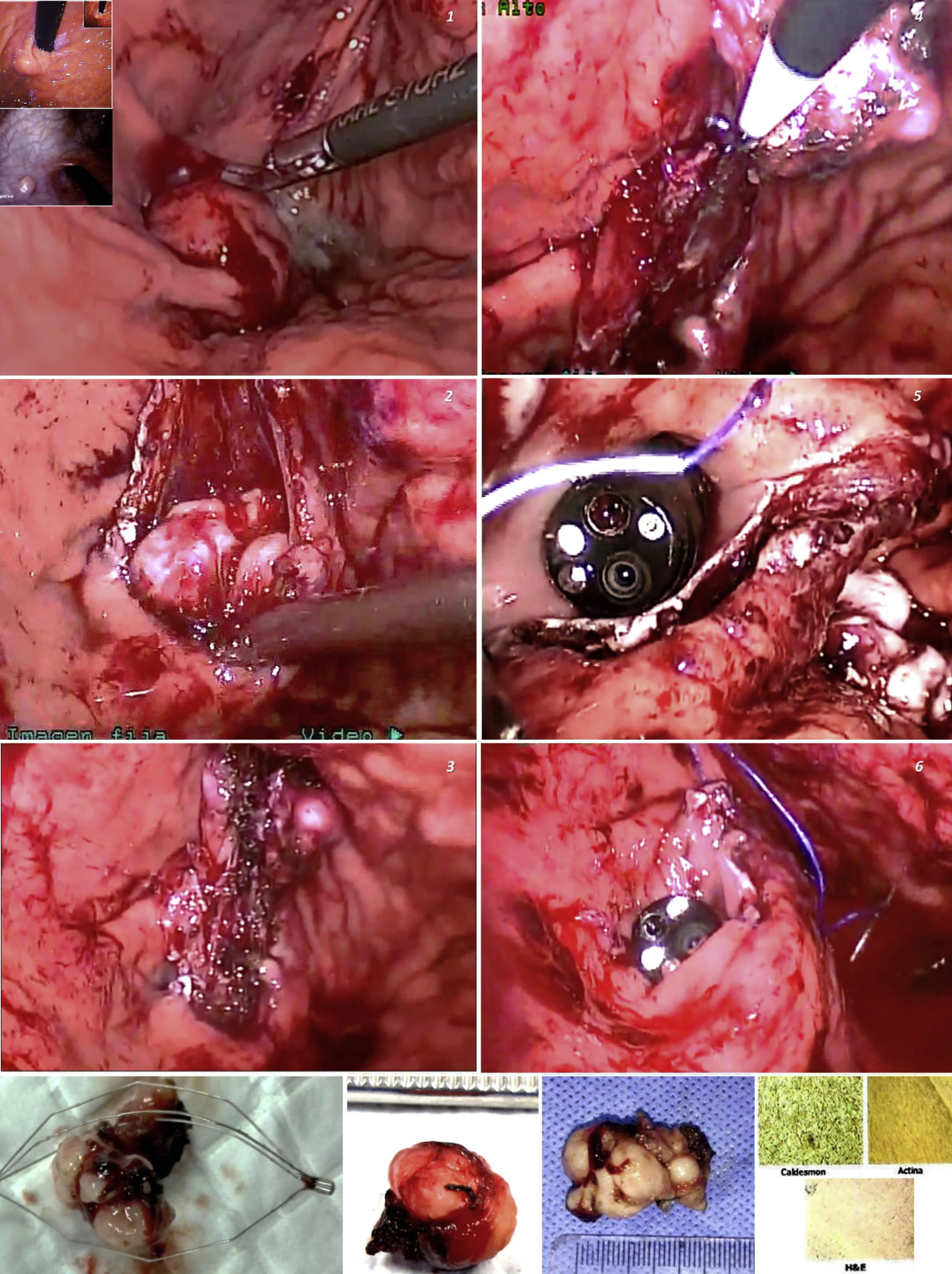
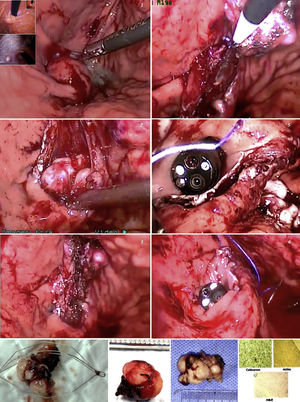
About one-fifth of gastric subepithelial lesions (SLs) are leiomyomas.1 Those located in the cardia or proximal to the esophagogastric junction (EGJ) are surgically demanding and associated with risks for stricture, leaks, or postoperative physiologic alterations. Many of those lesions can currently be resected through endoscopic submucosal dissection (ESD), a technique begun in Asia and subsequently carried out in the rest of the world. In Latin America, the experience with said procedure is still developing. The morbidity rate associated with SL resection using ESD ranges from 6.7 to 55.4% and includes bleeding, perforations, subcutaneous emphysema, pneumomediastinum, and fistulas secondary to failed closure of incised layers.2 Likewise, the manipulation of lesions greater than 40 mm can be complex and result in incomplete resections.3,4 The combination of flexible videoendoscopy (FVE) and transgastric intraluminal laparoscopy (TGIL) is a therapeutic alternative and a strategy of special interest for the treatment of gastric SLs.
