Chronic kidney disease is one of the main comorbidities affecting liver transplant recipients. Most of those patients have some degree of acute or chronic kidney dysfunction at the time of transplantation, moreover they can also develop de novo chronic kidney disease once transplanted. An important increase in the incidence of chronic kidney disease in the «MELD era» has been observed. This phenomenon has partially been attributed to the weight that kidney function carries for organ allocation. In addition, the generalized use of calcineurin inhibitors has also been a contributing factor. It is of the utmost importance for us to be familiar with the current methods for evaluating kidney function before and after a liver transplantation. The two main biomarkers available today for that purpose are serum creatinine and cystatin C. Several equations have been derived from those biomarkers and have been tested in that context with mixed results, due to their biologic variability and the lack of standardization in their measurement. The gold standard continues to be the direct determination of the glomerular filtration rate through different methods, however that is only done for research purposes. It is also essential to know the current classification of acute kidney injury and chronic kidney disease in order to make early diagnosis. The present review focuses on the recognition, diagnosis, and classification of chronic kidney disease and acute kidney injury in liver transplantation recipients.
La enfermedad renal crónica (ERC) es una de las principales comorbilidades que afectan a los pacientes receptores de trasplante hepático. Muchos de ellos son trasplantados con cierto grado de disfunción renal aguda o crónica, sin embargo, también pueden desarrollar ERC de novo una vez trasplantados. Se ha observado un incremento importante de la incidencia de ERC en la «era MELD», en parte se ha atribuido este fenómeno al peso que tiene la función renal para la asignación de órganos. Por otro lado, el uso generalizado de inhibidores de calcineurina también ha contribuido a este fenómeno. Es de suma importancia familiarizarnos con los métodos actuales para evaluar la función renal antes y después de un trasplante hepático. Los 2 principales biomarcadores disponibles hoy en día para este fin son la creatinina sérica y la cistatina C. Existen varias ecuaciones derivadas de estos biomarcadores que han sido probadas en este contexto, obteniéndose resultados mixtos debido a su variabilidad biológica y a la falta de estandarización en su determinación. El estándar de oro continúa siendo la medición directa de la tasa de filtrado glomerular por diversos métodos, sin embargo, esto solo se realiza con fines de investigación. También es importante conocer la clasificación actual de la lesión renal aguda y de la ERC con el fin de poder realizar un diagnóstico temprano. Esta revisión se centra en el reconocimiento, diagnóstico y clasificación de la ERC y la lesión renal aguda en pacientes receptores de trasplante hepático.
Chronic kidney disease (CKD) and acute kidney injury (AKI) are frequent complications in patients with advanced liver disease that are on the liver transplantation (LT) waiting list. Likewise, they are the main comorbidities that present, once those patients are transplanted. Currently, more than 75001 liver transplants are carried out annually in the United States with excellent long-term results. That success is partially due to the introduction of the calcineurin inhibitors (CNIs) as maintenance immunosuppressive therapy. However, those agents have been shown to be intrinsically nephrotoxic, through a mechanism mediated by chronic kidney vasoconstriction. They also develop other potentially adverse effects for the kidney, such as: dyslipidemia, high blood pressure, and diabetes.2 In their study on kidney biopsies performed in recipients of LT, O’Riordan et al. reported that the main causes of CKD were: toxicity due to CNIs (48%), diabetic nephropathy (19%), membranoproliferative glomerulonephritis (17%), IgA nephropathy (9%), acute tubular necrosis (4%), and crescentic glomerulonephritis (3%).3 Gonwa et al. attributed 73.3% of the causes of end-stage renal disease (ESRD) to CNI toxicity, followed by hepatorenal syndrome, in which kidney function was not recovered.2
The high prevalence of acute or chronic kidney function alterations in solid-organ recipients and its association with reduced overall survival,4 makes opportune diagnosis and treatment essential. The prevalence of kidney dysfunction in patients on the LT waiting list varies greatly. The prevalence of CKD has been reported from 30 to 90% and of AKI from 25 to 50%.5 That variability is partially due to the fact that the current diagnostic methods are inaccurate. At the end of the 1990s, Nair et al. evaluated the presence of kidney dysfunction in almost 20,000 LT recipients between 1988 and 1996. At the time of transplantation, the glomerular filtration rate (GFR) was altered in 33% of the patients. Those authors showed that a pre-transplantation estimated GFR (eGFR) < 40ml/min/1.73 m2 was associated with lower short-term and long-term survival in LT recipients. Lastly, they were the first authors to suggest the performance of combined liver-kidney transplantation (CLKT) in patients with an eGFR under 30ml/min/1.73 m2.6
