Patients with gastroesophageal reflux disease (GERD) are treated with proton pump inhibitors (PPIs). Those that do not achieve symptom relief, or non-responders, usually undergo esophageal pH monitoring off PPIs in order to confirm the presence of GERD.
AimsTo assess the efficacy of the reverse-PPI trial in evaluating the presence of GERD or its recurrence rates, as well as to identify a correlation between the symptom recurrence rates and GERD severity determined by 48-hour Bravo esophageal pH-monitor testing.
MethodsA final total of 205 patients that underwent the 48-hour Bravo esophageal pH-monitoring study were retrospectively included. Patients discontinued PPI usage for at least 7 days prior to testing, and completed symptom questionnaires during the 2-day test. The Bravo test was considered positive if the percentage of time with esophageal pH <4 was >4.4%.
ResultsA total of 363 patients underwent 48-hour Bravo testing and of those patients, 205 were eligible for the study. Ninety-two patients reported symptoms as being «same/better» and 113 as being «worse» after stopping PPIs. Of the 92 patients with improved symptoms, 44 (48%) had documented acid reflux during the Bravo study, compared with 65 of 113 (58%) patients with worsening symptoms that also complained of acid reflux. Of the 109 patients found to have confirmed GERD upon pH monitoring, 65 (59.6%) reported a worsening of symptoms, compared with 48 of 96 (50.0%) patients without GERD (p=0.043). Main symptoms stated to be worse included heartburn, chest pain, regurgitation, nausea, and belching (p<0.05). Of the 205 patients, 103 were off PPIs for 7 days. Seventy-two of them (68.9%) reported a worsening of symptoms, compared with 40 of the 102 (41.2%) patients that were off PPIs for>7 days (p=0.042)
ConclusionSymptom exacerbation following PPI cessation for at least 7 days correlated with acid reflux severity assessed by Bravo testing. Patients off PPIs for 7 days had a higher likelihood of experiencing worsening symptoms, compared with those off PPIs for more than 7 days. These findings suggest that when PPIs are held for 7 days or less prior to Bravo testing, acute worsening of upper GI symptoms due to the abrupt discontinuation of therapy may influence the Bravo results. The etiology of this may be related to rebound acid hypersecretion and needs to be further elucidated in future studies.
Los pacientes con enfermedad por reflujo gastroesofágico (ERGE) son tratados con inhibidores de la bomba de protones (IBP). Aquellos que no logran el alivio de los síntomas, o los no respondedores, usualmente son sometidos a la monitorización de pH esofágico, habiendo suspendido los IBP, para así confirmar la presencia de la ERGE.
ObjetivosValorar la eficacia de la prueba con IBP invertida en la evaluación de la presencia de ERGE o sus tasas de recurrencia, así como la identificación de una correlación entre las tasas de recurrencia de los síntomas y la gravedad de la ERGE determinada por pruebas de monitorización de pH esofágico Bravo de 48 h de duración.
MétodosUn total final de 205 pacientes sometidos a las pruebas de monitorización de pH esofágico Bravo de 48 h de duración fueron incluidos retrospectivamente. Los pacientes descontinuaron el uso de los IBP por al menos 7 días previo a la prueba y completaron cuestionarios sintomáticos durante los 2 días de la prueba. La prueba Bravo se consideró positiva si el porcentaje de tiempo con pH esofágico < 4 era > 4.4%.
ResultadosUn total de 363 pacientes se sometieron a la prueba Bravo de 48 h, y de esos pacientes, 205 fueron elegibles para el estudio. Noventa y dos pacientes reportaron síntomas de estar «igual/mejor» y 113 de estar «peor» después de la suspensión de IBP. De los 92 pacientes con mejoría de síntomas, 44 (48%) tenían reflujo ácido durante la prueba Bravo documentado, comparado con 65 de 113 (58%) pacientes con empeoramiento de síntomas que también se refirieron tener reflujo ácido. De los 109 pacientes en los cuales se confirmó la ERGE con la monitorización de pH, 65 (59.6%) reportaron un empeoramiento sintomático, comparado con 48 de 96 (50.0%) pacientes sin ERGE (p=0.043). Los síntomas principales declarados como empeorados incluyeron acidez, dolor de pecho, regurgitación, náuseas y eructación (p<0.05). De los 205 pacientes, 103 no tomaron los IBP por 7 días. Setenta y dos de ellos (68.9%) reportaron un empeoramiento de síntomas, comparados con 40 de los 102 (41.2%) pacientes que no tomaron IBP por>7 días (p=0.042).
ConclusiónLa exacerbación sintomática seguida de la suspensión de los IBP de por al menos 7 días correlacionó con la severidad de reflujo ácido evaluado por la prueba Bravo. Los pacientes que no tomaron IBP por 7 días presentaban mayor probabilidad de experimentar empeoramiento de síntomas comparados con aquellos que no tomaron los IBP por más de 7 días. Estos hallazgos indican que cuando los IBP son suspendidos por 7 días o menos previo a la prueba Bravo, el empeoramiento agudo de los síntomas gastrointestinales superiores, debido a la descontinuación abrupta de la terapia, puede influir los resultados de la prueba Bravo. La etiología de esto puede estar relacionada con la hipersecreción ácida de rebote y requiere ser elucidada aún más en estudios a futuro.
