Bariatric surgery is the best method for treating obesity and its comorbidities. Our aim was to provide a detailed analysis of the perioperative outcomes in Mexican patients that underwent surgery at a high-volume hospital center.
Materials and methodsA retrospective study was conducted on all the patients that underwent bariatric surgery at a single hospital center within a time frame of 4 and one-half years. Demographics, the perioperative variables, complications (early and late), weight loss, failure, and type 2 diabetes mellitus remission were all analyzed.
ResultsFive hundred patients were included in the study, 83.2% of whom were women. Mean patient age was 38.8 years and BMI was 44.1kg/m2. The most common comorbidities were high blood pressure, dyslipidemia, and diabetes. Laparoscopic gastric bypass surgery was performed in 85.8% of the patients, sleeve gastrectomy in 13%, and revision surgeries in 1%. There were 9.8% early complications and 12.2% late ones, with no deaths. Overall weight loss as the excess weight loss percentage at 12 and 24 months was 76.9 and 77.6%. The greatest weight loss at 12 months was seen in the patients that underwent laparoscopic gastric bypass. A total of 11.4% of the patients had treatment failure. In the patients with type 2 diabetes mellitus, 68.7% presented with complete disease remission and 9.3% with partial remission. There was improvement in 21.8% of the cases.
ConclusionsIn our experience at a high-volume hospital center, bariatric surgery is safe and effective, based on the low number of adverse effects and consequent weight loss and type 2 diabetes mellitus control. Long-term studies with a larger number of patients are needed to determine the final impact of those procedures.
La cirugía bariátrica es el mejor método contra la obesidad y sus comorbilidades. El objetivo fue analizar detalladamente los resultados perioperatorios y la evolución de pacientes latinos sometidos a cirugía en un centro de alto volumen.
Material y métodosEstudio retrospectivo, con todos los pacientes sometidos a cirugía bariátrica en un solo centro, durante 4 años y medio. Se realizó análisis demográfico, perioperatorio, de complicaciones (tempranas y tardías), pérdida de peso, falla y remisión de diabetes mellitus tipo 2.
ResultadosSe incluyeron 500 pacientes, un 83.2% mujeres, con edad media de 38.8 años e IMC de 44.1kg/m2. Las comorbilidades mayormente observadas fueron hipertensión, dislipidemia y diabetes. Se realizó bypass gástrico laparoscópico en el 85.8%, manga gástrica en el 13% y cirugía revisional en el 1%. Hubo un 9.8% de complicaciones tempranas y un 12.2% de tardías, sin mortalidad. La pérdida de peso global en porcentaje de exceso de peso perdido a 12 y 24 meses fue del 76.9% y 77.6%; el BPGL tuvo mayor pérdida de peso a los 12 meses. La falla al tratamiento fue del 11.4%. En los pacientes con DM2 existió remisión completa en el 68.7%, remisión parcial en el 9.3% y una mejoría en el 21.8% de los casos.
ConclusionesEn nuestra experiencia como centro de alto volumen, la cirugía bariátrica es segura y efectiva basada en el bajo número de efectos adversos, y la pérdida de peso con control de la DM2. Estudios a largo plazo y con un mayor número de pacientes son necesarios para determinar el impacto final de dichos procedimientos.
Obesity is a chronic disease that continues to expand worldwide and is now considered an epidemic. It affects 33.3% of the Mexican adult population,1,2 positioning the country in second place in the world in the number of patients with obesity, just behind the United States. Obesity and its associated diseases lead to a progressive increase in morbidity and mortality, reducing life expectancy.3,4
Bariatric surgery is the best treatment for those problems, resulting in important and sustained weight loss in the majority of patients.4–7 Among the bariatric procedures, laparoscopic gastric bypass (LGB) and laparoscopic sleeve gastrectomy (LSG) are the most widely performed across the world.8,9 They are considered safe procedures, with a reported mortality < 0.5% and early morbidity (30 days) of 7-23%.10–12 Even though a large part of the reduction in morbidity and mortality is associated with the advent of the laparoscopic era, there has also been a positive impact from a systematization of processes and patient management in high-volume centers.13 Regarding the Mexican population, there are few studies, and those that exist have short follow-up periods,14–16 or focus exclusively on surgical techniques,17–19 revision surgeries,20,21 metabolic surgery, 22–24 and postoperative changes.25,26 The aim of the present study was to describe the overall result of the first 500 patients operated on at a high-volume center dedicated exclusively to the management of obesity, evaluating the numerous variables of great importance.