Impact of MELD on the development of chronic kidney diseaseIn an attempt to reduce the mortality rate in patients on the transplant waiting list, giving priority to the “most gravely ill”, the Model for End-stage Liver Disease (MELD) score was adopted in the United States in February of 2002 for organ allocation. That model predicts mortality at 3 months and utilizes 4 variables: serum creatinine (SCr), total bilirubin, INR, and renal replacement therapy (RRT).7
Said model was adopted as the main instrument for organ allocation in the United States. The weight of SCr has been postulated to be disproportionate in the MELD formula, and therefore, since its introduction, patients with kidney dysfunction (acute or chronic) have had priority on the waiting list.8 In their retrospective analysis of 25,023 transplantations performed in the post-MELD era, Gonwa et al. found a significant increase in the number of transplanted patients with SCr > 1.0mg/dl or on hemodialysis, as well as over twice the number of CLKTs. The patients with a high MELD score had an even higher risk for developing CKD and its complications.9
Thus, it is not surprising that the incidence of advanced CKD has increased in the post-MELD era. Sharma et al. analyzed a cohort of 221 patients that received a LT within the time frame of February 2002 and February 2007. They defined post-liver transplantation (post-LT) CKD as an eGFR < 30ml/min/1.73 m2 for 3 months or longer, initiation of RRT, or being placed on the kidney transplantation list. With a mean follow-up of 2.6 years (range: 0.01-5.99), they found that 31 patients developed CKD with a 5-year cumulative incidence of 22%. At the time of transplantation, 36% of their patients had an eGFR < 60ml/min/1.73 m2 and the pre-transplantation eGFR was the only independent predictor for the development of post-LT CKD (HR = 1.33; p < 0.05).10
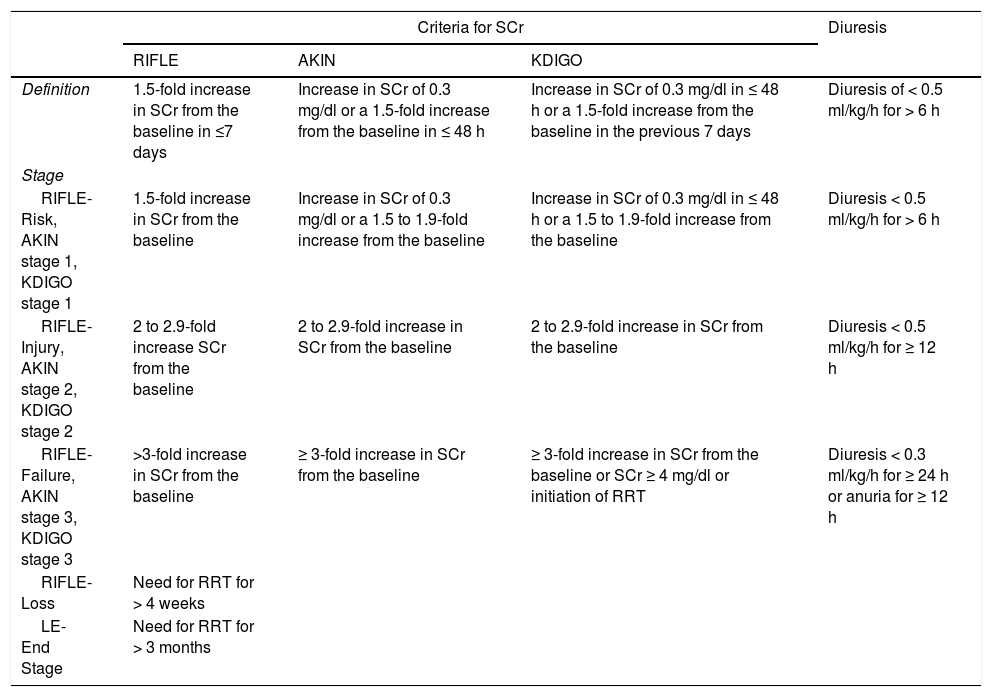
Not only has the risk for CKD increased in the post-MELD era, but also the risk for end-stage renal disease (ESRD), meaning those patients that need renal replacement therapy. That was apparent in the study by Sharma et al. in which they compared the incidence of ESRD before and after the introduction of MELD. They included patients that were LT recipients from April 1995 to December 2008 (n = 59,242), with data obtained from the Scientific Registry of Transplant Recipients (fig. 1). Their main finding was that the relative risk for developing ESRD had increased by 15% (p = 0.0049) since 2002. That condition was also strongly associated with mortality (HR = 3.32; p < 0.0001).11
Trend in the covariance-adjusted incidence of CKD according to the year in which liver transplantation was performed. Before 2002 (pre-MELD era), the post-liver transplant CKD rate significantly decreased 5.1% per year (RR 0.949; 95% CI 0.924 to 0.975); p = 0.0001). However, that trend was reversed in 2002 (MELD era), showing an annual increase in ESRD of 7.6% (p = 0.0001). MELD: Model for End-stage Liver Disease. Source: adapted from Sharma et al..11
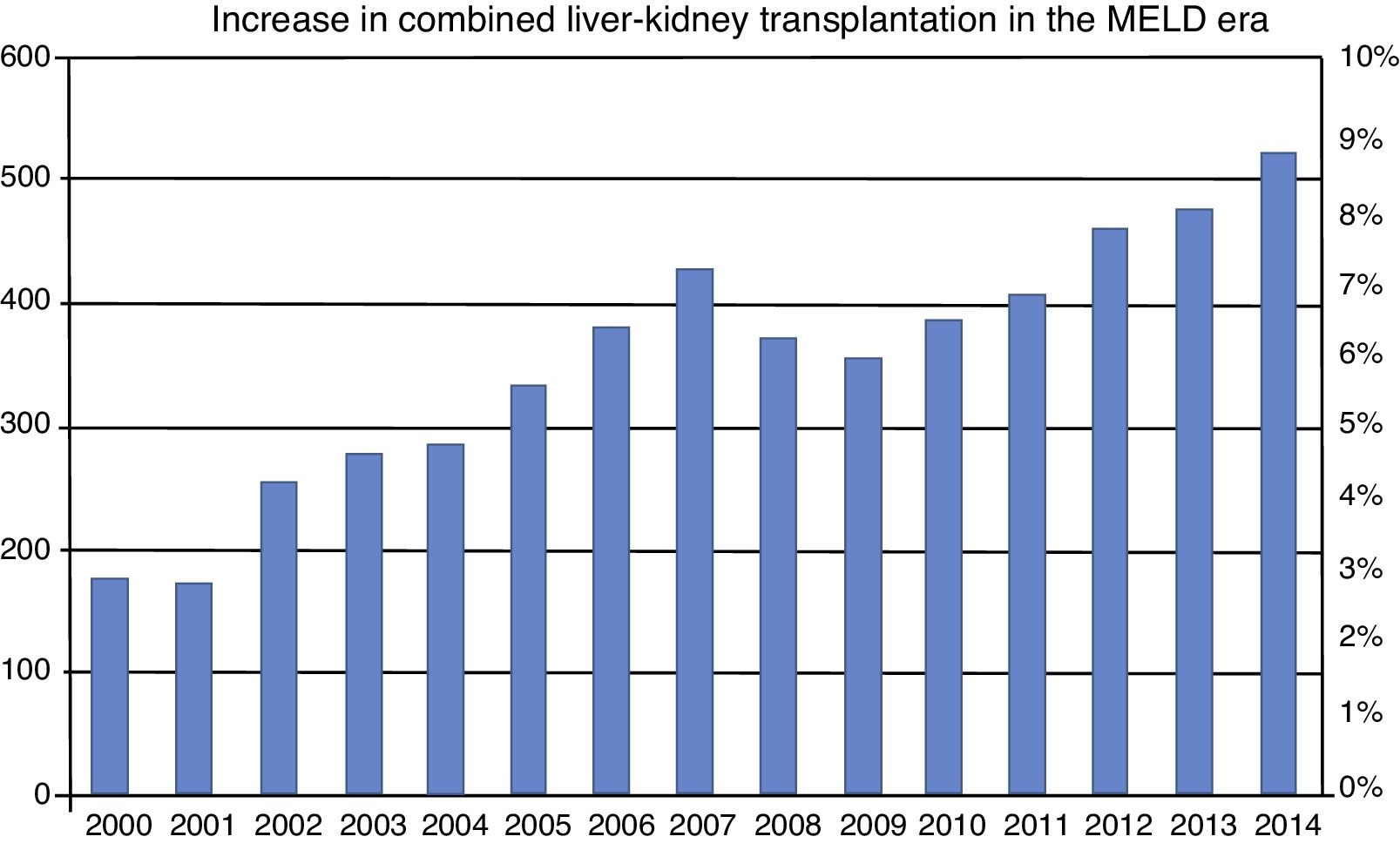
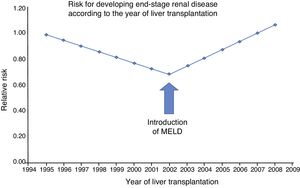
Furthermore, an important increase in the number of CLKTs has been observed in the post-MELD era. Simply in the 2005 yearly report in the United States, the number of CLKTs performed was 425, signifying a 200% increase with respect to the pre-MELD era12 (fig. 2). Table 1 lists the currently accepted criteria for a patient to be considered a candidate for a CLKT.
Incidence of combined liver-kidney transplantation from 2000 to 2014. The number of combined transplants has notably increased since the inclusion of MELD in February of 2002. Incidence of that type of transplant in the pre-MELD era was under 3%. It has increased to more than 8% in the MELD era. MELD: Model for End-stage Liver Disease. Source: adapted from Sharma et al.38
Criteria for a CLKT in patients on the waiting list for LT.
| Candidates with persistent AKI > 4 weeks plus one of the following: |
| a. Stage F (RIFLE) |
| b. GFR ≤ 35 ml/min/1.73 m2 (estimated by MDRD) or ≤ 25 ml/min (measured through 125I-iothalamate clearance) |
| Candidates with KDOQI-defined CKD for at least 3 months, plus one of the following: |
| a. GFR ≤ 40 ml/min/1.73 m2 (estimated by MDRD) or ≤ 30 ml/min (measured through 125I-iothalamate clearance) |
| b. Proteinuria > 2 g/day |
| c. Kidney biopsy with > 30% glomerular sclerosis or > 30% interstitial fibrosis |
| d. Presence of metabolic disease |
AKI: acute kidney injury; CKD: chronic kidney disease; CLKT: combined liver-kidney transplantation; GFR: glomerular filtration rate.