Gastroesophageal reflux disease (GERD) is one of the most common gastrointestinal disorders worldwide. In the US and the Western world, approximately 40% of the overall adult population presents with some variety of reflux symptoms and approximately 20% of the population experiences symptoms at least once a week1,2. The American College of Gastroenterology endorses proton-pump inhibitor (PPI) therapy, based on a presumptive diagnosis, for patients that present with common GERD-related symptoms, such as heartburn, chest pain, and dysphagia3. The most common treatment plan for GERD involves acid suppression, mainly with PPIs. Symptom improvement with PPIs is used as evidence to confirm the diagnosis of GERD. However, it has been estimated that between 10 and 40% of GERD patients are considered partial or non-responders to a standard PPI dose4. Persistent reflux symptoms refractory to PPI treatment are a common presentation of GERD in hospital and clinical settings. To further evaluate these patients, a pH monitoring study is frequently employed, requiring PPI discontinuation for at least 7 days prior to testing.
Although this is currently the common method to diagnose or rule out GERD, studies have demonstrated false positive and negative ratios. Bjornson et al. showed that in a randomized group of PPI users, more than 27% of the patients taken off PPIs did not have symptom recurrence. In patients that discontinued short-term PPIs given for Helicobacter pylori eradication, symptoms also remained the same and did not worsen5,6. Similarly, false-negative results may also lead to under-treatment or incorrect diagnosis in symptomatic GERD patients7. Nevertheless, the empiric PPI trial is still an inexpensive and effective way to diagnose GERD.
Given that the PPI trial has been such a key diagnostic tool in GERD management, it may be useful to examine whether recurrence, worsening, or absence of symptoms after stopping therapy has any significance. For example, symptom recurrence after discontinuing therapy in a chronic PPI user can further substantiate the need to continue PPI therapy. Likewise, an improvement or no change in symptoms might suggest that a patient's symptoms are refractory to PPI therapy. Thus far, studies evaluating GERD recurrence or non-recurrence post PPI cessation have been scarce.
Our aim was to assess the efficacy of the reverse-PPI trial in determining the presence or absence of GERD in patients that discontinued PPI therapy to undergo pH testing, by studying the symptom recurrence rates. An additional aim was to identify a correlation between the symptom recurrence rates and GERD severity determined by the 48-hour Bravo esophageal pH-monitoring test.
MethodsWe designed a retrospective medical chart review to examine the overall recurrence and characterization of patient symptoms after discontinuing PPIs for at least one week in preparation for the 48-hour Bravo wireless esophageal pH monitoring study, within the time frame of January 2012 to July 2014. In essence, we carried out the exact opposite of a therapeutic PPI trial and called it a reverse-PPI trial, after which we studied the overall symptom changes and their influencing factors.
The study cohort included 363 GERD patients referred to our clinic at Temple University Hospital due to lack of symptom response to standard PPI therapy for at least 8 consecutive weeks. All patients underwent 48-hour Bravo studies after discontinuing PPI treatment for at least 7 days. Patients were excluded if they had any of the following: a history of requiring narcotics or other pain medications, previous diagnosis of Barrett's esophagus or peptic stricture, or significant physical or psychiatric comorbidities. All patients, the day of probe placement, completed a questionnaire on GERD-related symptomatology and PPI usage, as well as a Patient Assessment of GI Symptoms (PAGI-SYM) survey during the 2-day test. Using the PAGI-SYM survey, patients characterized their overall symptomatology as “better/same” or “worse” after stopping PPIs. Only data from the 205 patients that met all 3 eligibility criteria were included in our analysis.
The institutional review board at Temple University Hospital approved the present study and informed consent was obtained from all participating patients.
Questionnaire on the day of the testOn the day of the 48-hour Bravo wireless esophageal pH monitoring study, each patient completed a questionnaire covering the following aspects: demographic information, medical and surgical histories, main symptoms while on the previous PPI therapy, duration of GERD-related symptoms, and number of days since the last PPI dose. This information was used in addition to data gathered from the electronic medical records (EMRs). Informed consent was obtained from the patients regarding the potential use of these data in similar future studies.
48-hour Bravo wireless esophageal pH monitoring studyThe 48-hour Bravo wireless esophageal pH monitoring study is a test that is performed for the clinical evaluation of patients with esophageal symptoms. Patients received standard instructions prior to undergoing the study. Participating patients could not eat or drink anything after midnight the night before the study and were to suspend any prokinetic agents, narcotics, sedatives, and/or antispasmodics. All patients underwent an upper endoscopy under monitored anesthesia care. The Bravo capsule was attached to the esophageal mucosal wall 6cm proximal to the esophagogastric junction during the endoscopy.
Following the procedure, the patients were given a paper diary to register the timing of their meals, symptoms, and supine periods, as well as detailed instructions regarding the proper use of the data receiver. The 48-hour Bravo wireless esophageal pH monitoring study was subsequently performed and the results were generated by the pH monitoring software8,9.
Upon completion of the 48-hour study, the patient data were downloaded from the receiver to the analysis software (Given Imaging, Yoqneam, Israel). Recorded parameters included: total number of acid reflux episodes, number of episodes > 5min, longest reflux episode, and percentage of time with pH < 4.0. A cumulative data set was also calculated and reported for both days. Patients whose percentage of time with pH < 4.0 was greater than 4.4% were classified as having acid reflux8,9.
