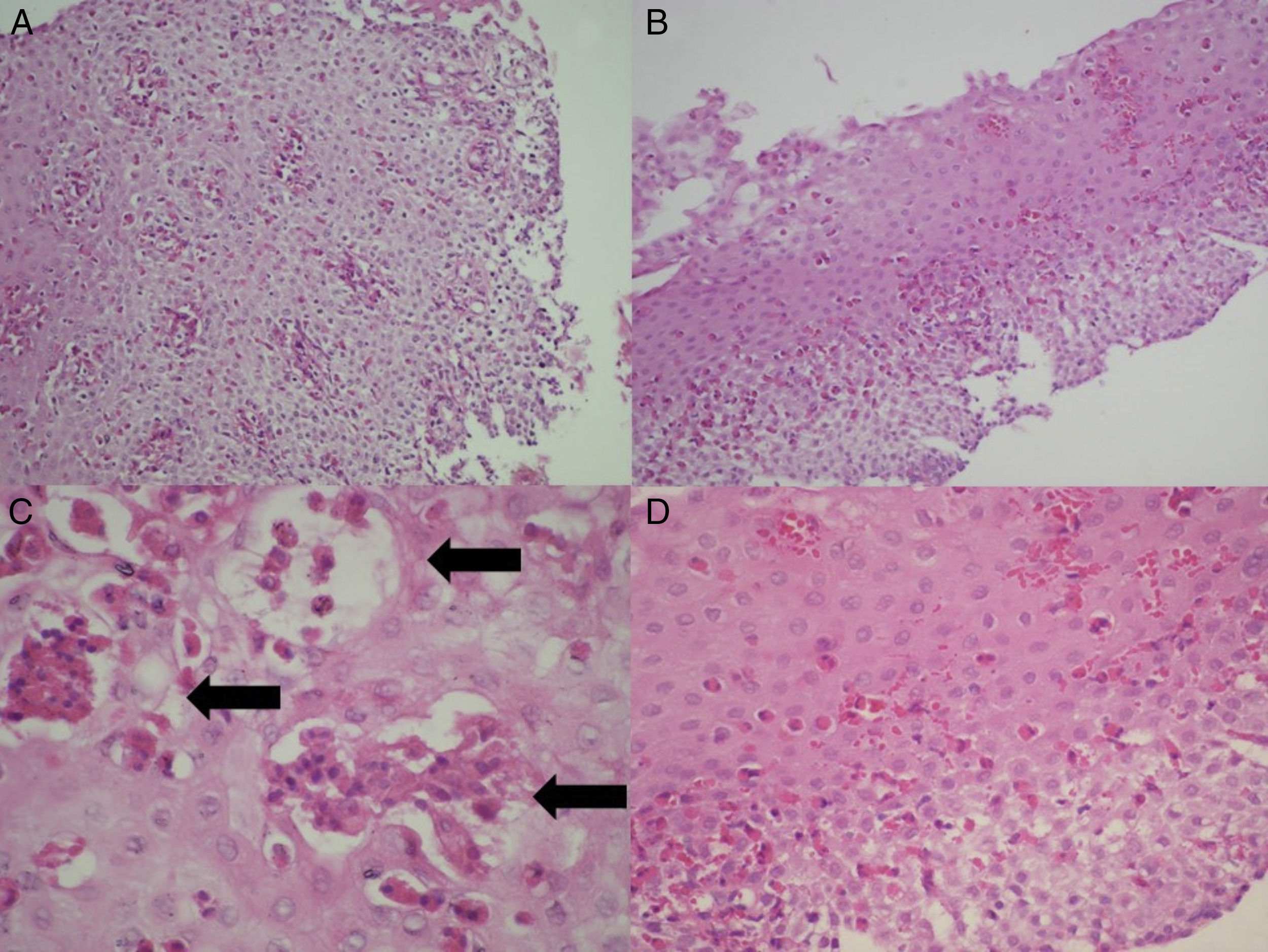
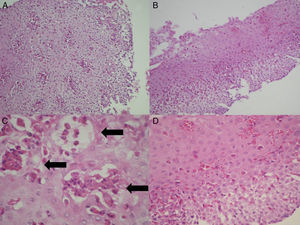
Histologic evaluationThe biopsy slides of the 27 patients were collected, randomized, mixed, and blinded, to be evaluated in a minimum of 5 fields by 2 certified pathologists (CR, CG), establishing their diagnostic correlation in only one evaluation. In addition to the density of eosinophils per high power field, the presence of eosinophilic degranulation, eosinophilic microabscesses (groupings of more than 4 contiguous eosinophils), and the presence of spongiosis or fibrosis of the lamina propria were evaluated (fig. 3). The histologic diagnosis of GERD was made based on the presence of a predominantly polymorphonuclear inflammatory infiltrate, hyperplasia of the papillae, or pseudoacanthosis of the epithelium. Barrett's esophagus was considered in the presence of intestinal metaplasia with goblet cells.
