Ductal adenocarcinoma of the pancreas is the most common biliopancreatic tumor. It accounts for 90% of those lesions and has a survival rate of 8.3%. According to the National Cancer Institute, the pathology makes up approximately 3% of all cancers in the United States and 7% of cancer deaths.1 Cholangiocarcinomas constitute 3% of gastrointestinal tumors, and tumors of the ampulla of Vater, called ampullomas, make up 2% of all digestive tract tumors and can arise from any of the 3 types of epithelium that form the papilla.2
Pancreatoduodenectomy was described by Allessandro Codivilla in 1898. In 1909, Kausch successfully performed the first regional resection of a periampullary tumor and the procedure was later popularized by Allen O. Whipple (1935). It is considered one of the most complex operations of the digestive tract due to the combined challenge of careful dissection in close proximity to critical vascular structures and the restoration of the enteric continuity through the construction of 3 anastomoses (pancreatojejunostomy, hepaticojejunostomy, and gastrojejunostomy).3
The da Vinci surgical system is composed of a robot with 3 or 4 arms operated by a surgeon from a console. Robotic-assisted surgery overcomes many of the main deficiencies of traditional laparoscopy, which include monocular vision, limited degrees of freedom, and the effects of pivot and fulcrum. In contrast, the robotic platform provides the surgeon with three-dimensional stereoscopic vision of the operating field. It restores hand-eye coordination, eliminating hand tremor, and the instrumentation favors complex angles of attack. Thus, the surgeon’s skills are enhanced, especially those involved in highly complex suturing.4,5
The first robotic-assisted pancreatoduodenectomy was reported on in 2003 by Giulianotti et al. They performed the first 6 pancreatoduodenectomies utilizing a hybrid technique: laparoscopic resection and robotic-assisted reconstruction. The resection and reconstruction in the last 2 procedures were completely carried out through robotic assistance. Mean surgery duration was 490 min and the morbidity and mortality rates were 37.5% and 12.5%, respectively.6
The development of minimal invasion in pancreatoduodenectomy in Mexico has advanced relatively slowly. At our hospital, the first laparoscopic pancreatoduodenectomy was performed by the physicians Carlos Morales, José Luis Beristaín, and Adolfo Cuendis, on March 7, 2013. Enthusiasm for the approach was then demonstrated by numerous surgical groups and in October 2015 the da Vinci Robotic System was introduced at our hospital. Complex hepatopancreatobiliary surgery immediately benefited from that approach, with the first hybrid pancreatoduodenectomy performed on April 28, 2016, with laparoscopic resection and robotic-assisted reconstruction.
We present herein the first case of fully robotic pancreatoduodenectomy performed at the Hospital General “Dr. Manuel Gea González”.
A 66-year-old man had a history of 8-year progression of controlled diabetes mellitus. His current illness began one year prior to diagnosis, with acholia, choluria, jaundice of the skin and teguments, 20 kg weight loss, and pain in the right hypochondrium. He presented with symptoms of cholangitis, prior to his referral to our service, that required management with endoscopic retrograde cholangiopancreatography (ERCP) and endostent as periampullary tumor palliation. The initial post-ERCP laboratory tests showed obstructive jaundice with total bilirubin of 22 mg/dl, mainly dependent on direct bilirubin levels, no leukocytosis, and a CA 19-9 level of 116 U.
