To identify and discuss the efficacy of dietary interventions, antioxidant supplementation, physical activity, and nutritional and psychologic counseling in the treatment of children and adolescents with non alcoholic fatty liver disease associated with obesity.
Materials and methodsA scoping review of studies on nutritional and educational interventions and physical activity in pediatric patients with non alcoholic fatty liver disease was conducted. A search for randomized clinical trials or quasi-experimental studies published up to December 2017 was carried out, utilizing seven databases (Medline, EBSCO, OVID, Science Direct, JSTOR, Wiley, and Biblioteca Digital UDG).
ResultsFrom a total of 751 articles, 729 were excluded due to the criteria of age, design, language, diagnostic method, and outcome variables. The analysis included 22 articles. The most frequently used intervention variables were diet and physical activity. The interventions had different durations, but most were carried out for one year. Some authors employed ascorbic acid, vitamin E, or omega-3 fatty acid supplementation. There were varying degrees of improvement in the variables analyzed in the majority of the studies, such as a decrease in ALT levels, a reduced frequency of steatosis determined through imaging studies, and a decrease in body mass index.
ConclusionsThe dietary interventions, omega-3 fatty acid supplementation, physical activity, and nutritional and psychologic counseling were identified as efficacious measures in the treatment of non alcoholic fatty liver disease associated with obesity in children and adolescents, according to biochemical or imaging study indicators, within the time frame of the intervention.
Identificar y discutir la eficacia de intervenciones nutricias, suplementación de antioxidantes, actividad física y consultoría nutricia y psicológica en el tratamiento de niños y adolescentes con hígado graso no alcohólico asociado a obesidad.
Material y métodosSe realizó una revisión sistemática de estudios de intervención nutricia, educativa y con actividad física en pacientes pediátricos con hígado graso no alcohólico. Se revisaron siete bases de datos (Medline, EBSCO, OVID, Science Direct, JSTOR, E, Wiley y Biblioteca Digital UDG) para la búsqueda de ensayos clínicos aleatorizados o cuasiexperimentales publicados hasta diciembre de 2017.
ResultadosDe un total de 751 artículos se excluyeron 729 por criterios como edad, diseño, idioma, método de diagnóstico y variables de desenlace; el análisis se realizó con 22 trabajos. Las variables de intervención empleadas con mayor frecuencia fueron la dieta y la actividad física por tiempo variable, en la mayor parte de los trabajos durante un año. Algunos autores suplementaron con ácido ascórbico, vitamina E o ácidos grasos omega-3. En la mayor parte de los trabajos evaluados se consiguieron grados variables de mejoría de las variables evaluadas como disminución de niveles de ALT, disminución de la frecuencia de esteatosis por estudios de imagen y disminución del índice de masa corporal.
ConclusionesLas intervenciones dietéticas, la suplementación de ácidos grasos omega-3, la actividad física y la consejería nutricia y psicológica se identificaron como medidas eficaces en el tratamiento del hígado graso no alcohólico asociado a obesidad en niños y adolescentes de acuerdo a indicadores bioquímicos o de imagen durante el período de la intervención.