Materials and methodsA retrospective study captured prospective data and analyzed the case records of all patients that underwent bariatric surgery within the time frame of January 1, 2013 to May 31, 2017 at a single center. The preoperative (demographics, anthropometry, and comorbidities), perioperative (types of surgery, days of hospital stay, and early and late complications), weight loss at 12 and 24 months measured by body mass index (BMI), and percentage of excess weight loss (EWL%) were described. A sub-analysis between LGB and LSG was carried out that compared weight loss at 12 months, as well as the report of patients with weight loss failure at 24 months and the prevalence of comorbidities in those patients. Secondarily, an independent analysis was done on the remission (partial or total) or improvement of type 2 diabetes mellitus (DM2) in patients that reached one year of follow-up and had complete laboratory tests.
Patient selectionPatient selection was based on the statutes of the U.S. National Institutes of Health (NIH) and the Mexican Official Norm NOM-008-SSA3-2010 for the comprehensive treatment of overweight and obesity.27,28 Informed consent, specifically for bariatric surgery, was obtained from all the patients and all procedures were approved by a multidisciplinary team (psychology, nutrition, physiotherapy, and endocrinology). The type of procedure was chosen in conjunction with the multidisciplinary team, according to patient characteristics and preferences, and the risks and benefits of each surgery were thoroughly explained to the patients beforehand (except if there were formal contraindications for a specific type of surgery).
Surgical techniqueThe “simplified” Roux-en-Y LGB was performed as previously described,17 with an antecolic segment and mechanical anastomosis calibrated at 2cm. The biliary and digestive tract segments measured 70 and 150cm, respectively. Both anastomoses were performed latero-laterally with linear staplers (staple height was 2.5mm). The major omentum was divided and both mesenteric spaces were closed. The methylene blue test and drain placement were systematically carried out.
LSG was performed at 5-6cm from the pylorus with 36-Fr calibration. The first two shots (60-mm staplers) were systematically performed with staples with 4.1mm staple height. The following shots (3-4 extra shots) were done with staples with 3.5mm staple height. The stapled line in all the cases was over-stitched with nonabsorbable suture (2-0 polypropylene). The methylene blue test and drain placement were systematically carried out.
Type 2 diabetes mellitus remissionThe definitions of DM2 remission and improvement were based on those suggested by the American Diabetes Association in their criteria established in 2009 and by the Clinical Practice Guidelines. Partial remission was defined as hyperglycemia under the DM2 threshold (100-125mg/dl) and HbA1c < 6.5% with no medications, for at least one year. Complete remission was defined as normal glycemia (< 100mg/dl) and HbA1c < 5.7% with no medication, for at least one year. Improvement was defined as the reduction in the number and/or dose of medications (including insulin) associated with better control of glucose values.29–31
Statistical analysisResults were expressed as mean ± standard deviation (SD) or percentage, depending on the variable. The Student's t test was used for the independent or dependent variables, as required. The categorical variables were compared through the chi-square test for distribution. Statistical significance was set at a p < 0.05. The statistical analysis was performed using the NCSS 2007 (NCSS, Kaysville, Utah, USA) program. The present study was exempt from the corresponding committee review due to its retrospective design.
ResultsOver a period of 4 years and 6 months, 505 procedures were performed, 5 (0.9%) of which were not completed due to intraoperative findings or complications that included: 2 giant wall hernias with loss of content, one case of macronodular cirrhosis, one gastrointestinal stromal tumor in the stomach, and one trocar-associated hematoma in the mesentery.
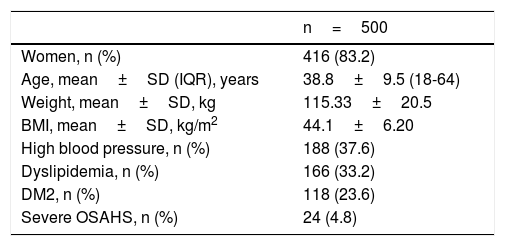
The final analysis was carried out on 500 patients. A total of 83.2% were women, mean patient age was 38.8 ± 9 years, and mean BMI was 44.1 ± 6.2kg/m2. The 3 most widely-observed comorbidities were high blood pressure (37.6%), dyslipidemia (33.2%), and DM2 (23.6%). Table 1 shows the rest of the initial demographics.