Statistical analysisThe study population was characterized using descriptive statistics. The bivariate analysis was employed to compare the case-control groups. Data were analyzed depending on the outcome variable using nonparametric tests (Fisher's exact text and Mann-Whitney U test), calculating sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) through contingency tables. Cohen's kappa coefficient was calculated for the interpretations of the endoscopies and biopsies. The statistical analysis was carried out using the SPSS (version 20, IBM Corp., Armonk, New York, USA) software program.
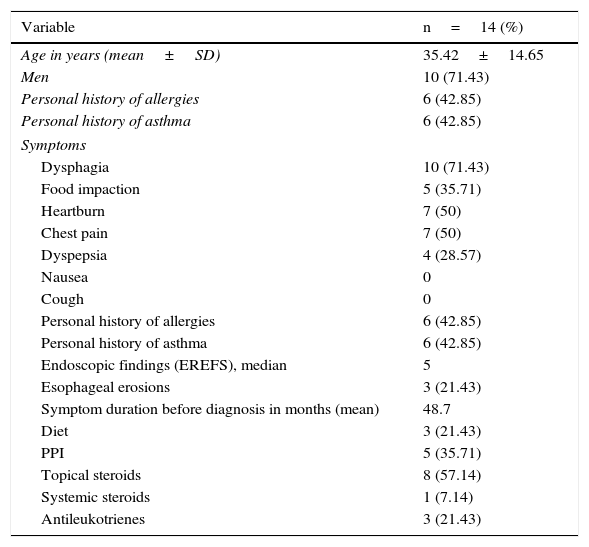
ResultsFourteen patients with confirmed diagnosis of eosinophilic esophagitis were included in study 1 for the description of the case series. The majority of these patients were men (71%) and the mean patient age was 35 years. Forty-two percent of the patients had a past history of allergies and asthma and the predominant symptom was dysphagia (71%). Mean symptom duration prior to diagnosis was 48.7 months, and some of the patients presented with heartburn and intermittent chest pain as the only symptoms. All the patients had endoscopic findings described for EoE and the median EREFS score was 5 points. The most prevalent treatment was topical steroids (57%). One patient received systemic steroids (oral prednisone) prescribed by another physician. The rest of the population characteristics are shown in Table 1.
Characteristics of the patients with eosinophilic esophagitis (n=14).