PAGI-SYMAfter the procedure, patients completed a PAGI-SYM questionnaire assessing their GERD-related symptoms. Patients were asked to rate the severity of their primary symptom and other GERD-related symptoms on a scale of 0 (none) to 5 (very severe). GERD-related symptoms included heartburn, chest pain, dysphagia, regurgitation, nausea, vomiting, coughing, wheezing, belching, and hoarseness. Next, they were asked to select which day the symptoms felt worse: Day 1, Day 2, or similar on both days. Finally, patients were asked to characterize their overall change in symptoms since discontinuing PPIs as “same/better” if their main symptom scores remained unchanged or decreased by one point or more. Likewise, they were asked to characterize their symptom change as “worse” if their symptom scores increased by one point or more.
Data analysisPatient data were put in a Microsoft Excel Database and identifying factors were coded to protect patient privacy. Microsoft Excel was used to record data from the Bravo capsule reports, the pre-study questionnaire, and the PAGI-SYM survey. The Student's t test was used to compare the data of the 2 groups of patients: the group that characterized overall symptoms as “same/better” versus the group that characterized overall symptoms as “worse”.
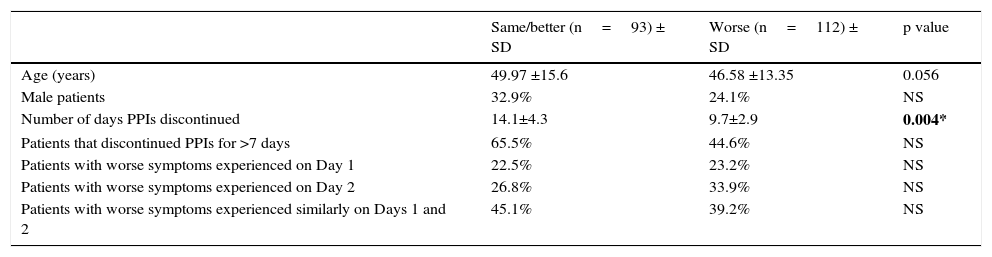
ResultsDemographics, proton pump inhibitor symptomatology, number of days of discontinued proton pump inhibitor use, and number of patients enrolled in the studyPatients were classified into 2 groups based on their characterization of overall symptom change after discontinuing PPIs for at least 7 days (Table 1). In general terms, patients that reported “same/better” symptoms had longer PPI discontinuation than those with “worse” symptoms (14.4 days ± 4.3 days vs 9.7 days ± 2.9 days, p ≤ 0.05). As a group, patients that presented with a worsening of symptoms had a lower mean age and the percentage of men was lower. The distribution of days during which the patients felt their symptoms were worse was similar in both groups. The highest percentage of patients felt their symptoms were similar on both days, followed by those that felt their symptoms were worse on Day 2. The lowest percentage of patients in both groups felt their symptoms were worse on Day 1 (Table 1).
Patient demographics.
| Same/better (n=93) ± SD | Worse (n=112) ± SD | p value | |
|---|---|---|---|
| Age (years) | 49.97 ±15.6 | 46.58 ±13.35 | 0.056 |
| Male patients | 32.9% | 24.1% | NS |
| Number of days PPIs discontinued | 14.1±4.3 | 9.7±2.9 | 0.004* |
| Patients that discontinued PPIs for >7 days | 65.5% | 44.6% | NS |
| Patients with worse symptoms experienced on Day 1 | 22.5% | 23.2% | NS |
| Patients with worse symptoms experienced on Day 2 | 26.8% | 33.9% | NS |
| Patients with worse symptoms experienced similarly on Days 1 and 2 | 45.1% | 39.2% | NS |
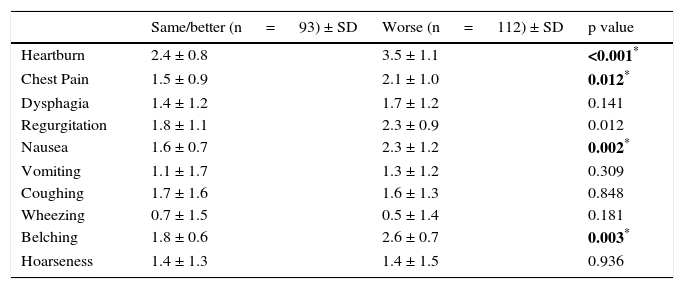
According to the PAGI-SYM survey, the patients whose scores for symptoms worsened, experienced higher levels of heartburn, chest pain, regurgitation, nausea, and vomiting (p ≤ 0.05) (Table 2). Symptom scores for dysphagia, vomiting, coughing, wheezing, and hoarseness were statistically insignificant and both groups reported similar severities (Table 2). Therefore, patients with a worsening of overall symptoms were more likely to experience an exacerbation of certain specific symptoms such as heartburn, chest pain, regurgitation, nausea, and vomiting.