Nonalcoholic fatty liver disease (NAFLD) is characterized by an accumulation of fat in the hepatocytes that is not associated with alcohol ingestion or metabolic diseases. For more than a decade, it has been considered the most frequent cause of chronic liver disease in children worldwide.1,2 Its clinical and histopathologic spectrum can range from simple steatosis without inflammation, to steatohepatitis, cirrhosis, and hepatocellular carcinoma.2,3
In Mexico and different Latin American countries, and in the United States, the prevalence of NAFLD has a linear relation to the prevalence of obesity, in the sense that when there is a higher prevalence of obesity, there is a higher prevalence of NAFLD.4 That epidemiologic behavior establishes a close association between the two problems and places obesity as a central pathophysiologic factor in the occurrence of NAFLD in pediatric-aged patients. The disease also shares covariables in the context of an unhealthy lifestyle, such as being sedentary, poor nutrition, and insufficient physical activity. Both obesity and NAFLD in childhood and adolescence clinically and biochemically express insulin resistance, dyslipidemia, hyperglycemia, and high blood pressure.1,2 However, even though obesity is the most important risk factor for NAFLD, not all obese individuals present with it, suggesting the participation of other specific associated factors, such as the excessive consumption of fructose, soft drinks, saturated fat, and cholesterol and the insufficient consumption of fiber, fish, omega-3 fatty acids, and vitamin E. Recent advances have been made in relation to the consumption of fructose, antioxidants, and omega-3 fatty acids.5,6 A group of factors that may be defining in the occurrence of NAFLD are genetic. Loci associated with Mendelian expressions of obesity, such as loci related to the central regulation of appetite, have been demonstrated, signifying a complex polygenic interaction.7
The prevalence of NAFLD varies from 2.8 to 25% in the general population, albeit that variability could depend on the diagnostic criteria employed. In the United States, the European Union, Australia, and Asia, NAFLD affects 10 to 24% of the population.8 In the Third National Health and Nutrition Examination Survey (NHANES III) in which 2,450 children and adolescents between 12 and 18 years of age were evaluated, alanine aminotransferase (ALT) levels were elevated in 75 adolescents, corresponding to 3% of that age group.9 In a study on 810 obese Japanese children from 4 to 12 years of age, employing ultrasound imaging, there was a 2.6% prevalence of NAFLD.10 Based on those studies, the prevalence of NAFLD in the pediatric population of the United States and Asia can be estimated at around 3%. In different studies in Mexico, the prevalence of NAFLD in children and adolescents with obesity has been estimated to range from 12 to 42%.11–14 Nevertheless, population studies have not been conducted in Mexico and therefore the real prevalence of NAFLD is not known.
There is also not enough information related to the treatment of NAFLD in the Mexican pediatric population. Lifestyle modifications, particularly those related to dietary nutrition and exercise, are considered the fundamental pillars in the therapeutic intervention directed at reducing adiposity -the variable that defines obesity- and they continue to be regarded as the most efficacious treatment for NAFLD.
In that context, the aim of the present scoping review was to identify and discuss the efficacy of nutritional interventions, antioxidant supplementation, and nutritional and psychologic counseling in the treatment of children and adolescents that present with obesity-associated NAFLD.
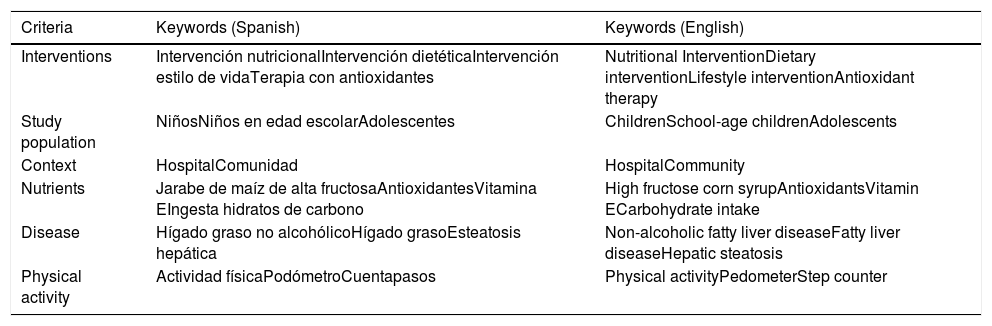
MethodologyThe present scoping review included articles published up to December 2017.15–17 The databases searched were the Biblioteca Digital de la Universidad de Guadalajara, Medline, EBSCO (Elton B. Stephens Co.), OVID, Science Direct, JSTOR (Journal Storage), and Wiley. Randomized controlled clinical trials and uncontrolled intervention studies on school-age children (6 to 11 years) and adolescents (12 to 18 years) diagnosed with obesity and NAFLD were selected. The operational definition of obesity from the World Health Organization (z-BMI > 2 SD), the Centers for Disease Control and Prevention (BMI > 95 percentile), and the International Obesity Task Force (BMI > 30) were those utilized in the studies chosen.18–20 The criteria for diagnosing NAFLD were alanine aminotransferase or aspartate aminotransferase values two times higher than the upper limit of normal for those enzymes, according to sex and/or the identification of hepatic steatosis through ultrasound or magnetic resonance imaging.1 The inclusion criteria were: nutritional interventions with diets modified in relation to energy intake and/or nutrients, interventions with physical activity or exercise, and interventions with antioxidant supplementation (vitamin C, vitamin E, and omega-3, 6, and 9 essential polyunsaturated fatty acids). Likewise, the present review included studies on individual, group, or family nutritional and psychologic counseling, as well as one mixed-design article that encompassed both a qualitative and quantitative intervention. Once the aim of the review was established, a list of keywords and their later combination using Boolean operators, were carried out. Table 1 shows the keywords employed for the initial search and Table 2 shows the keyword combinations that produced the highest number of results. The present scoping review was made up of 22 studies,21–43 and the number of useful references selected from the databases were: 4 from the Biblioteca Digital de la Universidad de Guadalajara, 2 from EBSCO, 7 from Medline, 6 from Ovid, 2 from Wiley, and 1 from JUSTOR. Only the studies that reported BMI and transferase values in the results and that utilized ultrasound and other imaging studies in the methodology were chosen. Twenty-seven studies were excluded. The main reasons for exclusion were: populations above 18 years of age, works that were review articles or meta-analyses, and articles not written in English or Spanish. The data analysis was performed through a systematization matrix of the previously selected articles.