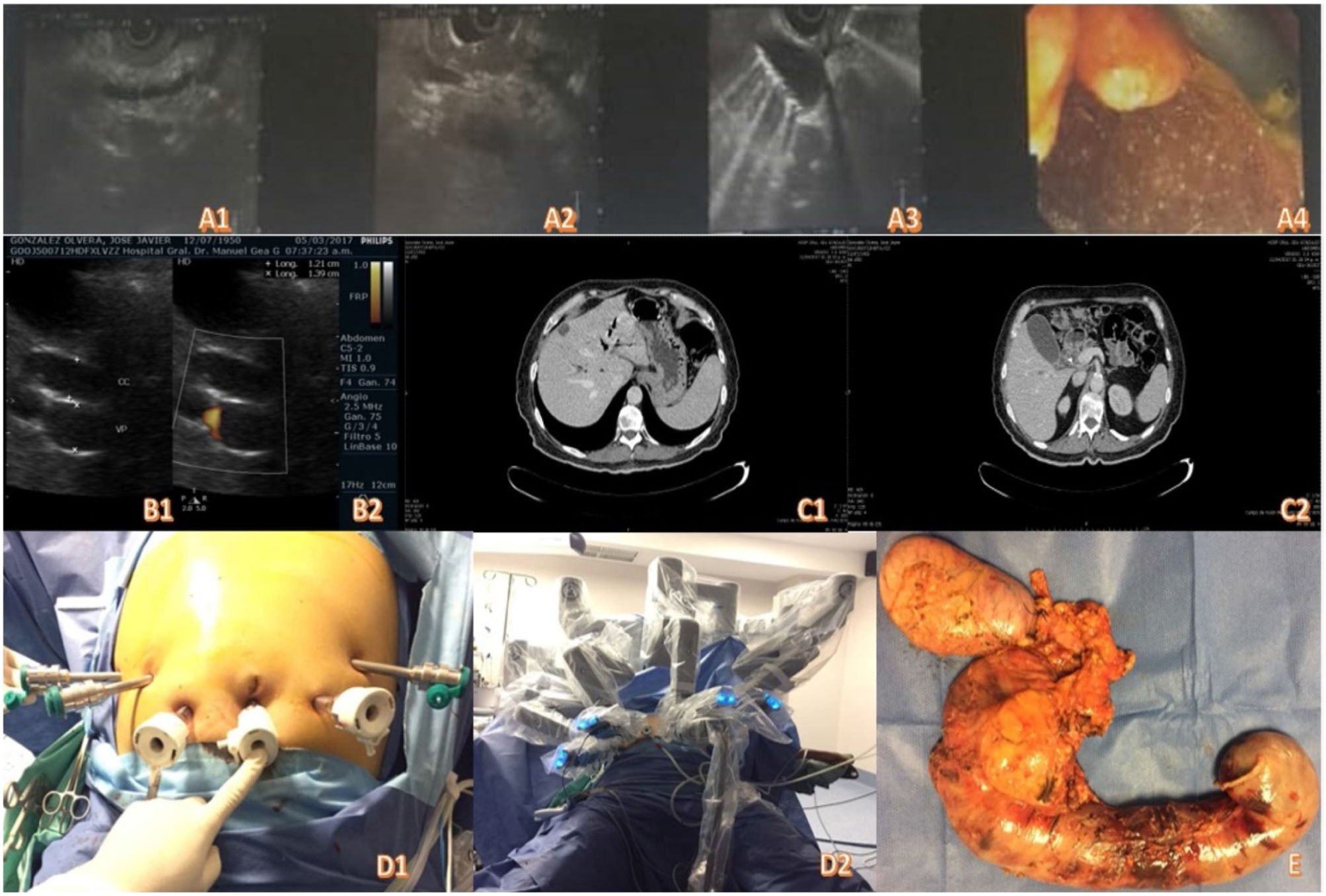
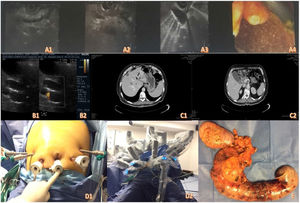
As part of the preoperative protocol, endoscopic ultrasound was carried out, diagnosing a tumor of the ampulla of Vater. The uT2 lesion biopsy was positive for adenocarcinoma of the ampulla (Fig. 1A1-A4). Ultrasound of the liver and biliary tract was ordered, identifying an enlarged gallbladder (Fig. 1B1-B2). A contrast-enhanced abdominal computed tomography scan revealed a periampullary tumor with associated bile duct and pancreatic duct dilation up to 8 mm, with no signs of vascular invasion or distant disease (Fig. 1C1-C2).