PAGI-SYM scores from both groups of patients enrolled in the study.
| Same/better (n = 93) ± SD | Worse (n = 112) ± SD | p value | |
|---|---|---|---|
| Heartburn | 2.4 ± 0.8 | 3.5 ± 1.1 | <0.001* |
| Chest Pain | 1.5 ± 0.9 | 2.1 ± 1.0 | 0.012* |
| Dysphagia | 1.4 ± 1.2 | 1.7 ± 1.2 | 0.141 |
| Regurgitation | 1.8 ± 1.1 | 2.3 ± 0.9 | 0.012 |
| Nausea | 1.6 ± 0.7 | 2.3 ± 1.2 | 0.002* |
| Vomiting | 1.1 ± 1.7 | 1.3 ± 1.2 | 0.309 |
| Coughing | 1.7 ± 1.6 | 1.6 ± 1.3 | 0.848 |
| Wheezing | 0.7 ± 1.5 | 0.5 ± 1.4 | 0.181 |
| Belching | 1.8 ± 0.6 | 2.6 ± 0.7 | 0.003* |
| Hoarseness | 1.4 ± 1.3 | 1.4 ± 1.5 | 0.936 |
Table 3 shows the data collected from the 205 study patients. Of the 205 patients analyzed, 92 (44.9%) characterized their overall symptom change as “same/better” and 113 (55.1%) patients characterized their symptoms as “worse”. All patients discontinued the use of PPIs for at least 7 days prior to completing the PAGI-SYM survey. Of the 92 patients that had symptom improvement, 44 (47.8%) reported acid reflux. According to the Bravo pH monitoring studies, 109 (53.2%) patients had reflux (percentage of time with pH < 4.0 greater than 4.4%) and 96 (46.8%) patients did not have reflux (percentage of time with pH < 4.0 less than 4.4%). Forty-four (40.4%) of the 109 patients with reflux reported their symptoms as “same/better”, whereas the other 65 (59.6%) patients characterized their symptoms as “worse”. Likewise, 48 (50.0%) of the 96 patients with no reflux reported their symptoms as “same/better”, whereas the other 48 (50%) characterized their symptoms as “worse”.
48-hour Bravo pH monitoring studyThe Bravo data from Table 4 shows that patients that had a worsening of symptoms after stopping PPIs, had a higher acid exposure time (8.9% ± 1.2% vs 5.8% ± 1.6%) (p=0.081), more reflux episodes (93.3 ± 18.5 vs 72.9 ± 22.4; p=0.013), and longer reflux episodes (reflux > 5min) (8.4min ± 2.2min vs 6.3min ± 1.4min; p=0.045), compared with those that reported no worsening of symptoms. This suggests that patients with a higher frequency and longer duration of reflux episodes and higher acid exposure are more likely to experience a worsening of their symptoms once they discontinue PPIs.
48-h Bravo pH monitoring study results.
| Same/better (n=93) ± SD | Worse (n=112) ± SD | p value | |
|---|---|---|---|
| Day 1, total time pH < 4.0 | 6.3% ± 2.4% | 9.4% ± 3.3% | 0.094 |
| Day 2, total time pH < 4.0 | 6.7% ± 4.4% | 5.9% ± 2.3% | 0.491 |
| Total time pH < 4.0 | 5.8% ± 1.2% | 8.9% ± 1.6% | 0.082 |
| Total number of reflux episodes | 72.9 ± 22.4 | 93.3 ± 18.5 | 0.013* |
| Number of episodes > 5 min | 6.3min ± 4.4min | 8.4min ± 3.2min | 0.045 |
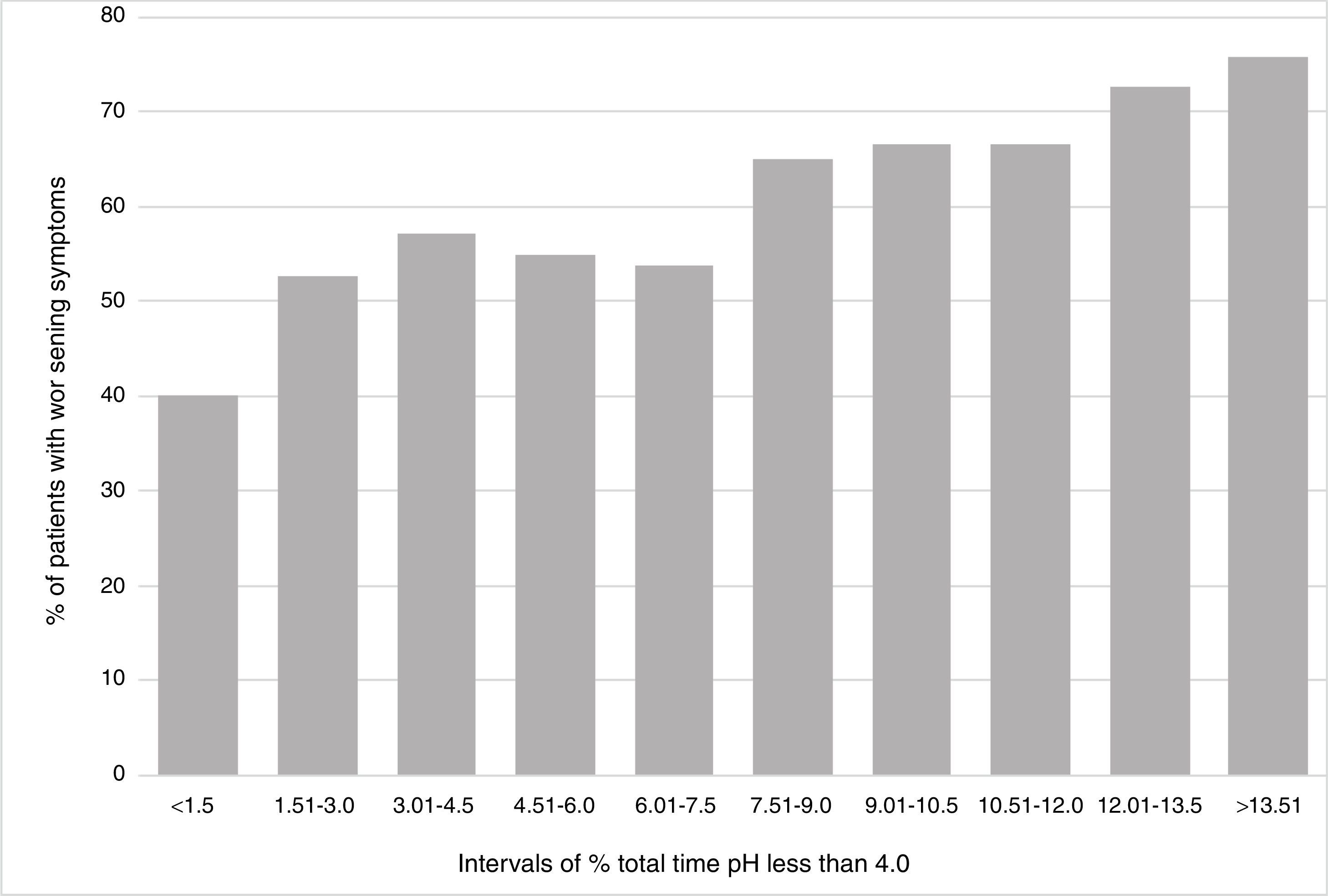
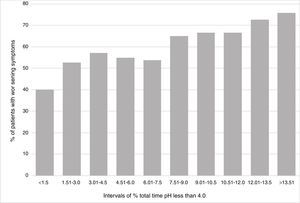