We describe herein the technique employed and evaluated the results of the resection of the SLs in the cardia, in three patients operated on within the time frame of June 2012 and October 2017. Variables were expressed in absolute values. The arithmetic mean and range of the continuous variables were calculated, and the categorical variables were expressed as frequencies. The preoperative clinical evaluation, gastrointestinal videoendoscopy, triphasic abdominal CT, endoscopic ultrasound, and fine-needle aspiration cytology (FNAC) were included. The cytology results were consistent with leiomyomas in cases 2 and 3. Table 1 shows the characteristics of each patient. Other isolated factors that prompted the surgical indication were the diagnostic doubt in the FNAC, which could not be repeated due to cost (case 1), and the insistence of the patients on the resection of a “tumor” lesion in a “difficult” location, given the possibility of growth, which was a decision supported by the treating gastroenterologist.
Case series of patients with laparoscopic-endoscopic resection of subepithelial lesions in the cardia. Clinical characteristics, extension study findings, and postoperative results.
| Patient | Sex/age | BMI (kg/m2) | Endoscopic ultrasound | Surgical indication | Additional procedures | Diagram of lesion location | Surgery duration | Blood loss | Intraoperative adverse events | Follow-up (months) |
|---|---|---|---|---|---|---|---|---|---|---|
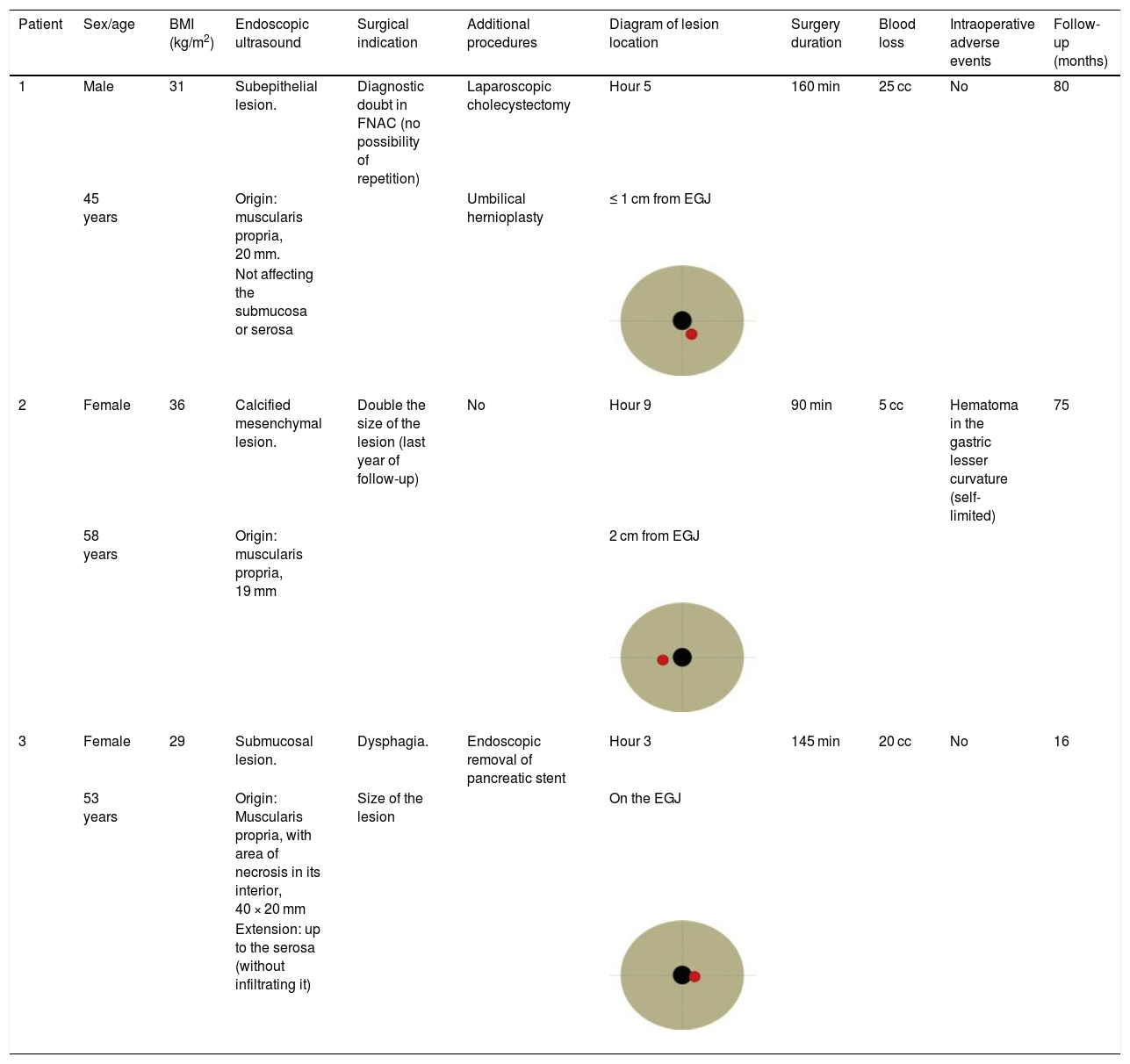
| 1 | Male | 31 | Subepithelial lesion. | Diagnostic doubt in FNAC (no possibility of repetition) | Laparoscopic cholecystectomy | Hour 5 | 160 min | 25 cc | No | 80 |
| 45 years | Origin: muscularis propria, 20 mm. | Umbilical hernioplasty | ≤ 1 cm from EGJ | |||||||
| Not affecting the submucosa or serosa | ||||||||||
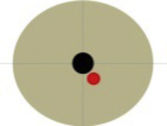
| 2 | Female | 36 | Calcified mesenchymal lesion. | Double the size of the lesion (last year of follow-up) | No | Hour 9 | 90 min | 5 cc | Hematoma in the gastric lesser curvature (self-limited) | 75 |
| 58 years | Origin: muscularis propria, 19 mm | 2 cm from EGJ | ||||||||
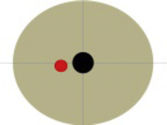
| 3 | Female | 29 | Submucosal lesion. | Dysphagia. | Endoscopic removal of pancreatic stent | Hour 3 | 145 min | 20 cc | No | 16 |
| 53 years | Origin: Muscularis propria, with area of necrosis in its interior, 40 × 20 mm | Size of the lesion | On the EGJ | |||||||
| Extension: up to the serosa (without infiltrating it) |
Under general anesthesia, we placed three abdominal ports and carried out exploratory laparoscopy. We endoscopically verified the location of the lesions, fixed the stomach to the abdominal wall with suture threads, and under endoscopic control, through transillumination, we introduced the transgastric trocars, avoiding the vessels of the stomach wall. Using diathermia in the dissection, we extracted the lesions through the mouth and sutured the mucosa with synthetic absorbable suture. Upon finishing, we performed a pneumatic test and endoscopically and laparoscopically verified the closure of the gastric ports. All the lesions had endophytic growth and the immunohistochemistry result was consistent with leiomyomas (positive Caldesmon antibodies and actin) (Fig. 1). Surgery duration was a mean 131.6 minutes, with blood loss of 16.6 cc. One case presented with an adverse event (Table 1). The postoperative esophagrams ruled out leaks, oral diet was then begun, and the patients were released from the hospital. There were no early or late postoperative complications and the mean follow-up period was 57 months.