| Variable | n=14 (%) |
|---|---|
| Age in years (mean±SD) | 35.42±14.65 |
| Men | 10 (71.43) |
| Personal history of allergies | 6 (42.85) |
| Personal history of asthma | 6 (42.85) |
| Symptoms | |
| Dysphagia | 10 (71.43) |
| Food impaction | 5 (35.71) |
| Heartburn | 7 (50) |
| Chest pain | 7 (50) |
| Dyspepsia | 4 (28.57) |
| Nausea | 0 |
| Cough | 0 |
| Personal history of allergies | 6 (42.85) |
| Personal history of asthma | 6 (42.85) |
| Endoscopic findings (EREFS), median | 5 |
| Esophageal erosions | 3 (21.43) |
| Symptom duration before diagnosis in months (mean) | 48.7 |
| Diet | 3 (21.43) |
| PPI | 5 (35.71) |
| Topical steroids | 8 (57.14) |
| Systemic steroids | 1 (7.14) |
| Antileukotrienes | 3 (21.43) |
For study 2, the case-control analysis included 9 of the patients with EoE (which was the total number of cases at that moment) and 18 controls paired by age and sex that presented with GERD. Nine of those controls had confirmed diagnosis of Barrett's esophagus. The mean age for the cases was 33±14 years and 33.6±16.8 years for the controls, of which 66% and 72%, respectively, were men (Table 2). There were more patients that presented with a personal history of asthma (44%) in the EoE group, and the difference with the GERD patients (0%) was statistically significant. In relation to symptoms, the Carlsson-Dent questionnaire showed no difference between the groups when a value of 4 or more was considered positive. However, the patients with EoE had more food impaction, with respect to the GERD patients (37% vs 0%) and the difference was statistically significant, with 100% specificity and a PPV of 100% for the diagnosis of interest (Table 3).
EoE/GERD comparison.
| Variable | EoE (9) | GERD (18) | p |
|---|---|---|---|
| Age (years) | 33±14 | 33.6±16.8 | NS |
| Sex (M) | 6 (66%) | 13 (72%) | NS |
| Asthma | 4 (44%) | 0/18 | 0.0023 |
| Dysphagia | 6 (66%) | 4 (30%) | NS |
| Chest pain | 4 (44%) | 2 (15%) | NS |
| Food impaction | 3 (37%) | 0 | 0.04 |
| Carlsson-Dent questionnaire (> 3) | 4/5 (80%) | 12/12(100%) | NS |
| First endoscopic evaluation | 17/32 (53%) | 5/60 (8%) | < 0.0001 |
| Second endoscopic evaluation | 32/32 (100%) | 17/60 (28%) | < 0.0001 |
Relative diagnostic usefulness of clinical symptoms and endoscopic signs.
| Variable | S (%) | E (%) | PPV (%) | NPV (%) |
|---|---|---|---|---|
| Dysphagia | 66 | 69 | 60 | 75 |
| Chest pain | 50 | 84 | 66 | 73 |
| Food impaction | 37 | 100 | 100 | 72 |
| Carlsson-Dent questionnaire (>3) | 80 | 0 | 25 | 0 |
| First endoscopic evaluation | 53 | 91 | 77 | 78 |
| Second endoscopic evaluation | 100 | 71 | 65 | 100 |
In the initial evaluation of the endoscopic findings, 53% of the interpretations were correct for EoE. Eight percent of the interpretations were falsely positive for EoE in the images of the GERD group (sensitivity 53%, specificity 91%, PPV 77%, NPV 78%). In the second evaluation, the number of correct diagnoses for EoE rose to 100%. However, the number of false positive interpretations in the GERD group also increased to 28% (sensitivity 100%, specificity 71%, PPV 65%, NPV 100%). The kappa statistic for interobserver agreement in the initial image evaluation was 0.32 (observers 1 and 2) and 0 (observers 3 and 4), with a mean of 0.16. In the second image evaluation it was 0.82 (observers 1 and 2) and 0.6 (observers 3 and 4), with a mean of 0.71. Diagnostic agreement of the pathologists was 88% with a kappa statistic of 0.6 for EoE.
DiscussionEoE is seen in Mexico, as well as in the western countries.9–12 Its clinical manifestations are nonspecific and are a product of dysfunction or inflammatory stricture. They are often indistinguishable from those that cause dysfunction and stricture in GERD. The distinction between EoE and GERD is often difficult, especially given the latter's high frequency, resulting in the coexistence of the two conditions in which GERD can potentiate EoE. This has led some experts to consider EoE as an additional complication of GERD.5 Furthermore, up to half of the patients with esophageal eosinophilia have a clinical, histologic, and histopathologic response to PPI administration,20 known as PPI-responsive esophageal eosinophilia. This disorder is not completely understood, but is known to share genetic, pathophysiologic, and proteomic features with EoE, and not with GERD.