After being approved by the multidisciplinary committee of the hospital, a fully robotic pylorus-preserving pancreatoduodenectomy was performed, with an operative time of 8 h, utilizing 4 robotic arms, with the patient in the French position (Fig. 1D1-D2). Surgery duration was longer than that reported with a standardized curve in the literature, precisely because it was the first procedure of that type performed by our group. However, our operative time with that approach was similar to the ones reported by Giulianotti in 2003. As reported in large case series, surgery duration will progressively decrease as more such procedures are performed. Resection took up most of the time of the procedure, with a duration of 4 h 45 min, and 3 h 15 min for reconstruction. Operative time for open surgery at teaching and training centers, such as ours, is around 5 h. However, minimally invasive approaches provide a shorter recovery period, less pain, fewer hernias, and lower infection rates. A 2-layer duct-to-mucosa pancreatojejunostomy with no stenting, a single-layer side-to-side hepaticojejunostomy, and a 2-layer end-to-side duodenojejunostomy were performed, achieving R0 with intraoperative blood loss of 400 cc.
Postoperative recovery was favorable, with no development of complications, and the patient was discharged from the hospital on the 9th day after surgery. We are aware that the time of release in the literature is on average 3 days shorter, but because it was our first case, initiation and tolerance of oral intake took longer than usual.
The pathology report indicated moderately differentiated adenocarcinoma (grade II) of the ampulla of Vater, invading the pancreas and duodenal wall, with metastasis to the peripancreatic lymph nodes. The tumor measured 5 × 3.5 cm, the surgical margins were disease-free, and it was staged as T3 N1 M0, with a lymph node harvest of 25 nodes. The patient was referred for multimodal treatment in good condition (Fig. 1E). Upon his release, the patient was referred for follow-up and multimodal treatment, receiving adjuvant therapy. However, he presented with disease recurrence and progression at month 14 of follow-up.
To the best of our knowledge, the present case of fully robotic pancreatoduodenectomy performed by the multidisciplinary group of our hospital center is the first to be reported in Mexico. We are aware that the management of hepatobiliary malignant disease requires highly specialized personnel at high-volume centers. In that respect, we are a team that is made up of surgeons trained in minimally invasive hepatopancreatobiliary surgery, oncology surgeons trained in minimally invasive surgery, and oncology surgeons with formal training in hepatopancreatobiliary surgery. As a whole, our group performs more than 40 surgeries per year.
Fig. 1A1-A4) Endoscopic ultrasound images. B1-B2) Abdominal ultrasound. C1-C2) Contrast-enhanced abdominal tomography. D1-D2) Positioning of the trocars and docking of the robot. E) Surgical specimen.
Ethical disclosuresBefore performing the surgical procedure, a written statement of informed consent was requested of the patient and his responsible relative, observed by 2 witnesses.
Before the surgical procedure, the case was presented to the multidisciplinary committee of the Hospital General Dr. Manuel Gea González, which evaluated its risks and benefits. However, because it was a case report and not a research study, as such, authorization by an Ethics Committee was not requested.
The authors declare that the present article contains no personal information of any type that could identify the patient.
Financial disclosureNo financial support was received in relation to this article.
Conflict of interestThe authors declare that there is no conflict of interest.
The authors wish to thank the Hospital General Dr. Manuel Gea González for the opportunity to work with this technology and all the members of the team that worked together, seeking the wellbeing of the patient.
Please cite this article as: Cuendis-Velázquez A, López y López MA, Bada-Yllan O, Zozaya-García JM, Melchor-Ruan J, Vela-Sarmiento I, et al. Pancreatoduodenectomía totalmente robótica. Primer procedimiento en México. Revista de Gastroenterología de México. 2020;85:100–102.