Keywords in Spanish and English employed in the bibliographic search in the databases consulted.
| Criteria | Keywords (Spanish) | Keywords (English) |
|---|---|---|
| Interventions | Intervención nutricionalIntervención dietéticaIntervención estilo de vidaTerapia con antioxidantes | Nutritional InterventionDietary interventionLifestyle interventionAntioxidant therapy |
| Study population | NiñosNiños en edad escolarAdolescentes | ChildrenSchool-age childrenAdolescents |
| Context | HospitalComunidad | HospitalCommunity |
| Nutrients | Jarabe de maíz de alta fructosaAntioxidantesVitamina EIngesta hidratos de carbono | High fructose corn syrupAntioxidantsVitamin ECarbohydrate intake |
| Disease | Hígado graso no alcohólicoHígado grasoEsteatosis hepática | Non-alcoholic fatty liver diseaseFatty liver diseaseHepatic steatosis |
| Physical activity | Actividad físicaPodómetroCuentapasos | Physical activityPedometerStep counter |
The combination of keywords that produced the most results.
| Age group | Fatty liver disease and children and interventionFatty liver disease or steatosis and childrenNon-alcoholic fatty liver disease or steatosis and children |
| Non-alcoholic fatty liver disease and adolescents and intervention | |
| Fatty liver disease and pediatric and intervention | |
| Diet | Non-alcoholic fatty liver disease and children and diet therapy intervention |
| Fatty liver disease and children and diet | |
| Non-alcoholic fatty liver disease and children and diet therapy | |
| Non-alcoholic fatty liver disease and children and high fructose corn syrup | |
| Exercise | Non-alcoholic fatty liver disease and children and exercise |
| Fatty liver disease and children and physical activityFatty liver disease and children and physical activity or exercise | |
| Antioxidants | Fatty liver disease and children and antioxidant supplementation |
| Fatty liver disease and children and antioxidant therapy | |
| Fatty liver disease and children and vitamin E | |
| Fatty liver disease and children and vitamin C | |
| Non-alcoholic fatty liver disease and children and antioxidant therapy | |
| Lifestyle | Non-alcoholic fatty liver disease and children and lifestyle intervention |
| Non-alcoholic fatty liver disease and children and lifestyle intervention | |
| Non-alcoholic fatty liver disease and children and lifestyle changes | |
| Non-alcoholic fatty liver disease and children and lifestyle modification |
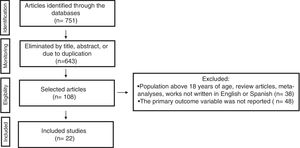
A total of 751 articles were identified and 643 were excluded due to their titles, abstracts, or to duplication. Additional database filters were used on the 108 remaining articles, in relation to age group and type of study (controlled and randomized clinical trial, quasi-experimental study). Thirty-eight of those articles were eliminated because they had populations above 18 years of age, they were review articles or meta-analyses, or they were in languages other than English or Spanish, whereas 47 were eliminated because they did not adequately report the primary result of interest or outcome variable. Finally, 22 references were included that identified BMI values, serum transferase concentrations, and ultrasound and other imaging study findings in the methodology and results sections, (fig. 1). Twenty-two clinical trials classified as empirical-analytic studies were also included, one of which had a mixed qualitative-quantitative design, (Table 3). There were results from 13 countries and the majority of the studies selected were from the United States (40%) and Italy (31%). Only 2 clinical trials were conducted in Latin America. A total of 65% of the clinical trials were considered quasi-experimental or open trials because they did not have control groups for comparison purposes. The rest of the works were considered longitudinal intervention studies or randomized controlled clinical trials with a control group.