Likewise, the rate of sequential kidney transplantations, that is to say, those performed in liver transplantation recipients, rose. The total number of sequential kidney transplantations increased from 37 to 124 per year in 2008 (0.9% of all kidney transplantations carried out). Gonwa et al. analyzed the kidney transplants performed from 1997 to 2008, utilizing information from the Organ Procurement Transplant Network (OPTN) database. Within that time frame, 157,086 kidney transplantations were carried out, and of those, 680 deceased-donor transplants and 410 living-donor transplants were performed on patients with a previous LT.13
Evaluation of kidney function before liver transplantationAccurate determination of kidney function is a critical aspect of the evaluation of patients that are candidates for receiving a LT. It is relevant for the fair allocation of organs destined for a solitary transplantation and for decisions concerning combined transplants. Different studies have shown that the equations based on SCr are not sufficiently precise for determining GFR in cirrhotic patients that are candidates for LT.14–15 The introduction of new formulas for estimating GFR based on serum cystatin C (CysC) levels or in combination with SCr renewed the hope of having a noninvasive method that would enable us to know the kidney function of those patients, but the results have been mixed.
The most widely used biomarkers and equationsSCr is the most widely used biomarker and its utilization goes back to 1920. It is an amino acid with a molecular weight of 113Da, end product of the non-enzymatic degradation of creatine in skeletal muscle. It is distributed throughout the total body water, is freely filtered, is not bound to proteins, and is not metabolized in the kidneys. Those characteristics could make it a good glomerular filtration marker, but several extrarenal determinants influence its serum concentration, thus decreasing its usefulness. The main factors are: age, sex, race, height, and diet, among others. Of those, muscle mass is the most relevant, given that the majority of patients with cirrhosis of the liver have some grade of malnutrition. Moreover, approximately 10-20% of SCr excretion is due to tubular secretion and its degradation by the gut microbiota. Those factors reduce its concentration, resulting in a tendency to overestimate the GFR. Drugs, such as trimethoprim or cimetidine, reduce its tubular secretion and have the opposite effect on the eGFR.16
The equations for calculating eGFR based on SCr are the most widely used worldwide and those with which there is the most experience. The Cockcroft-Gault formula is named after the authors that proposed it in 1976. It estimates creatinine clearance based on age, sex, and weight. It was developed utilizing data from 249 male patients and is adjusted in females, assuming less creatinine production. It has currently fallen into disuse due to its lack of accuracy, because it systematically overestimates the GFR in patients with overweight or edema.17 The formula derived from the Modification of Diet in Renal Disease (MDRD) Study uses age, sex, race, and standardized SCr for calculating the eGFR. It was described in the study population of 1628 patients with chronic kidney disease that had measured GFR (mGFR) computed through 125I-iothalamate clearance. Given the population in which it was developed, it is not accurate in cases of high GFR levels (> 60ml/min/1.73 m2).18 There are 2 versions of that formula, one with 6 variables (that also takes BUN or urea and serum albumin into account) and the classic one with 4 variables, both of which have equal levels of accuracy.19
In 2009 another equation based on creatinine was described by the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) group that included a more diverse population: youths, diabetics, transplanted individuals, and persons with and without CKD diagnosis, in an effort to improve the limitations of the MDRD equation. It utilizes the same 4 variables as the MDRD but corrects GFR underestimation in young persons or those with better kidney function. In other words, it is as accurate as the MDRD equation for eGFR < 60ml/min/1.73 m2, but more accurate in values > 60ml/min/1.73 m2.18
Another biomarker that has recently gained relevance is CysC, a cysteine protease inhibiting enzyme of 13 KDa that is present in all nucleated cells of the organism. It is distributed in the extracellular space and freely filtered. Posteriorly, it is totally reabsorbed into the tubules, where it is degraded. Thus, under normal conditions, it cannot be recovered from urine. It has no tubular secretion or significant extrarenal elimination. Serum concentration of CysC is not dependent on sex, age, muscle mass, or nutritional status. Those characteristics make it an excellent marker for GFR, most likely superior to SCr. Nevertheless, its concentration can be altered in thyroid diseases, neoplasias, inflammatory states, and with glucocorticoid use.20 In general, its behavior in healthy individuals is known, but it could have a different behavior in sick patients. One of the main advantages that CysC has over SCr is its lack of dependence on muscle mass, making it more accurate in diverse body compositions.16 On the other hand, its quantification is more expensive than that of SCr, and not all laboratories have the standardized assays.
Serum CysC has been shown to better predict GFR because it is less affected by age, race, sex, and nutritional status than SCr, and therefore has been considered a potential alternative for calculating GFR.21 Likewise, CysC is a better predictor of cardiovascular mortality or mortality due to any cause, than SCr.22 Nevertheless, the equations that are based solely on CysC have not been shown to be more accurate than those based on SCr, due to the non-GFR determinants of CysC levels.23
In 2012, Inker et al. from the CKD-EPI group, in collaboration with the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), published a watershed study for the calculation of eGFR utilizing both markers. Their data was retrieved from 5352 patients from 13 different studies that had mGFR determined through 125I-iothalamate clearance. They developed 2 equations: one based solely on CysC (CKD-EPI-CysC) and another that combined SCr and CysC (CKD-EPI-Cr-CysC). They concluded that the CKD-EPI-Cr-CysC equation performed better and was more accurate for GFR, compared with 125I-iothalamate clearance, and that said equation could be useful for confirming CKD.23 A calculator that includes those equations can be accessed online at: www.kidney.org/professionals/kdoqi/gfr_calculator.