Initial demographic and comorbidity analysis of all patients that underwent bariatric surgery.
| n=500 | |
|---|---|
| Women, n (%) | 416 (83.2) |
| Age, mean±SD (IQR), years | 38.8±9.5 (18-64) |
| Weight, mean±SD, kg | 115.33±20.5 |
| BMI, mean±SD, kg/m2 | 44.1±6.20 |
| High blood pressure, n (%) | 188 (37.6) |
| Dyslipidemia, n (%) | 166 (33.2) |
| DM2, n (%) | 118 (23.6) |
| Severe OSAHS, n (%) | 24 (4.8) |
BMI: body mass index; DM2: type 2 diabetes mellitus; IQR: interquartile range; N: number; OSAHS: Obstructive sleep apnea-hypopnea syndrome; SD: standard deviation.
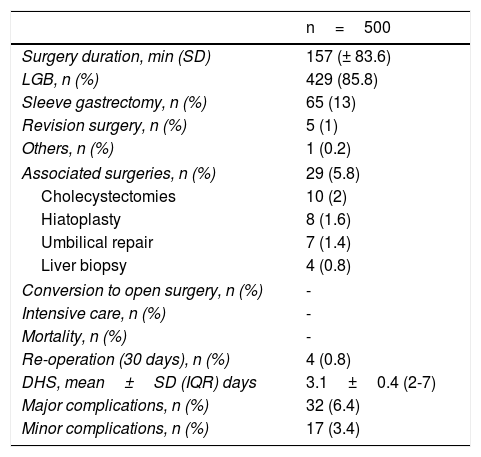
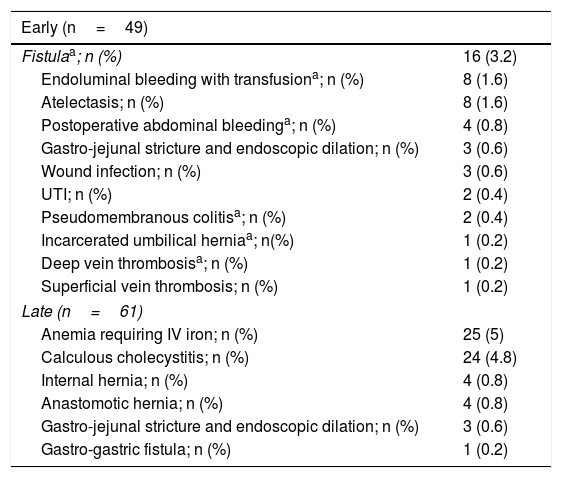
Regarding the perioperative analysis, LGB was performed in 85.8% of the patients and LSG in 13%. Revision surgery (conversion of a previous bariatric surgery to a different type) was carried out in 1% of the cases. Other procedures associated with bariatric surgery (cholecystectomy, hiatoplasty, wall repair, and liver biopsy) were performed in 5.8% of the cases. There were no conversions to open surgery, admission to the intensive care unit, or deaths (Table 2). In relation to adverse effects, there were a total of 49 (9.8%) early complications (< 30 days), 6.4% of which were major. The early re-operation rate was 0.8%. There were 61 (12.2%) late (after 30 days) complications, the main ones of which were anemia, calculous cholecystitis, and internal hernia (Table 3). The late re-operation rate was 3.4%. Mean hospital stay was 3.1±0.4 days for patients with no complications and 3.7±1 days for patients that presented with a complication.