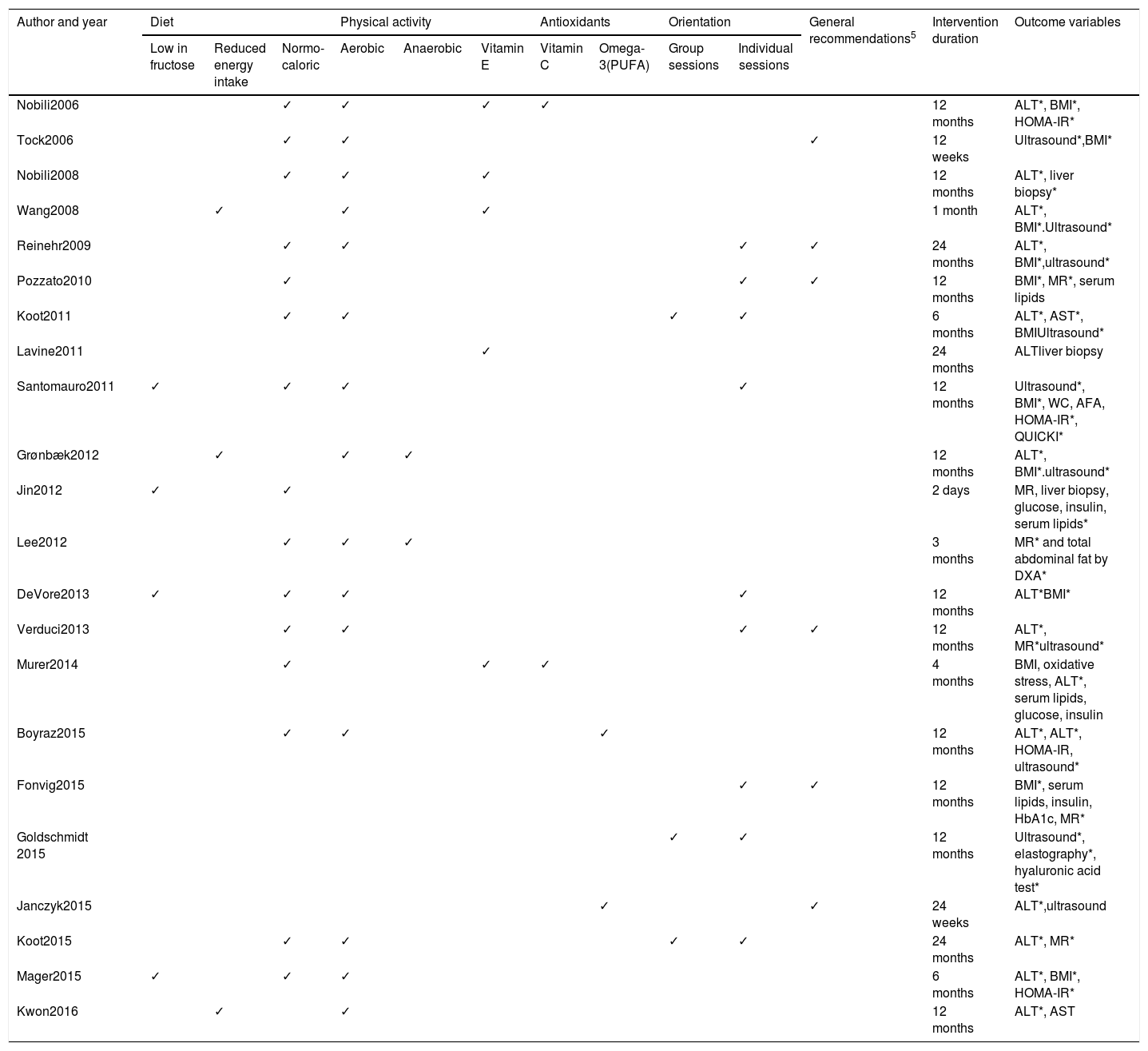
Intervention and outcome variables of the 22 intervention studies on pediatric patients with NAFLD included in the scoping review. The outcome variables with improvement after the intervention reported by the study authors are identified with an asterisk (*) in the last column.