Equation performance for calculating eGFR in cirrhotic patientsDe Souza et al. conducted the first study comparing the performance of the equations described by the CKD-EPI groups (CKD-EPI-Cr, CKD-EPI-CysC, and CKD-EPI-Cr-CysC) with the most well-known formulas (MDRD-4, MDRD-6 and Hoek)24 in cirrhotic patients evaluated for LT. Renal clearance of inulin was the gold standard employed. The population sample consisted of 202 subjects assessed for LT that were mainly Caucasian and whose causes of liver failure were predominantly alcoholic cirrhosis of the liver and chronic hepatitis C virus or B virus infection. The sample was divided into the following subgroups: subjects with ascites classified according to severity and the grade of liver dysfunction (MELD ≤ 15 or > 15) and renal dysfunction (Kidney Disease Outcomes Quality Initiative [KDOQI] classification). In general, the mean GFR was 80 ± 31ml/min/1.73 m2, 74% of the subjects had a mGFR ≥ 60ml/min/1.73 m2, and it was < 60ml/min/1.73 m2 in 26%. A total of 32% of the subjects had Child-Pugh class C disease and 18% had refractory ascites. The mean MELD score was 11 ± 4 and 84% of the subjects had a MELD score < 15. In general, the equations based on CysC (CKD-EPI-CysC and CKD-EPI-Cr-CysC) showed better performance (less bias and better accuracy between 10 and 30%) in the 3 ascites groups and the 2 subgroups (MELD and Child-Pugh). As expected, the performance of the MDRD-4, MDRD-6, and CKD-EPI-Cr equations was poorer, especially as the grade of ascites worsened. The CKD-EPI-CysC equation performed better in patients with severe ascites and with an initial mGFR < 60ml/min/1.73 m2. Nevertheless, only 47-67% of the LT candidates were correctly classified according to the KDOQI classification. In that study, the authors found that the equations based on SCr had a tendency to overestimate GFR, whereas those based on CysC tended to underestimate it, especially in subjects with a mGFR > 60ml/min/1.73 m2.25
Mindikoglu et al. compared the performance of the CKD-EPI-Cr-CysC equation (using the parameters: bias, precision, and accuracy) with the Cockcroft-Gault, MDRD-4, MDRD-6, CKD-EPI-Cr, and CKD-EPI-CysC equations and 24-h SCr clearance. They analyzed 72 patients with cirrhosis of the liver using mGFR measured through 125I-iothalamate as the reference. The majority of the subjects (40%) had liver cirrhosis that was secondary to HCV infection. A total of 35% of the subjects did not present with ascites, 37% had ascites that responded to diuretics, and 28% had refractory ascites. With respect to CKD, 29% of the subjects had mGFR < 60ml/min/1.73 m2 It was significantly lower in subjects with ascites (71.1 vs. 106.9ml/min/1.73 m2, respectively, p = 0.0001). Unlike the study by De Souza et al.,25 those authors excluded patients diagnosed with hepatocellular carcinoma. They found that the accuracy of the CKD-EPI-Cr-CysC equation was significantly better than the others analyzed. However, when compared with the mGFR, 23.6% of the subjects had a difference > 30% (1-P30) and 44.4% had a difference greater than 20% (1-P20). On the other hand, the CKD-EPI-Cr-CysC equation was more precise and accurate in patients with mGFR < 60ml/min/1.73 m2. 26
The results of those last 2 studies concur with that previously reported in the literature. The MDRD-6, CKD-EPI-CysC, and CKD-EPI-Cr-CysC equations showed acceptable performance and clinical applicability, but they were lower in patients with advanced CKD. The equations based on SCr tended to overestimate GFR, whereas those based on CysC tended to underestimate it. That is especially relevant when the eGFR nears 40ml/min/1.73 m2, the limit at which CLKT is recommended.27
A tendency for the traditional formulas to perform more poorly in patients with advanced cirrhosis or with ascites has been observed. According to the study by De Souza et al.,25 such is not the case with the new equations based on CysC, which could be one of their main advantages. However, said study included patients with predominantly low MELD scores. All in all, those data support the use of the new equations that utilize CysC, but unfortunately that marker is not available in the majority of centers.25–26,28.
eGFR = 45.9 x (SCr-.836 in ?mol/l) x (urea-0.229 in mmol/l) x (INR-0.113) x (age-0.129) x (Na-0.972 in mmol/l) x (0.809 if female) x (0.92 if ascites is moderate or severe)
Recently, Kalafateli et al.29 developed and validated a formula (using multiple linear regression by stages) for estimating GFR in cirrhotic patients and compared it with some of the existing formulas (MDRD-4, MDRD-6, CKD-EPI-Cr, CKD-EPI-CysC, and CKD-EPI-Cr-CysC). They included 469 subjects assessed for LT within the time frame of 2011 to 2014, and 2 cohorts for the internal and external validation of 82 and 174 patients, respectively. mGFR was determined through the clearance of a radioactive isotope. The derived equation with the maximal r2 (74.6%) is the following:
Said equation was named the Royal Free Hospital (RFH) Cirrhosis GFR and showed better performance and accuracy for predicting mGFR, with P10, P30, and P50 (the percentile variation with respect to mGFR) values of 56.1, 89, and 98.8%, respectively, in the external validation cohort and 45.4, 88.5, and 96.6% in the internal validation cohort, which was not influenced by the grade of liver dysfunction. The authors also reported a higher degree of accuracy compared with the equations based on CysC. However, they did not use a standardized method for their measurements and so did not consider it a relevant result.
If possible, our first option for determining GFR in cirrhotic patients that are LT candidates would be to measure it directly through one of the standard methods, such as inulin or 125I-iothalamate clearance. The second option would be to use the CKD-EPI-CysC or CKD-EPI-Cr-CysC equations, but not all of the assays for measuring CysC are standardized, their use is not generalized, and they are costlier. Finally, if there is no access to any of those methods, we can use the equation described by Kalafateli et al.29 (which has not been studied in a Mexican population) or the MDRD equation. We must not forget that the performance of the formulas for estimating GFR is poor in cirrhotic patients. The most reasonable explanation is that they were not described or validated in patients with cirrhosis of the liver and therefore their performance is sub-optimum. The RFH equation was designed to calculate GFR in cirrhotic patients, but it has not been validated in different populations, including those in Latin America.
Evaluation of kidney function after liver transplantationThe evaluation of kidney function after a LT is of vital importance for the later follow-up of those patients. The risk for CKD, or its progression to ESRD, in solid-organ recipients is high. In addition, having CKD confers greater morbidity and mortality on those patients.4
Post-LT acute kidney injuryThe development of AKI after a LT is one of the most frequent early complications. AKI has recently been described as one of the main risk factors for a future development of CKD and ESRD, thus the importance of its prevention, identification, and opportune treatment.30 Due to the numerous definitions used in different studies, the reported incidence varies widely. For example, when AKI is defined as an increase in SCr ≥ 50% higher than the preoperative level, postoperative incidence of 56.6% has been reported. The 2 most studied systems for defining AKI in the context of LT are the Risk, Injury, Failure, Loss, End-stage renal disease (RIFLE)28 and the Acute Kidney Injury Network (AKIN) classifications.31 When the definition of AKI according to the AKIN group is used, post-transplantation incidence is 60.1%.32 The Kidney Disease Improving Global Outcomes (KDIGO) guidelines on AKI published in 2012 combined the most important aspects of the previous classifications. Nevertheless, the older classifications are found in the majority of the articles in the literature33 (Table 2).
Definition of acute kidney injury.
| Criteria for SCr | Diuresis | |||
|---|---|---|---|---|
| RIFLE | AKIN | KDIGO | ||
| Definition | 1.5-fold increase in SCr from the baseline in ≤7 days | Increase in SCr of 0.3 mg/dl or a 1.5-fold increase from the baseline in ≤ 48 h | Increase in SCr of 0.3 mg/dl in ≤ 48 h or a 1.5-fold increase from the baseline in the previous 7 days | Diuresis of < 0.5 ml/kg/h for > 6 h |
| Stage | ||||
| RIFLE-Risk, AKIN stage 1, KDIGO stage 1 | 1.5-fold increase in SCr from the baseline | Increase in SCr of 0.3 mg/dl or a 1.5 to 1.9-fold increase from the baseline | Increase in SCr of 0.3 mg/dl in ≤ 48 h or a 1.5 to 1.9-fold increase from the baseline | Diuresis < 0.5 ml/kg/h for > 6 h |
| RIFLE-Injury, AKIN stage 2, KDIGO stage 2 | 2 to 2.9-fold increase SCr from the baseline | 2 to 2.9-fold increase in SCr from the baseline | 2 to 2.9-fold increase in SCr from the baseline | Diuresis < 0.5 ml/kg/h for ≥ 12 h |
| RIFLE-Failure, AKIN stage 3, KDIGO stage 3 | >3-fold increase in SCr from the baseline | ≥ 3-fold increase in SCr from the baseline | ≥ 3-fold increase in SCr from the baseline or SCr ≥ 4 mg/dl or initiation of RRT | Diuresis < 0.3 ml/kg/h for ≥ 24 h or anuria for ≥ 12 h |
| RIFLE-Loss | Need for RRT for > 4 weeks | |||
| LE-End Stage | Need for RRT for > 3 months | |||
RRT: Renal replacement therapy; SCr: Serum creatinine.