The data from the Bravo studies reporting the percentage of time with pH < 4 for each individual day, as well as the percentage of the overall time for the 2 days, were analyzed. These percentages of times with pH < 4 were divided into 10 separate, equally spaced percentage intervals from less than 1.5% to greater than 13.51%. There was a correlation between the percentage of the total time with pH < 4 and the percentage of patients with symptom worsening after suspending PPIs (R2=0.896). The patient data showed a positive association between GERD severity (measured by the percentage of time with pH < 4 that was > 4) and the overall rates of symptom worsening (fig. 1).
Impact of time of symptom onset on acid refluxTable 5 presents symptom duration and its impact on patient symptomatology. A total of 71 patients reported that the onset of their GERD-related symptoms occurred during the past year, whereas 134 patients reported an onset longer than one year. Of the 71 patients with onset in the past year, 35 (49.3%) had reflux and 36 (50.7%) did not have reflux. Thirty-six (50.7%) of the 71 patients reported their symptoms as “same/better”, whereas 35 (49.3%) patients characterized them as “worse”. Of the 134 patients that reported symptom onset of more than one year, 74 (55.2%) had reflux and 60 (44.8%) did not have reflux. Fifty-six (41.8%) of those patients reported symptoms as “same/better” and 78 (58.2%) characterized the symptom change as “worse”. In our data, patients with symptom duration longer than one year had higher rates of symptom worsening and acid reflux, compared with the patients whose symptom onset was less than one year.
Impact of symptom and PPI discontinuation duration on acid reflux.
| Symptom duration ≤ 1 year | ||
|---|---|---|
| Symptoms | Acid reflux present | Acid reflux absent |
| Same/better | 15 | 21 |
| Worse | 20 | 15 |
| Total | 35 | 36 |
| Symptom duration > 1 year | ||
|---|---|---|
| Symptoms | Acid reflux present | Acid reflux absent |
| Same/better | 14 | 42 |
| Worse | 60 | 18 |
| Total | 74 | 60 |
| PPI stopped for 7 days | ||
|---|---|---|
| Symptoms | Acid reflux present | Acid reflux absent |
| Same/better | 25 | 17 |
| Worse | 37 | 22 |
| Total | 62 | 39 |
| PPI stopped >7 days | ||
|---|---|---|
| Symptoms | Acid reflux present | Acid reflux absent |
| Same/better | 10 | 40 |
| Worse | 37 | 17 |
| Total | 47 | 57 |
A total of 101 patients reported suspending their respective PPIs for exactly 7 days prior to the Bravo procedure. Of those patients, 62 (61.4%) had reflux and 39 (38.6%) did not have reflux. In that group, 42 (41.6%) reported their symptoms as “same/better” and 59 (58.4%) characterized the symptom change as “worse”. In the group of 104 patients that discontinued their respective PPIs for more than 7 days prior to the Bravo procedure, 47 (45.2%) had reflux and 57 (54.8%) did not have reflux. Fifty (48.1%) of the 104 patients reported their symptoms as “same/better” and the remaining 54 (51.9%) described the symptom change as “worse”. As a result, our data indicated higher rates of symptom worsening and acid reflux in the patient group that stopped PPI usage for exactly 7 days and lower rates of symptom worsening and acid reflux in the patient group that discontinued PPI usage for more than 7 days (Table 5).