In the case series presented herein, EoE was identified and diagnosed according to the guidelines of the 2011 North American consensus, and GERD was excluded as the cause of the eosinophilia documented in the middle third of the esophagus, after a minimum 2-month PPI therapy, and in 3 patients, also after 24-h pH study.
Nevertheless, distinguishing and specifying these entities is sometimes impossible. The anti-inflammatory action of PPIs has recently been recognized in regard to their usefulness in esophageal eosinophilia. However, it must be emphasized that treatments for GERD (PPI and antireflux surgery) are insufficient for EoE and, more importantly, surgical treatment can worsen dysphagia in these patients.21 Thus, patients with refractory esophageal symptoms should have a thorough diagnostic evaluation before receiving any additional therapeutic intervention.
Our report is the largest case series conducted in Mexico and describes the clinical and endoscopic manifestations of EoE. The study also compares EoE and GERD patients in a search for keys to their differential diagnosis. This comparison was previously explored by Dellon et al. in North Carolina.7 Their study included 151 patients with EoE and compared them with 226 patients with GERD. They demonstrated how eosinophilic infiltration of the esophageal mucosa could be predicted when there was younger age, a higher frequency of dysphagia, white exudates, esophageal rings and/or furrows in the mucosa, as well as absence of hiatal hernia. With these data, a useful diagnostic index was calculated, albeit complex and impossible to carry out in daily clinical practice. The guiding data in our study were the presence of asthma and food impaction. The history of asthma coincided with that reported in other case series, including a previously published Mexican study.10
In two prospective studies on patients that underwent endoscopy due to dysphagia, there was a 10 to 15% prevalence of EoE.22,23 In those studies, in a total of 33 patients, Prasad et al. documented the predominance of men, age under 40 years, history of food impaction, and endoscopic images of esophageal rings, linear furrows, stricture, or white exudates. In 31 patients, McKenzie et al. identified young patients with a past history of food allergies and asthma.
In our case series of Mexican patients there was also a predominance of young men (10/14 with a mean age of 35 years), a past history of allergies, asthma, rhinitis-sinusitis or atopic dermatitis (6/14), and the presentation of esophageal symptoms. However, the data analyzed were also insufficient for differentiating EoE from GERD, despite examining the clinical indices designed to objectively qualify esophageal symptoms, such as the Carlsson-Dent scale for GERD and the Straumann scale for EoE.
In Mexico, EoE has been considered an exceptional diagnosis and thought to be a consequence of differences in diet or the genetic polymorphisms resulting from the mixed racial ancestry in the Mexican population. It should be mentioned that the predominant phenotype in our study population was Indo-European, even though 2 of the patients were clearly mestizos.
A recent multi-center retrospective study of referral hospitals in the United States showed a lower prevalence of EoE in African Americans and Hispanic Americans.24 From a total of 793 patients, 660 were white (83%), 77 black (10%), 18 of Latin American origin, and only 2 were American Indian. Comparing the symptoms, there was no significant difference between men and women, but there was a significant difference between white subjects and those of other races. The relative frequency of dysphagia (75 vs 50%) and food impaction (35 vs 13%) was lower in black subjects and those of Latin American origin, but they had a predominance of GERD manifestations, even in the presence of EoE. There was also a lower prevalence of stricture and esophagi with trachealization in blacks and Hispanics (46 vs 20%), with a relatively higher frequency in the latter of white plaques and edema with markings or furrows. Those authors concluded that there could be a lower intensity or perception of complaints suggestive of EoE in the cases of these minority populations.