Upon their comparison, we found significant differences between the RIFLE and AKIN classifications for determining the incidence of AKI in post-LT patients. Karapanagiotou et al. applied both criteria to determine AKI incidence in a cohort of 71 patients. According to the RIFLE classification, 39.4% had post-LT AKI, but according to the AKIN criteria, 52.1% of the patients presented with AKI, resulting in a significant difference.34 In a recent study, Hilmi et al. determined that the incidence of AKI (using the KDIGO classification) within the first 72h post-LT was 52%, a similar rate to that previously published using the AKIN classification for AKI.35
The hemodynamic profile of patients with advanced cirrhosis of the liver, as well as the interventions utilized for their treatment, confer upon them an especially high risk for the development of AKI and/or CKD. That becomes even more relevant if the patient in question also has decompensated cirrhosis. AKI in cirrhotic patients can be classified into 3 large groups:5
- 1.
Intrinsic kidney damage, such as acute tubular necrosis or glomerulonephritis: 29%.
- 2.
Functional alterations due to reduced kidney blood flow, such as hepatorenal syndrome: 70%.
- 3.
Obstructive problems of the urinary tract: 1%.
Those different causes of kidney dysfunction are not mutually exclusive and may coexist, or even be part of a continuum in which intrinsic kidney damage can be preceded by a functional or obstructive alteration.36
Considering that a significant percentage of post-transplantation patients can develop AKI, it is important to know which factors are associated with its development. The risk factors identified include: a history of hepatitis C virus infection, diabetes mellitus, high blood pressure, kidney dysfunction (acute or chronic) prior to transplantation, diuretic use, the MELD and Child-Pugh scores, post-transplantation ICN use, time at which ICN use was begun, body mass index (BMI), and the need for packed red blood cell transfusion.5,35,37
The majority of those variables reflect the severity of the patient's condition prior to LT. The transfusion of blood derivatives, mainly packed red blood cells during surgery, reflects the bleeding and a reduction in effective intravascular volume. That then negatively impacts kidney function after the procedure, through a mechanism of local hypoxia and production of oxygen free radicals upon the re-establishment of normal circulation (ischemia-reperfusion).38 Obesity has been observed to affect the dosing and distribution volume of potentially nephrotoxic medications. Finally, CNIs, such as tacrolimus, induce transitory vasoconstriction of the renal artery, leading to deterioration in kidney function through the ischemia-reperfusion mechanism.39
Currently, there is no formula for estimating GFR in AKI. The customary formulas were described in patients with no acute or abrupt kidney function alterations, that is to say, at baseline kidney function. Therefore, their use in acute settings is inadequate, given that they overestimate the true GFR.
AKI and CKD prevention strategies are a theme that warrants a complete review, but they are worth mentioning briefly below:40
- 1.
Maintain adequate volume status chiefly using balanced solutions to prevent the appearance of hyperchloremic metabolic acidosis.41
- 2.
Prevent volume overload following the conceptual model of the 4 resuscitation stages: rescue, optimization, stabilization, and de-escalation.42 Bouchard et al. defined that condition as an increase in body weight ≥ 10% and associated it with an increase in mortality at 60 days in patients with AKI and with poorer kidney function recovery.43
- 3.
Ensure adequate kidney perfusion with a mean arterial pressure (MAP) > 65mmHg through the appropriate use of crystalloid or colloid (albumin) solutions to restore the effective circulating volume, as well as the early use of vasoactive amines (norepinephrine).40 In patients with a history of high blood pressure, an objective MAP of 80-85mmHg was associated with better kidney outcomes (AKI and RRT) in septic patients, but they also presented with a higher incidence of atrial fibrillation.44
- 4.
Different studies suggest that the surgical technique utilized in LT also plays a role in the development of AKI. In their study, Cabezuelo et al. posited that the so-called “piggy-back” technique significantly reduced the risk for postoperative AKI, compared with the customary technique (with or without venovenous bypass), which was attributed to the fact that with said technique there was no need for retrocaval dissection, reducing retroperitoneal blood loss.45