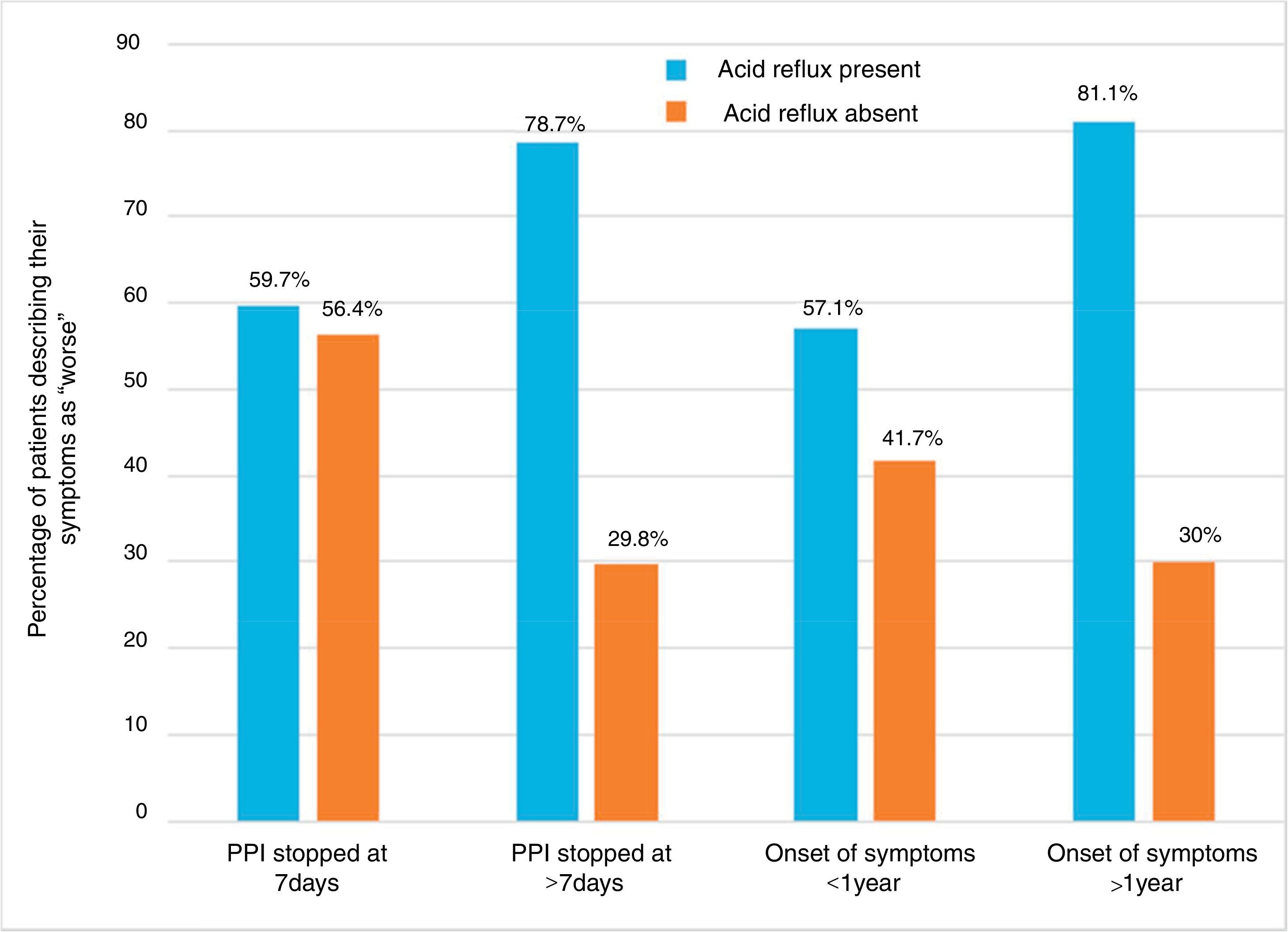
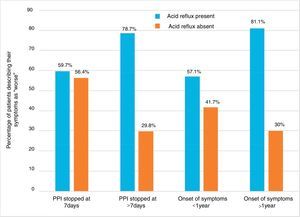
Impact of symptom onset and duration of proton pump inhibitor suspension on symptom worsening rates in patients with and without acid refluxPatients were divided into different groups based on PPI suspension and symptom onset. Within each group, patients were further divided into those that presented with reflux and those that did not. The rates of patients reporting a worsening of symptoms were studied in each of the 8 subgroups. The highest rates of symptom worsening were found in patients with reflux that discontinued PPIs for more than 7 days (78.7%), as well as in patients with reflux that had symptoms for more than one year (80.1%). The lowest rates were observed in patients without reflux that discontinued PPIs for more than 7 days (29.8%), as well as in those with reflux that had symptoms for more than one year (30.0%) (fig. 2). These data suggest that patients currently presenting with acid reflux that discontinue PPIs for more than 7 days and those with symptom onset prior to the past year will experience the highest rates of symptom worsening.