| Author and year | Diet | Physical activity | Antioxidants | Orientation | General recommendations5 | Intervention duration | Outcome variables | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Low in fructose | Reduced energy intake | Normo-caloric | Aerobic | Anaerobic | Vitamin E | Vitamin C | Omega-3(PUFA) | Group sessions | Individual sessions | ||||
| Nobili2006 | ✓ | ✓ | ✓ | ✓ | 12 months | ALT*, BMI*, HOMA-IR* | |||||||
| Tock2006 | ✓ | ✓ | ✓ | 12 weeks | Ultrasound*,BMI* | ||||||||
| Nobili2008 | ✓ | ✓ | ✓ | 12 months | ALT*, liver biopsy* | ||||||||
| Wang2008 | ✓ | ✓ | ✓ | 1 month | ALT*, BMI*.Ultrasound* | ||||||||
| Reinehr2009 | ✓ | ✓ | ✓ | ✓ | 24 months | ALT*, BMI*,ultrasound* | |||||||
| Pozzato2010 | ✓ | ✓ | ✓ | 12 months | BMI*, MR*, serum lipids | ||||||||
| Koot2011 | ✓ | ✓ | ✓ | ✓ | 6 months | ALT*, AST*, BMIUltrasound* | |||||||
| Lavine2011 | ✓ | 24 months | ALTliver biopsy | ||||||||||
| Santomauro2011 | ✓ | ✓ | ✓ | ✓ | 12 months | Ultrasound*, BMI*, WC, AFA, HOMA-IR*, QUICKI* | |||||||
| Grønbæk2012 | ✓ | ✓ | ✓ | 12 months | ALT*, BMI*.ultrasound* | ||||||||
| Jin2012 | ✓ | ✓ | 2 days | MR, liver biopsy, glucose, insulin, serum lipids* | |||||||||
| Lee2012 | ✓ | ✓ | ✓ | 3 months | MR* and total abdominal fat by DXA* | ||||||||
| DeVore2013 | ✓ | ✓ | ✓ | ✓ | 12 months | ALT*BMI* | |||||||
| Verduci2013 | ✓ | ✓ | ✓ | ✓ | 12 months | ALT*, MR*ultrasound* | |||||||
| Murer2014 | ✓ | ✓ | ✓ | 4 months | BMI, oxidative stress, ALT*, serum lipids, glucose, insulin | ||||||||
| Boyraz2015 | ✓ | ✓ | ✓ | 12 months | ALT*, ALT*, HOMA-IR, ultrasound* | ||||||||
| Fonvig2015 | ✓ | ✓ | 12 months | BMI*, serum lipids, insulin, HbA1c, MR* | |||||||||
| Goldschmidt 2015 | ✓ | ✓ | 12 months | Ultrasound*, elastography*, hyaluronic acid test* | |||||||||
| Janczyk2015 | ✓ | ✓ | 24 weeks | ALT*,ultrasound | |||||||||
| Koot2015 | ✓ | ✓ | ✓ | ✓ | 24 months | ALT*, MR* | |||||||
| Mager2015 | ✓ | ✓ | ✓ | 6 months | ALT*, BMI*, HOMA-IR* | ||||||||
| Kwon2016 | ✓ | ✓ | 12 months | ALT*, AST | |||||||||
AFA: arm fat area; ALT: alanine aminotransferase; AST: aspartate aminotransferase; BMI: body mass index; DXA: dual-energy x-ray absorptiometry; HbA1c: glycosylated hemoglobin; HOMA-IR: homeostatic model assessment for insulin resistance; MR: magnetic resonance; QUICKI: quantitative insulin-sensitivity check index; WC: waist circumference.
In none of the studies reviewed was the school-age patient group reported as the only group. Four studies were exclusively on adolescents and 20 studies combined both age groups. Patient recruitment was mixed, from the hospital context, as outpatients, and from events outside of health institutions, such as camps. In two studies, there was a minimum intervention duration of 4 weeks and a maximum of 24 months and 50% of the interventions were carried out for about one year.
Nutritional interventionsTreatment of children and adolescents with NAFLD was through dietary intervention in 15 of the 22 intervention studies reviewed (Table 3) and they differed in the type of dietary nutrition plan utilized. Maintenance diets with no energy intake or nutrient restrictions were managed in about 70% of the studies, together with general recommendations with respect to the quality of the diet. Energy intake restriction was utilized as part of the treatment in only 2 studies. In the trial conducted by Pozzato et al., the intervention was strictly nutritional and they reported a statistically significant decrease in BMI and hepatic steatosis, albeit there was no reduction in ALT and AST levels after one year of the intervention.26 In 4 studies in which a diet with a normal caloric intake was managed, fructose restriction was emphasized.27,32,33,37 Nutritional intervention was combined with planned physical activity in the majority of the studies reviewed, and in some cases there was antioxidant supplementation. In most of the studies that combined those intervention variables, there was improvement in the outcome variables, especially regarding the decrease in ALT levels, a reduced frequency of steatosis cases identified through imaging studies, and a slight decrease in body adiposity evaluated through body mass index and other methods.