Perioperative period and complication analysis in all the patients that underwent bariatric surgery.
| n=500 | |
|---|---|
| Surgery duration, min (SD) | 157 (± 83.6) |
| LGB, n (%) | 429 (85.8) |
| Sleeve gastrectomy, n (%) | 65 (13) |
| Revision surgery, n (%) | 5 (1) |
| Others, n (%) | 1 (0.2) |
| Associated surgeries, n (%) | 29 (5.8) |
| Cholecystectomies | 10 (2) |
| Hiatoplasty | 8 (1.6) |
| Umbilical repair | 7 (1.4) |
| Liver biopsy | 4 (0.8) |
| Conversion to open surgery, n (%) | - |
| Intensive care, n (%) | - |
| Mortality, n (%) | - |
| Re-operation (30 days), n (%) | 4 (0.8) |
| DHS, mean±SD (IQR) days | 3.1±0.4 (2-7) |
| Major complications, n (%) | 32 (6.4) |
| Minor complications, n (%) | 17 (3.4) |
DHS: days of hospital stay; IQR: interquartile range; LGB: laparoscopic gastric bypass; min: minutes; n: number
Early and late postoperative complications in 500 patients that underwent bariatric surgery.
| Early (n=49) | |
|---|---|
| Fistulaa; n (%) | 16 (3.2) |
| Endoluminal bleeding with transfusiona; n (%) | 8 (1.6) |
| Atelectasis; n (%) | 8 (1.6) |
| Postoperative abdominal bleedinga; n (%) | 4 (0.8) |
| Gastro-jejunal stricture and endoscopic dilation; n (%) | 3 (0.6) |
| Wound infection; n (%) | 3 (0.6) |
| UTI; n (%) | 2 (0.4) |
| Pseudomembranous colitisa; n (%) | 2 (0.4) |
| Incarcerated umbilical herniaa; n(%) | 1 (0.2) |
| Deep vein thrombosisa; n (%) | 1 (0.2) |
| Superficial vein thrombosis; n (%) | 1 (0.2) |
| Late (n=61) | |
| Anemia requiring IV iron; n (%) | 25 (5) |
| Calculous cholecystitis; n (%) | 24 (4.8) |
| Internal hernia; n (%) | 4 (0.8) |
| Anastomotic hernia; n (%) | 4 (0.8) |
| Gastro-jejunal stricture and endoscopic dilation; n (%) | 3 (0.6) |
| Gastro-gastric fistula; n (%) | 1 (0.2) |
IV: intravenous; UTI: urinary tract infection
The EWL% at 12 and 24 months was 76.9% and 77.6%, respectively. The analysis per procedure showed a baseline BMI of 40.4 ± 5.3kg/m2 for LGB vs. 38.5 ± 8.1kg/m2 for LSG (p=0.823). The EWL% at 12 months in the LGB and LSG comparison was 77.1±16.3% vs. 67.9±16.8% (p=0.035), respectively, corresponding to a BMI of 28.8±4kg/m2 for LGB and 30.5±4.1kg/m2 for LSG (p < 0.001).
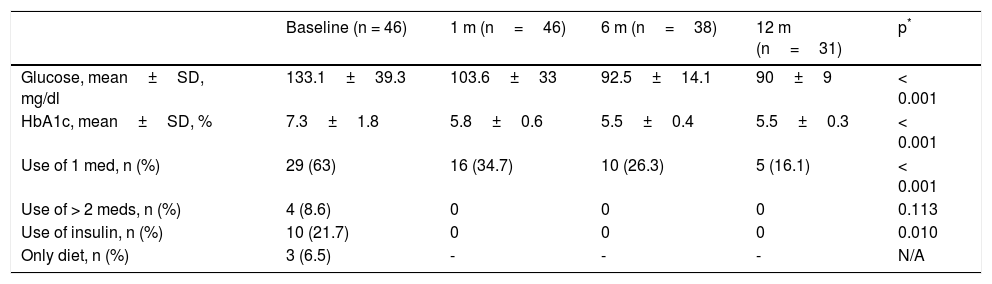
The main follow-up in the study focused on the first 24 months, which was reached by 259 patients. Of those patients, only 122 (47.1%) came to their follow-up appointment. There was surgery failure (EWL% less than 50%) in 7.4% of the cases (9 patients) who had an average EWL% of 30.8%. The prevalence of comorbidities at 24 months was 18.9% for the patients with DM2/prediabetes, 3.3% for those with high blood pressure, and 4.1% for patients with dyslipidemia. We found improvement in obstructive sleep apnea-hypopnea syndrome (OSAHS) in 100% of the patients, defined as interruption in the use of nasal continuous positive airway pressure (CPAP). The sub-analysis of patients with DM2 and complete follow-up with laboratory tests and medication use at 12 months (n=46) showed significant improvement in glucose, glycosylated hemoglobin, and medication use (Table 4). There was complete remission in 68.7% of the patients, partial remission in 9.3%, and improvement in 21.8% of the cases.