Symptom characterizationFrom their questionnaire answers, the patients that reported symptoms prior to stopping PPI therapy were grouped into 3 symptom categories: extra-esophageal, esophageal, and dyspeptic (Table 6). Patients whose primary symptoms included heartburn, nausea, vomiting, regurgitation, and belching were classified as having dyspeptic symptoms. Patients that experienced chest pain and dysphagia were considered to have esophageal symptoms of GERD. Finally, patients suffering from chronic cough, shortness of breath, and wheezing as primary symptoms were classified as having extra-esophageal symptoms. Overall, 21 patients had extra-esophageal symptoms, 150 patients had dyspeptic symptoms, and 34 had esophageal symptoms. Of the 21 patients with extra-esophageal symptoms, 9 were in the “worse” group and 12 were in the “same/better” group. Of the 150 patients with dyspeptic symptoms, 84 were in the “worse” group and 66 were in the “same/better” group. Finally, of the 34 patients in the esophageal group, 19 were in the “worse” group and 15 were in the “same/better” category.
Interestingly, all but 12 patients experienced a similar type of symptom upon discontinuation of PPI therapy, and none of those 12 patients had dyspeptic symptoms prior to stopping treatment. All the patients that experienced a change in their primary symptoms had dyspeptic symptoms after suspending the PPIs. Of the 12 patients, 3 were in the “same/better” group and 9 were in the “worse group”. Two of the 3 patients in the “same/better” group initially had esophageal symptoms, whereas the other patient presented with extra-esophageal symptoms prior to stopping therapy. Of the 9 patients in the “worse” group, 3 experienced esophageal symptoms and 6 experienced extra-esophageal symptoms prior to stopping treatment.
PPI dosage and duration of usageAll the patients in the study had been taking at least one of these drugs: dexlansoprazole, esomeprazole, omeprazole, and pantoprazole. Twenty-three patients had been on multiple therapy regimens and PPI therapy details for 8 other patients could not be found in the EMRs. Duration of PPI therapy was classified as long-term and short-term usage. There is currently no definitive length of time for determining long-term vs short-term PPI usage. However, numerous studies have indicated that PPI therapy for more than one year can be classified as long-term usage due to increased risk of side effects, such as hip fractures and malabsorption10. Therefore, patients that have been on therapy for over one year were classified as long-term users (Table 7).
PPI usage, dose, and usage duration.
| Dexlansoprazole | Same/better (n=13) ± SD | Worse (n=15) ± SD |
|---|---|---|
| Dose (mg/day) | 65.0 ± 5.0 | 72.9 ± 6.6 |
| % of long-term users (>1 year) | 58.3% | 86.7% |
| Esomeprazole | Same/better (n=21) ± SD | Worse (n=28) ± SD |
|---|---|---|
| Dose (mg/day) | 47.8 ± 4.3 | 51.4 ± 3.5 |
| Long-term users (>1 year) | 66.7% | 64.3% |
| Omeprazole | Same/better (n = 41) ± SD | Worse (n = 48) ± SD |
|---|---|---|
| Dose (mg/day) | 44.4 ± 4.2 | 37.0 ± 2.6 |
| % of long-term users (>1 year) | 60.1% | 63.3% |
| Pantoprazole | Same/better (n = 14) ± SD | Worse (n = 10) ± SD |
|---|---|---|
| Dose (mg/day) | 49.2 ± 4.9 | 56.0 ± 6.5 |
| Long-term users (>1 year) | 57.1% | 40.0% |
Data shows that mean PPI dosage was higher in the “worse” group for the following drugs: dexlansoprazole (65.0mg ± 5.0mg vs 72.9mg ± 6.6mg), esomeprazole, (47.8mg ± 4.3mg vs 51.4mg ± 3.5mg), and pantoprazole (49.2mg ± 4.9mg vs 56.0mg ± 6.5mg). PPI dosage was greater in the “same/better” group among those patients taking omeprazole (44.4mg ± 4.2mg vs 37.0mg ± 2.6mg), and dexlansoprazole (58.3 vs 86.7%) and omeprazole (60.1 vs 63.3%) had higher levels of long-term users in the “worse” group than in the “same/better” group. On the other hand, esomeprazole (66.7 vs 64.3%) had higher levels of long-term users in the “same/better” group than in the “worse” group (Table 7).
DiscussionOur study demonstrated that a worsening of symptoms after stopping PPIs for at least 7 days was positively associated with acid reflux severity during Bravo pH monitoring, and was therefore suggestive of GERD. To have an accurate measure of patient acid reflux, we collected our data from patients that adhered to the current protocol of PPI discontinuation for at least 7 days prior to the study. This indicates that patients with severe levels of GERD will have a greater likelihood of symptom worsening upon discontinuation of PPIs, and those with lower levels of GERD will have a lesser likelihood of symptom worsening. Our survey data showed that these patients also complained of a worsening of specific symptoms, such as chest pain, nausea, heartburn, and belching. Therefore, we assume that the worsening of these specific symptoms may lead to patients feeling an overall worsening of their acid reflux. Other symptoms, such as wheezing, hoarseness, coughing, and dysphagia were not as statistically significant in predicting a worsening of symptoms.
In addition, our reverse-PPI trial was a better diagnostic predictor of GERD in patients that stopped PPIs for more than 7 days, in comparison with those that stopped for just 7 days. In the latter, rates of symptom worsening were similar in the patients with acid reflux and without acid reflux. The trial did not provide much benefit in predicting GERD in that group. However, 78.7% of the patients with reflux that stopped PPIs for more than 7 days experienced a worsening of symptoms and only 29.8% of the patients without acid reflux in that same group experienced a worsening of symptoms. This was markedly different from the 7-day group.