Physical activityThe most recurrent recommendation in all the clinical trials reviewed was that of physical activity and/or systematic exercise, and related results were reported in approximately 90% of the studies (Table 3). However, rigorous exercise plans were not carried out in all the cases. There were indications for increasing physical activity and reducing sedentary behavior, such as including 30-60min of aerobic activity 3-6 times a week. It should be clarified that physical activity was not used as the sole intervention variable in any of the works reviewed, given that variables related to diet, antioxidant supplementation, or educational activities were managed in all of them.
Three interventions were carried out in contexts in which the patients had to attend summer camps or spend some time in the hospital, and therefore the physical activity was more structured, and the times of activity were monitored. The activities included swimming, cycling, gymnastics, track and field, and basketball, for at least one hour 6 times a week. Most of the physical activity interventions focused on aerobic exercise. Only Grønbæk and Lee reported results of interventions related to anaerobic exercise, and Lee described improvement in insulin sensitivity.28,31
Antioxidant supplementationAntioxidant supplementation has been a recurrent theme in the treatment of NAFLD in the pediatric population (Table 3). The most widely used antioxidants have been alpha-tocopherol, ascorbic acid antioxidants, and the omega-3 fatty acids. Supplementation with alpha-tocopherol and ascorbic acid was reported by 4 authors. Nevertheless, those supplements were not used alone, but together with dietary and physical activity plans. No significant differences were demonstrated between the study groups in relation to the intervention variable of antioxidant supplementation, including the dose. The decreases in cases of hepatic steatosis and ALT and AST levels were attributed to lifestyle modifications, and not specifically to the supplementation. A multicenter study on 173 pediatric patients with NAFLD that included 10 universities in the United States could not demonstrate reduced ALT levels after the 24-month administration of metformin and vitamin E.30
Differences were shown in studies in which supplementation with omega-3 fatty acids was evaluated. Two studies included omega-3 supplementation, but as in the studies assessing other antioxidants, they were combined with lifestyle changes. Both studies showed significant differences between the intervention groups that received omega fatty acids and those that received placebo.
CounselingThe interventions for lifestyle modification included nutritional or psychologic counseling (Table 3). Seven trials provided individual nutrition counseling to the patients. Regardless of the dietary nutrition plans, said counseling was focused on diet quality and the recommendations were directed especially at reducing the consumption of simple carbohydrates, sweetened drinks, trans fats, and junk food, in addition to providing healthy recipes. Two trials included group counseling sessions. Only Koot and Goldschmidt included both individual and group counseling in their studies.38,39
Individual and group counseling with psychologists was included in the interventions of 6 studies. Among the themes dealt with were psychosocial skills, dietary behaviors, attitudes toward foods, sleep disorders, and hygiene. It should be pointed out that 7 of the 22 studies included psychologic and nutritional education counseling with parents and children.
DiscussionThe main finding from the present scoping review was that in 21 of the 22 studies, the authors reported improvement in the condition of the patients with NAFLD after undergoing the intervention. That improvement was related to the decrease or suppression of steatosis evaluated through imaging studies or liver biopsy, as well as to the improvement or normalization of biochemical indicators, especially serum liver enzyme concentrations, which occurred in a varying number of cases during the differing duration of the interventions. The decrease in adiposity evaluated through anthropometric or imaging study indicators was clearly related to the efficacy of the intervention carried out, observed even with marginal changes in BMI. Those results ratify the close association between NAFLD and obesity in children and adolescents and indicate that modification of the variables in the energy equation that explain obesity when energy intake exceeds energy expenditure can lead to a resolution, albeit partial or temporary, of NAFLD.