- 5.
Early CNI use has been associated with early AKI, but as mentioned at the beginning of the present review, the main problem is with their chronic use. Even though CNIs are an essential part of immunosuppression in the majority of solid-organ transplants, their long-term use can be nephrotoxic. Initially, they cause reversible vasoconstriction of the afferent arteriole, high blood pressure, metabolic acidosis, and hyperkalemia.46 Their chronic use produces CNI nephropathy, which is characterized by: arteriolar hyalinosis, obstructive arteriolopathy, atrophy, tubular vacuolization, interstitial fibrosis, and glomerular sclerosis. Some studies have shown that the late introduction of those drugs, their reduced dose, or CNI-free maintenance therapies substantially improve kidney function.5,47 On the other hand, Ishani et al. described a lower nephrotoxicity rate (defined as duplicating baseline SCr) associated with the use of tacrolimus, compared with cyclosporine A, in lung or heart transplantation patients.48
The prevalence of CKD in solid-organ recipients has been reported from 10 to 90%.37-38 That variability is explained by the different criteria used for its definition, the different methodologies of each study, but especially by the inherent limitations of the SCr-dependent formulas for calculating GFR. CKD diagnosis has been associated with a dramatic increase in adverse cardiovascular events, an increased hospitalization risk, and a 4-fold higher mortality rate, compared with patients with preserved kidney function.5
The largest and most complete study on that theme was conducted by Ojo et al., in which CKD was defined as a GFR < 30ml/min/1.73 m2 (calculated with the MDRD-4 formula). Those authors reported a 16% prevalence of CKD at 5 years in post-LT patients, surpassed only by patients with intestine transplantation, in whom prevalence was 21.3%4. It should be mentioned that if the latest CKD definition proposed by the KDIGO group had been used, which utilizes a GFR < 60ml/min/1.73 m2 and includes albuminuria, the prevalence would have been much higher. A recently published study by O’Riordan et al. examined the risk for CKD in 230 LT recipients, utilizing the KDOQI criteria. At 10 years, they found that 2.26% of those patients developed stage 5 CKD, 6.11% stage 4, 56.77% stage 3, and 23.7% stage 2. The rest of the patients had minimum or no kidney function deterioration.49 Likewise, the risk for developing ESRD has been reported at 5% per year in patients that have received a solid-organ transplantation or hematopoietic cell transplantation.50 Gonwa et al. reported an incidence of ESRD of up to 18% ten years after LT.2
The impact of kidney function on the survival of those patients was clearly described in the analysis by Allen et al. They conducted a prospective study on a cohort of LT recipients. Their aim was to determine the association of a decrease in GFR with patient survival. They included a total of 1211 transplantation recipients from the Mayo Clinic, where mGFR quantification through 125I-iothalamate clearance is part of the protocol in those patients. Twenty-five years after LT, 54% of the patients had died, 9% had undergone a kidney transplant, and 7, 21, and 18% had a mGFR > 60, 59 to 30, and < 30ml/min/1.73 m2, respectively. The mortality risk increased significantly when the mGFR decreased to < 30ml/min/1.73 m2: HR = 2.67 (95% CI: 1.8 to 3.96) for GFR of 29 to 15ml/min/1.73 m2 and HR = 5.47 (95% CI: 3.1 to 9.65) for GFR < 15ml/min/1.73 m2. It should be noted that the eGFR determined through the conventional formulas underestimated that risk in patients with a mGFR of 30 to 90ml/min/1.73 m2. 51
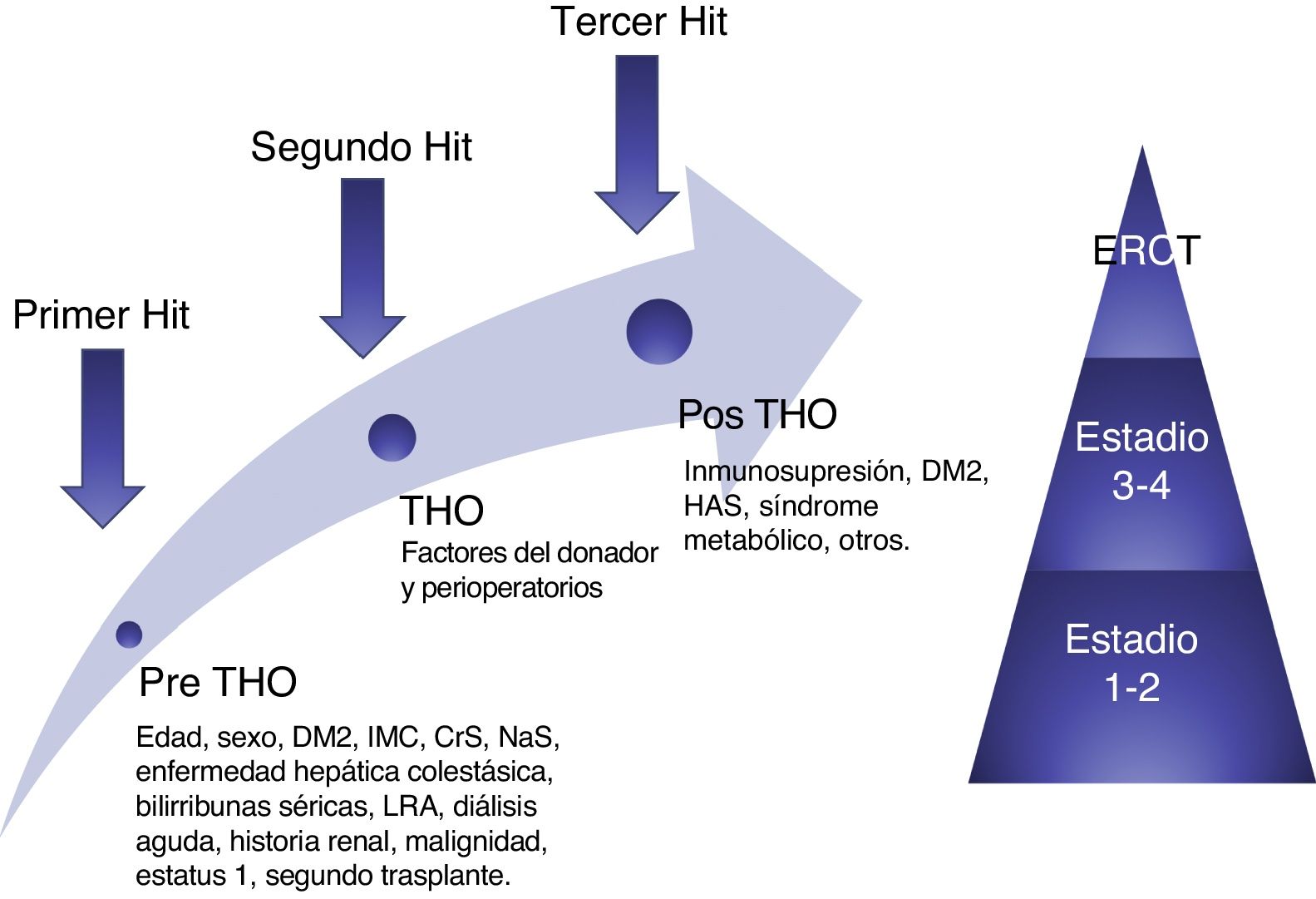
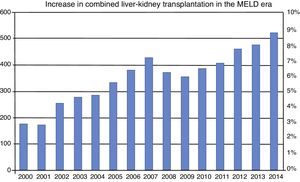
Sharma et al. recently proposed a theory of three “hits” for simplifying the complex pathophysiology of CKD. Prior to LT, patients with decompensated cirrhosis of the liver have compromised hemodynamics, especially renal hemodynamics, due to the presence of portal hypertension and systemic vasodilatation, constituting the first “hit”. During transplantation, there are inherent conditions, mainly hypovolemic episodes, that can affect kidney function, and are the second “hit”. The third “hit” is constituted by the factors that can eventually contribute to kidney function deterioration, such as: immunosuppressant use (CNIs), diabetes, high blood pressure, nephrotoxins, and subsequent AKI episodes37 (fig. 3).