This phenomenon has been observed in other developing countries considered to have low prevalence of the disease. In a study conducted in Saudi Arabia,25 8 cases of EoE were documented in adults over a 7-year period. The patient characteristics were similar to those described in North America and Europe. The authors concluded that it is unclear whether the low prevalence was due to a lack of disease suspicion, and that this has changed in recent years, resulting in an increase of reported cases. With this in mind, our study showed how 4 Mexican gastroenterologists-endoscopists initially interpreted 60% of the EoE cases correctly in a blinded evaluation of endoscopic images in patients with EoE and GERD. However, when key information on the EoE images was provided, correct interpretation was 100%. In our opinion, this intervention did not show the gastroenterologists typical images of EoE, but rather made them think more about EoE diagnosis by observing the subtle alterations (white exudates or edema with linear furrows). The lack of diagnostic accuracy of endoscopic images in patients with EoE has been greatly stressed, but this could improve with the systematic examination proposed by Hirano et al.14,15 More than 90% of the patients with EoE have images characteristic of eosinophilic infiltration, making the usefulness and precision of endoscopy much greater in this group of patients than in those with GERD. Only 15 to 30% of case series of GERD patients have images of marked esophagitis and in many cases there is no esophageal damage at all.26,27
Another possible explanation of the lower prevalence could be the quality of medical care that this group of marginalized patients receives. Insufficient semiology of esophageal complaints could be the cause of lower detection. In Mexico it is common practice for the clinical gastroenterologist to order the study, and for the specialist in gastrointestinal endoscopy to perform the examination without having interviewed the patient. A reflection of this could be the long period of progression in the EoE patients included in our case series, which was a mean 4 years before diagnosis was made. The directed interview as to the presence of dysphagia, and if that is the case, a detailed anamnesis of its frequency, intensity, and association with food impactions, guide the endoscopic evaluation, given that the lesions are often faint and nonspecific or even absent. Thus, when there is clinical suspicion, biopsies of the middle third of the esophagus are required. The frequency of these complaints in our case series fully coincided with those reported in Caucasians in the medical literature (70% dysphagia, 50% chest pain, and 35% food impaction). The majority of our study population was of European origin and therefore did not represent the number of races and racial mixing in the Mexican population, thus being a possible source of bias. Nevertheless, given the lower frequency of complaints suggestive of EoE in blacks and Hispanics, the patients in our environment with a history of GERD and associated atopic diseases, particularly those with dysphagia or food impaction, should be studied for eosinophilia, taking biopsies of the middle third and the distal portion of the esophagus, especially when there is no satisfactory PPI response.
In conclusion, our case series describes 14 patients with EoE according to the criteria established by the North American diagnostic consensus. The EoE symptoms are the same as those reported in European and North American populations. All our patients had macroscopic endoscopic alterations that enabled histopathologic diagnosis. In the blinded evaluation, diagnostic sensitivity increased from 53 to 96% when the disorder was contemplated and there was awareness of the abnormalities characteristic of esophageal eosinofilia. There are no specific symptoms for EoE diagnosis, but subtle alterations should be searched for at endoscopy in young men with a history of atopy and food impaction to confirm the diagnosis through histopathology.
Ethical responsibilitiesProtection of persons and animalsThe authors declare that the procedures followed conformed to the ethical standards of the responsible committee on human experimentation and were in accordance with the World Medical Association and the Declaration of Helsinki.
Data confidentialityThe authors declare that no patient data appear in this article.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Financial disclosureNo financial support was received in relation to this study.
Conflict of interestThe authors declare that there is no conflict of interest.
The authors wish to thank all of the gastroenterologists that participated in the study.
Please cite this article as: Soto-Solís R, Santana-de Anda K, González-Uribe N, Gallegos C, Romo-Aguirre C, Remes-Troche JM, et al. Cómo mejorar el diagnóstico de esofagitis eosinofílica: experiencia de una serie de casos en México. Revista de Gastroenterología de México. 2017;82:5–12.
See related content at DOI: 10.1016/j.rgmxen.2016.10.002, Souza RF. Diagnosing eosinophilic esophagitis: cytokine sizzleand fizzle - Mexican style. Revista de Gastroenterología de México.2017;82:1–4.