The nutritional intervention was the common denominator in the majority of the intervention studies. The dominant trend was the application of normocaloric dietary plans involving normal energy intake in accordance with the age and sex of each subject, with no restrictions and balanced (50% carbohydrates, 20% proteins, and 30% lipids), added to the recommendations for improving the quality of foods consumed. That criterion corresponds to the US practice guidelines for the diagnosis and management of NAFLD, which state that weight loss should be slow and constant during treatment and follow-up because big fluctuations in weight can increase lipid deposits in the liver, in addition to compromising linear growth.44
The antioxidant, α-tocopherol, is the most commonly evaluated agent in the treatment of NAFLD because its antioxidant activity is secondary to its effect of stabilizing cell membranes through protection against lipid peroxidation and the production of free radicals that cause cell damage. However, in the present review, the clinical trials that treated the patients with α-tocopherol reported a lack of statistically significant differences between the intervention groups and the controls. The same results were found in the studies that combined α-tocopherol with ascorbic acid. In two studies with omega-3 fatty acid supplementation, the results were more encouraging than those with α-tocopherol. Both studies showed improvement in the outcome variables considered NAFLD indicators, regardless of diet and physical activity.23,29 Omega-3 fatty acids have been shown to be efficacious in reducing lipogenesis, as well as inflammation, in the liver and are an alternate intervention route regarding the variables related to the energy equation in the treatment of NAFLD.45 Nevertheless, controlled clinical trials are needed to ratify their efficacy.
The intervention in some of the studies reviewed was multiple, encompassing the change in lifestyle concept, considered a central factor in the prevention and treatment of childhood obesity. In the study by Grønbæk et al., the subjects attended a 10-week summer camp at which they received dietary plans with no supplementation, controlled foods, structured physical activity, and nutritional and psychologic counseling.28 An ambulatory intervention was carried out in the study by Boyraz et al. and included a normocaloric dietary plan, recommendations for structured physical activity, and omega-3 fatty acid supplementation.41 The types of studies that had more than one intervention variable and achieved improvement in the outcome variables exemplify the most efficacious intervention models. They can be carried out in empiric clinical trials, which are closer to the everyday clinical reality.46
Among the intervention studies related to physical activity -the second component of the energy equation- three trials with structured and monitored interventions were identified, two of which combined aerobic and anaerobic physical activity. Only general recommendations were provided in the rest of the studies. Encouraging results, such as reducing the BMI and a decrease in some or all of the obesity and NAFLD indicators, were shown in the trials that combined diet and physical activity. The development of new physical activity interventions is likely, given that those recommendations and practices should be based on the daily life of the patients, with no need to restrict their performance to a specific location.28,31
An essential aspect for achieving intervention efficacy was nutritional and psychologic counseling. The interventions that involved both the subjects and their parents had successful results, regardless of intervention duration. For example, in the study by Koot et al., follow-up with nutritional and group behavioral counseling that included parents and children was carried out at 6 and 24 months, resulting in a reduced frequency of steatosis in the children, as well as a decrease in ALT values.29
In 2018, there was no consensus on the treatment of NAFLD in children and adolescents. The results of the present review clearly show that today, lifestyle modifications that include diet, exercise, and education are the fundamental pillars in therapeutic interventions. However, the results of the interventions described herein are partial, and probably temporary, and most likely are not the solution to the overall problem of obesity and NAFLD. An alternative solution would be to focus on pregnancy and the first three to five years of life, the stage of life at which the central factors for a healthy lifestyle are established, which are: metabolic programming, the partial structuring of body composition, and dietary and physical activity habits.
The strength of the present scoping review was the identification of the intervention variables that over the past 12 years have been shown to be efficacious in the partial, and probably temporary, modification of obesity-associated NAFLD in children and adolescents. The weakness of the review, inherent in the methodology of selected works, was the variability in intervention duration, intervention measures, and the outcome variables.
In conclusion, the present scoping review related to the efficacy of intervention studies in obese children with NAFLD identified the fact that changes in family lifestyle, a healthy diet, regular physical activity, and health education are the current resources in the treatment of pediatric patients with NAFLD associated with obesity. Those intervention measures tend to provide partial, and probably temporary, results.
Financial disclosureA grant from the Consejo Nacional de Ciencia y Tecnología (CVU # 385932) for doctoral studies was received.
Conflict of interestThe authors declare there is no conflict of interest.
Please cite this article as: Caro-Sabido EA, Larrosa-Haro A. Eficacia de la intervención nutricia y de la actividad física en niños y adolescentes con hígado graso no alcohólico asociado a obesidad: revisión sistemática exploratoria. Revista de Gastroenterología de México. 2019;84:185–194.