Metabolic progression and medication use in patients with DM2 and complete studies at 12 months.
| Baseline (n = 46) | 1 m (n=46) | 6 m (n=38) | 12 m (n=31) | p* | |
|---|---|---|---|---|---|
| Glucose, mean±SD, mg/dl | 133.1±39.3 | 103.6±33 | 92.5±14.1 | 90±9 | < 0.001 |
| HbA1c, mean±SD, % | 7.3±1.8 | 5.8±0.6 | 5.5±0.4 | 5.5±0.3 | < 0.001 |
| Use of 1 med, n (%) | 29 (63) | 16 (34.7) | 10 (26.3) | 5 (16.1) | < 0.001 |
| Use of > 2 meds, n (%) | 4 (8.6) | 0 | 0 | 0 | 0.113 |
| Use of insulin, n (%) | 10 (21.7) | 0 | 0 | 0 | 0.010 |
| Only diet, n (%) | 3 (6.5) | - | - | - | N/A |
HbA1c: glycosylated hemoglobin; med: medication; n: number; N/A: not applicable; SD: standard deviation.
The results of the present retrospective study on 500 patients that mainly underwent gastric bypass surgery and sleeve gastrectomy at a single center were similar to those reported worldwide, especially to results from referral centers in highly industrialized countries. In relation to type of surgery, LGB resulted in a higher EWL% at 12 months.
The performance of bariatric surgery began more than 50 years ago, but the manner in which it is carried out is quite different today, thanks to the advances in science, technology, patient selection, understanding of the disease, and surgical technique refinement.32 The great watershed in bariatric surgery was the application of laparoscopic techniques, with the first case of laparoscopic gastric bypass being reported in 1994,33 initiating a gradual decline in perioperative morbidity and mortality.
Its efficacy in relation to weight loss, control of comorbidities, and durability of results (compared with conservative management) has been shown for 30 years, thanks to the results of the Swedish Obese Subjects analysis.4,7 That study on thousands of patients demonstrated the positive impact of bariatric surgery, especially gastric bypass in the short, medium, and long term. An important characteristic of said study was the low morbidity (13%) and mortality (0.2%) rates, which were lower than those previously reported with the performance of open procedures. The Swedish Obese Subjects study continues to be an international guide, with multiple subsequent and long-term studies.4,34,35
In addition to those advances that greatly increased the safety of the bariatric procedures, better results have also been demonstrated in patients operated on at high-volume centers.36–38 A systematic review that included almost 500,000 patients described a reduction in morbidity and mortality, early re-admission, hospital stay, and hospital costs.13 A definition for a high-volume center has not been established in Mexico, but in the United States, the American College of Surgeons and the American Society for Metabolic and Bariatric Surgery implemented Bariatric Surgery Centers of Excellence hospital accreditation criteria. Said high-volume hospitals perform at least 125 procedures annually and have the resources of bariatric equipment and specialized personnel. Academic training programs have also had a positive impact on patient progression.37–40 Based on the above, we consider our clinic a high-volume center, given that approximately 135 surgeries are performed annually. Only in its first year of operation (2013) were fewer surgeries performed (82 LGBs and 2 LSGs).
Regarding safety, an average mortality rate of 0.3% and a severe adverse effect rate of 4.3% (including death, deep vein thrombosis, pulmonary embolism, re-intubation, requirement of endoscopy, interventional radiology, or reoperation) have been described.36,41 For high-volume centers, general early morbidity is reported at 17% (7-23%), and specifically for LGB at 21%.11,13 Depending on the time at which complications present, they are classified as early (first 30 days) and late. Rosenthal et al. reported complications at 31.8%, 18.7% of which were early and 9.4% were late.10 In our case series, the complication rate was 22%, with early complications at 9.8% and late ones at 12.2%. Even though our study had an apparently higher rate of late complications, that was because the Rosenthal study only reported complications directly related to the surgery (stricture, ulcers, trocar-associated hernias), whereas in addition to those complications, we reported anemia, gallbladder problems, and internal hernias. The majority of case series do not include minor complications. In our study we mainly found atelectasis, surgical site infection, and anastomotic stricture with no surgical management. The most common major complication was fistula (3.2%), but only one case required early re-intervention. The remaining cases were managed conservatively with fistula closure at an average of 2.5 weeks. The second most common complication was bleeding (extraluminal and intraluminal) that required transfusion.