Minimal access surgical approaches have been adapted to the location and characteristics of each organ and particular type of lesion. Different groups have employed FVE and TGIL, offering additional benefits to those of surgical or totally endoscopic resections,5 especially in large lesions, those located in the cardia, lesser curvature, or antrum, or those in the deep layers of the stomach wall.6 The technique is also a therapeutic alternative in centers, such as ours, with little experience in third space endoscopy. The combination of the two approaches aids in increasing the surgeon’s perspective, by having a simultaneous internal and external view of the organ. Triangulation of the laparoscopic instruments inside the gastric lumen enables the dissection and resection maneuvers to be controlled, providing safety during the suturing of the defects, with vessel sealing or closure with clips, sutures, or autosutures carried out in the same manner. Possible perforation or bleeding can also be treated through laparoscopy or endoscopy.
According to Ntourakis and Mavrogenis,5 the cooperative use of endoscopy and laparoscopy is grouped into three categories, depending on their roles: a) laparoscopic-assisted endoscopic resection, b) endoscopic-assisted laparoscopic resection, and c) combined laparoscopic-endoscopic resection. The procedure performed on our patients falls under the second category. Said technique was first described by Ohashi,7 in which the number of transgastric laparoscopic ports can vary, employing a single port, or combining FVE with the use of a robot, thus achieving additional benefits to those of robotic surgery. In our patients, the combination of FVE and TGIL was an efficacious and safe strategy for the resection of SLs in the cardia. The combined use of the two procedures potentiated the advantages of their individual use.
Ethical considerationsThe surgical procedure was explained to each of the patients. We obtained informed consent for both the surgical act and the publication of images (those documents are in the possession of the corresponding author), following the protocols of our work center on the publication of patient data, preserving their anonymity. We declare that no experiments on humans were conducted.
Financial disclosureNo financial support was received in relation to this article.
Conflict of interestThe authors declare that there is no conflict of interest.?
Please cite this article as: Pereira Graterol F, Salazar Marcano F, Venales Barrios Y, Caceres Cauro A. Resección laparoscópica-endoscópica transgástrica (intraluminal) de lesiones subepiteliales cardiales. Serie de casos. Revista de Gastroenterología de México. 2021;86:202–205.