Conceptual model of post-orthotopic liver transplantation CKD. The model shows the risk factors and 3 "hits" that result in kidney damage and finally contribute to the development of CKD. AKI: acute kidney injury; BMI: body mass index; CKD: chronic kidney disease; DM2: type 2 diabetes mellitus; ESRD: end-stage renal disease; OLT: orthotopic liver transplantation; SCr: serum creatinine; SNa: serum sodium. Source: adapted from Sharma et al.38
Identifying which patients are at higher risk for developing CKD in the medium term can be a great challenge. Several cohorts have been studied in an effort to identify the risk factors that have most frequently been associated with that complication. Historically, the most accepted are kidney function prior to transplantation, the demographic characteristics of each patient, patient comorbidities, AKI episodes in the peri-transplantation period, and chronic exposure to CNIs.52 In the study by Sharma et al., the main risk factors associated with the development of ESRD were: black race, hepatitis C, history of diabetes mellitus (DM) prior to LT, elevated pre-transplantation SCr levels, low serum albumin levels, low bilirubin levels, and serum sodium > 141 mEq/L at the time of transplantation.11
O’Riordan et al. developed a model that predicts the risk for reaching an eGFR below 30ml/min/1.73 m2 in the year following LT. They utilized a cohort of 368 patients from the United Kingdom that underwent LT, obtaining the patient data prospectively. The variables used in the model were: SCr, history of high blood pressure, grade of proteinuria, and duration of post-transplantation AKI. Those variables were evaluated before the transplant (except for the AKI episodes). In patients with a score above 2.16, their sensitivity and specificity were 99.2 and 100%, respectively (positive predictive value of 70%, negative predictive value of 100%), with an ROC area under the curve of 0.996 for predicting the development of CKD (GFR < 30ml/min/1.73 m2).52 They produced the following equation:
R = - 1.8 + [(0.001)(AKI duration in days) + (0.64)(proteinuria in g/24h) + (0.013)(SCr in ?mol/L)+(1.5 if there is a high blood pressure diagnosis)
Risk for progression to an eGFR < 30ml/min/1.73 m2at one year after transplantation
Israni et al. conducted a study on a cohort of LT recipients above 18 years of age, taking data from the Scientific Registry of Transplant Recipients (SRTR) and the US Renal Data System (USRDS), respectively, belonging to the MELD era. Patients with a previous transplant, multiorgan transplant, or those diagnosed with ESRD were excluded. The final sample analyzed included 39,611 patients. They developed a model for predicting the risk for developing ESRD at 6 months, and from 6 months to 5 years, after transplantation. The main independent predictors for the development of ESRD at 6 months were: age, history of DM, history of malignant neoplasias, BMI, SCr, hemodialysis the week prior to transplantation, and the donor risk index (DRI). With respect to the incidence of ESRD after 6 months, the main predictors were: DM at the time of transplantation, hepatitis C status, African American ethnicity, and the levels of serum albumin, total bilirubin, and SCr at the time of transplantation. The weakness of the last model was that it did not include the peri-transplant AKI episodes.53
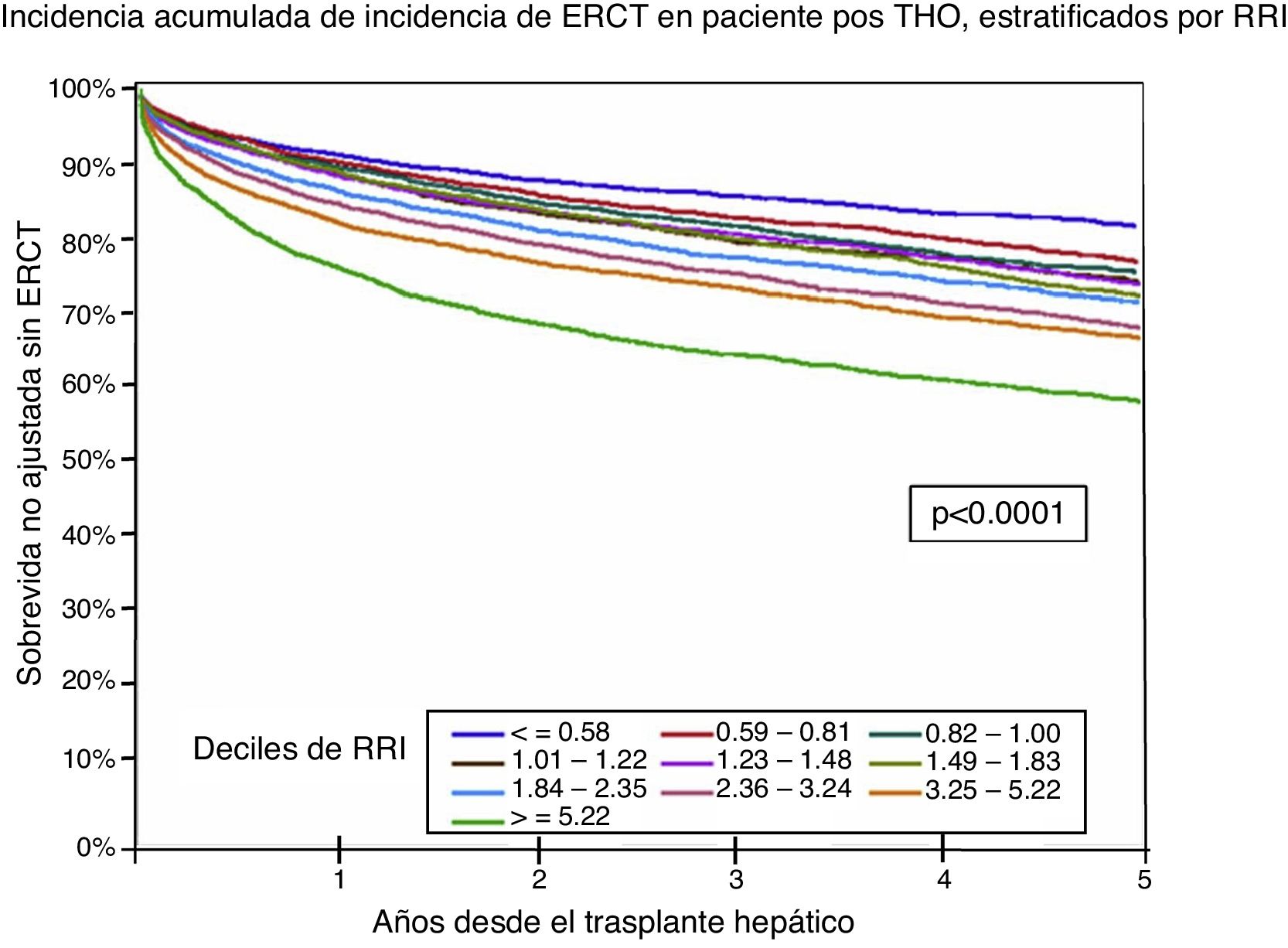
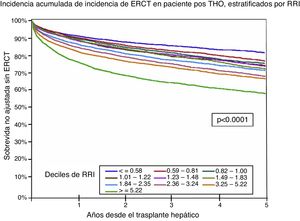
Finally, Sharma et al. carried out and validated a score for determining the risk for post-LT CKD, which they called the Renal Risk Index (RRI). That score was derived from a cohort of 43,514 LT recipients. It was based on 14 risk factors independently associated with the development of ESRD: age of the recipient at the time of transplantation, African American ethnicity, cholestatic disease, positivity for HCV, BMI, DM2 diagnosis, albumin, creatinine, bilirubin, and serum sodium levels, status 1, re-transplantation, history of TIPSS, and post-transplantation acute hemodialysis. The RRI was stratified into deciles based on the estimated risk, in which higher scores were associated with a higher rate of ESRD at 5 years. The RRI represents the relative risk for developing post-transplantation ESRD (95% CI: 0.75-0.78). That risk was also associated with a higher post-transplantation mortality rate54 (fig. 4).