There are numerous types of bariatric surgery.32 Some have gone into disuse (vertical banded gastroplasty, the Scoppinaro technique, and gastric banding), others are considered classic (LGB, LSG, and duodenal switch), while still others are investigational (such as gastric plication, electric implants, mini-gastric bypass, and ileal interposition).42 Even though LGB has been considered the gold standard, sleeve gastrectomy has displaced bypass as the most widely performed technique worldwide since 2016.9 That is related to its “technical simplicity”, shorter surgery duration, and lower long-term complication rate.32 On the other hand, the majority of studies show that LGB has better results with respect to weight loss, lower failure and revision rates, and better metabolic control.6,32 LSG was gradually adopted at our center and mostly performed on patients with a low BMI and no diseases, surgical high-risk patients, and patients with autoimmune diseases. Even though a larger number of LSGs have been performed at our clinic over the last 2 years, more LGBs continue to be carried out, with a bypass/sleeve ratio of 7:1.
Duodenal switch (DS) has been established as the most effective surgery in relation to weight loss, but it is technically demanding, has a higher percentage of complications, and lower long-term quality of life due to extreme and lasting malabsorption.3,5,6,43 LGB provides fewer effects than DS, but it is more tolerable because it is not as malabsorptive. Weight loss at 12 months of about 70-80% EWL is reported worldwide for LGB and of 51-80% for LSG,3,11,32 results which are similar to ours.
One of the reasons that the performance of bariatric surgery has increased is its impact on associated diseases, especially DM2. In Mexico, DM2 has reached epidemic proportions, with alarming prevalence figures of 9.4%.1 Similar to its effect on weight loss, bariatric surgery is the best treatment for the control and remission of DM2 in patients with obesity.44–46 It even forms part of the management algorithm in the latest guidelines and algorithms of the most important international associations.47–48 Remission rates have changed due to modifications in the definitions. The first case series described remission in > 85% of patients,5,49 but there was discrepancy in the manner of reporting. In 2009, the American Diabetes Association modified the established criteria, and current remission rates are reported at 45-70%.29,50,51 Nevertheless, cutoff levels for HbA1c are still a subject of debate. Lower cutoff values for HbA1c were used to define remission in the latest guidelines, which is why we used the stricter model of HbA1c of 5.7% in our analysis. We found complete remission in 68.7% of the patients, and those that required insulin did not use it again after hospital release, before any weight loss, due to the hormonal effect of incretins (especially GLP-1), which are responsible for the greater impact on postoperative metabolic control.52 Likewise, not all metabolic benefits are related to weight loss. Other factors, such as a prolonged period of time with obesity and DM2, the number medications used prior to surgery, advanced age, male sex, longer visceral adipocytes, and a higher number of associated comorbidities, are also influential.53
Follow-up and patient loss are common problems in bariatric surgery. The authors of a recent meta-analysis found that patient follow-up is inadequate in 60% of the bariatric literature, with the probable causes of economic origin, logistics, and a lack of education and communication with the patients.54 To improve patient follow-up at our clinic, the following has been implemented: a list with complete patient data, an emergency number patients can call 24/7, flexibility in the scheduling of appointments, and personalized attention. The creation of support groups, the use of technology, and better collaboration with primary care centers must be emphasized.
In conclusion, it is our experience that bariatric surgery is safe and effective, based on the low number of complications and on weight loss associated with important metabolic control. LGB surgery had the best weight-loss results during the first year. The main limitations of our study were its retrospective design and the fact that the focus of the follow-up period was the first 24 months, together with the loss of patients during follow-up. However, there are no other similar studies on a Mexican population.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Financial disclosureNo financial support was received in relation to this study/article.
Conflict of interestThe authors declare that there is no conflict of interest.
Please cite this article as: Guilbert L, Joo P, Ortiz C, Sepúlveda E, Alabi F, León A, et al. Seguridad y eficacia de la cirugía bariátrica en México: análisis detallado de 500 cirugías en un centro de alto volumen. Revista de Gastroenterología de México. 2019;84:296–302.