One of the hypotheses for this difference is a higher level of rebound acid hypersecretion (RAH). Data from Niklasson et al. show RAH was strongest in 1-week post-treatment cessation11. Therefore, higher levels of RAH may have occurred in the patients that discontinued PPIs for 7 days. However, a recent meta-analysis concluded that studies on patients with reflux disease found no evidence of symptomatic RAH12.
The data analysis also showed that the reverse-PPI trial was more effective in predicting GERD in patients with chronic GERD-like symptoms with onset of more than one year. Nearly 81% of the patients with GERD experienced a worsening of symptoms, whereas only 30% of the non-GERD patients experienced a worsening of symptoms. Patients with no acid reflux that discontinued PPIs for more than 7 days, but whose symptom onset began prior to the past year, experienced the lowest rates of overall symptomatology worsening. This suggests that in patients suffering with long-term GERD-related symptoms, the reverse-PPI trial could be another effective and practical diagnostic tool. However, the trial is less effective as a diagnostic tool in patients suffering with GERD for less than one year. Only 57% of patients with GERD for this time period felt their symptoms had worsened, whereas more than 41% without GERD experienced a worsening of their symptoms. Consequently, the reverse-PPI trial was not useful for predicting GERD in patients experiencing symptoms for a shorter period of time (< 1 year).
The reverse-PPI trial is a practical and useful treatment plan, given the widespread use of PPIs in GERD. However, exactly how sensitive and specific the trials are in correctly diagnosing and ruling out GERD remains unclear. A meta-analysis done by Mattis et al. reviewed 11 different therapeutic PPI trials and tested each trial's sensitivity and specificity in diagnosing GERD13. Their data showed a broad range of values from each of the trials. Sensitivity values ranged from 0.48-1.0 and specificity values ranged from 0.30-0.71. The sensitivity and specificity averages calculated for the collective group of studies were 0.71 and 0.41, respectively. The authors concluded that the PPI trial does not confidently establish or exclude the diagnosis of GERD and currently, the therapeutic treatment approach should be carried out on an individualized basis14. We hope that the reverse-PPI trial can serve as an additional test used in conjunction with the therapeutic PPI trial and that it may aid physicians in better managing their patients. The reverse-PPI trial, much like the therapeutic PPI trial, can be utilized in a very cost-efficient manner.
In addition to improving the clinical diagnosis of GERD, we believe that the reverse-PPI trial can reduce unnecessary PPI treatment. In the past few years, there has been a rampant overuse of PPIs. Chia et al. reported on a cohort from Singapore in which PPI use was prevalent in hospitals, but fewer than half of the hospitalized patients using PPIs had evidence-based indications that supported such use15. In addition, chronic long-term PPI overuse and misuse can have devastating side effects, including recurrent bacterial infections, increased bone fractures, hypomagnesemia, and hypochlorhydria16. Some studies also indicate that the recent rise in Clostridium difficile infections may be due in part to the overuse of PPIs in the last few years17,18. The cost and economic burden can also be very high on local clinics and patients12.
The present study is not without limitations. Our data relied on patient-reported symptom characterization and we felt that using a numeric scale to rate the change in symptomatology would allow us to gather a more detailed characterization of the change in symptoms upon PPI suspension, as opposed to simply asking the patients to characterize their symptoms as “same/better” or “worse”. In addition, because this study is based on patient surveys, there is a potential for error due to bias and misrepresentation of symptoms. However, conducting similar surveys with a greater number of patients and in different communities may provide further insight.
ConclusionsOur study demonstrated that a worsening of symptoms after discontinuing PPIs for at least 7 days was positively associated with acid reflux severity during Bravo pH monitoring, suggesting GERD. Results of the reverse-PPI trial (assessing symptom change) can potentially provide clues leading to the evidence that GERD reflux is the cause of symptoms in patients suspending PPIs for more than 7 days prior to the pH study and in patients that present with symptoms for more than one year. Consequently, a reverse-PPI trial can be a useful diagnostic tool in patients presenting with severe degrees of GERD, as higher rates of these patients will experience a worsening of their symptoms upon cessation of therapy. Nevertheless, further large-scale studies are required to substantiate our data.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that the procedures followed were in accordance with the regulations of the relevant clinical research ethics committee and with those of the Code of Ethics of the World Medical Association (Declaration of Helsinki).
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors have obtained the written informed consent of the patients or subjects mentioned in the article. The corresponding author is in possession of this document.
Financial disclosureNone.
Conflict of interestThe authors declare that there is no conflict of interest.
Please cite this article as: Schey R, Alla SP, Midani D, Parkman HP. Recurrencia sintomática relacionada con la enfermedad por reflujo gastroesofágico en pacientes que descontinúan los inhibidores de la bomba de protones durante el estudio inalámbrico de monitorización de pH esofágico Bravo. Revista de Gastroenterología de México. 2017;82:277–286.
See related content at DOI: http://dx.doi.org/10.1016/j.rgmxen.2017.07.019, Coss-Adame E. Symptomatic recurrence upon proton pump inhibitor discontinuation correlated with results of Bravo® wireless esophageal pH monitoring in patients with gastroesophageal reflux disease. Revista de Gastroenterología de México. 2017;82:275–276.