Survival without ESRD after an orthotopic liver transplantation stratified by the RRI. ESRD: end-stage renal disease; RRI: renal risk index. Source: adapted from Sharma et al.55
Being aware of those risk factors enables us to formulate strategies for the prevention of developing ESRD, and therefore its importance cannot be over-emphasized. For example, in a patient at high risk for developing ESRD, we should maximize our efforts to prevent AKI events in the peri-transplantation period. On the other hand, there are conditions that we cannot modify, such as neoplasias or donor-derived conditions. Likewise, we must pay special attention to the treatment of the comorbidities associated with kidney function deterioration, mainly DM2, high blood pressure, and hepatitis C virus infection, and minimize exposure to CNIs.53-54
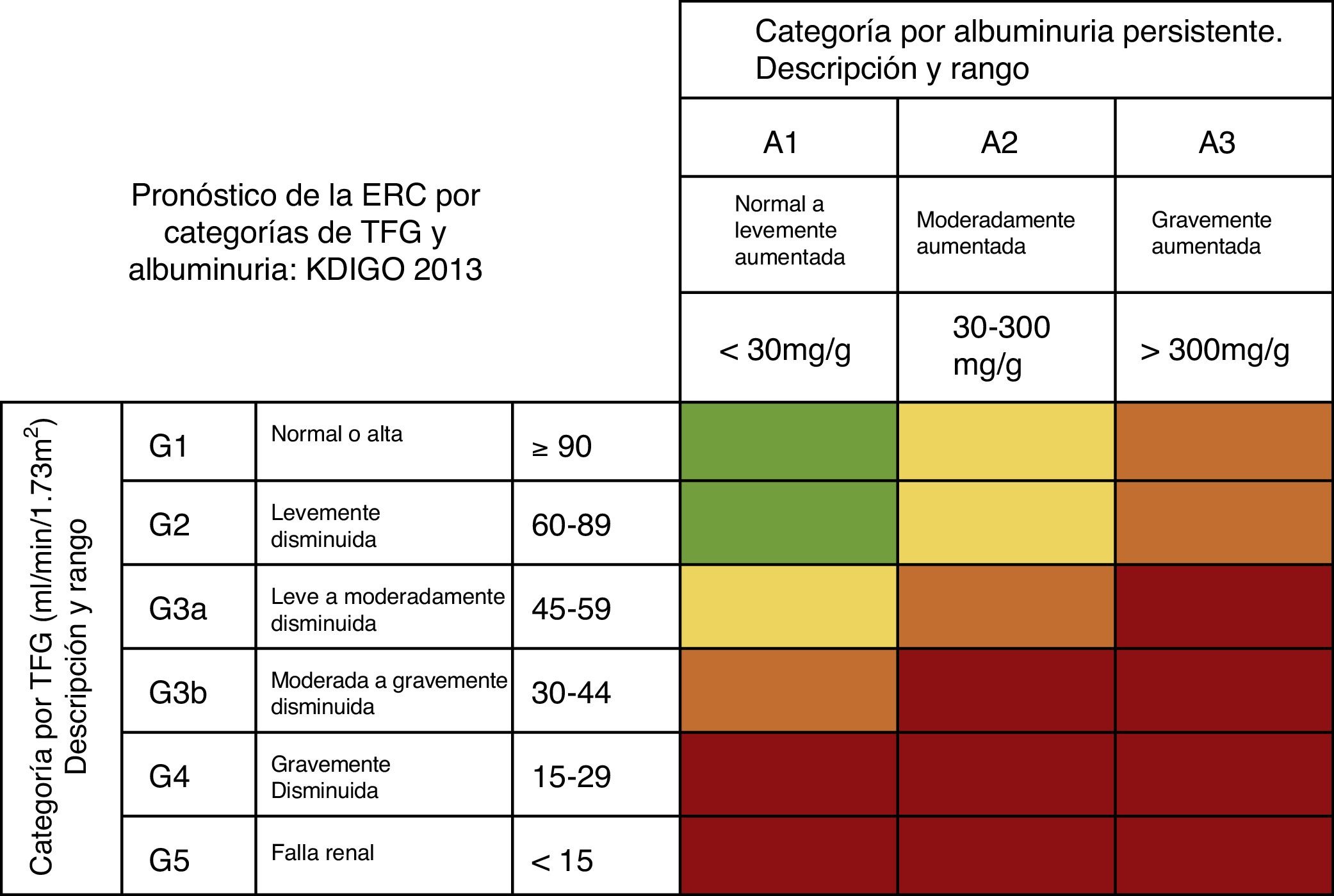
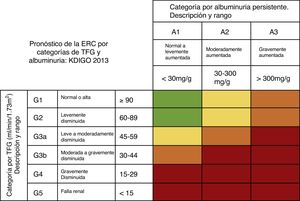
Diagnosis of chronic kidney disease after liver transplantationThe KDIGO group defines CKD as: the group of anatomic and functional kidney abnormalities present for more than 3 months, that have health implications. It is classified according to cause, GFR, and albuminuria27 (fig. 5).
Risk of progression and adverse outcomes of CKD: green: low risk (with no other kidney disease markers and considered not to be present); yellow: moderately increased risk; orange: high risk; red: very high risk. CKD: chronic kidney disease; GFR: glomerular filtration rate. Source: adapted from the KDIGO 2013 Guidelines.28
Correct GFR estimation is vitally important for the diagnosis and classification of CKD, but there is currently no precise and reliable method for its estimation in patients that have undergone a solid-organ transplantation. The “gold standard” is direct GFR measurement through the clearance of inulin, 125I-iothalamate, or other radio-markers. Those methods are expensive, technically difficult, and not readily available. Thus, the most recent CLKT guidelines, published in 2012, still recommend the estimation of GFR through the MDRD formula, because it is the one that less frequently overestimates GFR.55
Knowing the limitations of SCr, the study by Schück et al., published in 2002, demonstrated a negative correlation between kidney function in LT recipients and CysC levels. The study included 58 LT recipients (mean 14 ± 10 months) in whom SCr, CysC, and creatinine clearance were determined, comparing them with inulin clearance as the gold standard. They found a correlation with a decrease in inulin clearance ≤ 80ml/min, with some serum CysC levels ≥ 1.2mg/dl, with 96% sensitivity. However, they did not demonstrate a clear superiority of that parameter, compared with SCr.56
In a more recent study, Wagner et al. evaluated the formulas based on SCr and CysC for calculating GFR in LT recipient patients, using inulin clearance as the reference. The formulas analyzed were the MDRD 4, Cockcroft-Gault, Hoek, Larsson, and the CKD-EPI based on SCr and CysC. They included 49 LT recipients (67% were men), in whom SCr, CysC, and inulin clearance were measured 24 months after transplantation. Bias, precision, and accuracy were determined for each formula, compared with inulin clearance. Of the study group, 45% had a mGFR below 60ml/min/1.73 m2 at 2 years, post-transplantation. In general, the equations based on CysC were superior to those based on SCr for identifying the patients with a GFR < 60ml/min/1.73 m2. The formulas that correctly identified a larger number of patients with CKD were the Hoek, Larsson, and CKD-EPI-CysC equations (84, 88, and 88%, respectively). The MDRD 4 and CKD-EPI-Cr formulas only identified 48 and 40% of those patients, respectively.57
ConclusionsThe development of CKD is one of the most frequent comorbidities found in patients that have undergone solid-organ transplantation. It is of the utmost importance to precisely know the kidney function of patients on the transplantation waiting list, so that preventive measures can immediately be taken, thus preserving their kidney function. During surgery, the necessary measures should also be carried out to prevent the development of AKI, one of the main risk factors for developing chronic kidney disease. Unfortunately, the different methods, formulas, and instruments that we have for its diagnosis, treatment, and follow-up are far from ideal. GFR calculation is imprecise in cirrhotic patients and in post-LT patients, given that the majority of the formulas were not described in those populations. However, knowing the trajectory of kidney function of those patients will enable us to establish long-term prevention and/or treatment strategies. We believe it is important that patients presenting with kidney dysfunction, or those at high risk for renal complications, have a nephrology assessment as part of their pre-transplantation evaluation. Likewise, the threshold for nephrology referrals should be lowered for transplanted patients at high risk for progressing to CKD, or patients that present with adverse renal outcomes.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Financial disclosureNo financial support was received in relation to this article.
Conflict of interestThe authors declare that there is no conflict of interest.
Please cite this article as: Meraz-Muñoz A, García-Juárez I. Enfermedad renal crónica en el trasplante hepático: evaluación de la función renal. Revista de Gastroenterología de México. 2019;84:57–68.