Over the last few decades, multidisciplinary treatment of cancer patients has improved survival rates and the disease-free periods. However, the number of tumors associated with treatment, particularly radiotherapy, has also increased.1
That association has been made from the cases of persons that survived the nuclear disasters of Japan and Chernobyl. Its justification is based on the concept of oncogenesis due to radiation, established by Cahan et al. in 1948,2 and those criteria are still valid. Their only controversial point is the time necessary for cancer to be associated with radiotherapy. The original criteria stated 5 years, but it has currently been shown that the period can be shorter, with the appearance of radiation-induced tumors from 6 months to 20 years after treatment completion.3
It is known that the development of these tumors is the result of the interaction of many etiologic factors. The role of mutations has been studied in animal models, especially that of p53, the main form of which is the deletion of that gene (52% of mutations) in radiation-induced sarcomas, compared with sporadic sarcomas (less than 10%). However, this is not yet conclusive in humans.4,5 The p53 mutations, as well as genetic alterations due to genomic instability, are the most widely studied. Their mechanisms include abnormal intracellular signaling, cytokine production, the creation of free radicals, and the secondary triggering of an inflammatory response.6
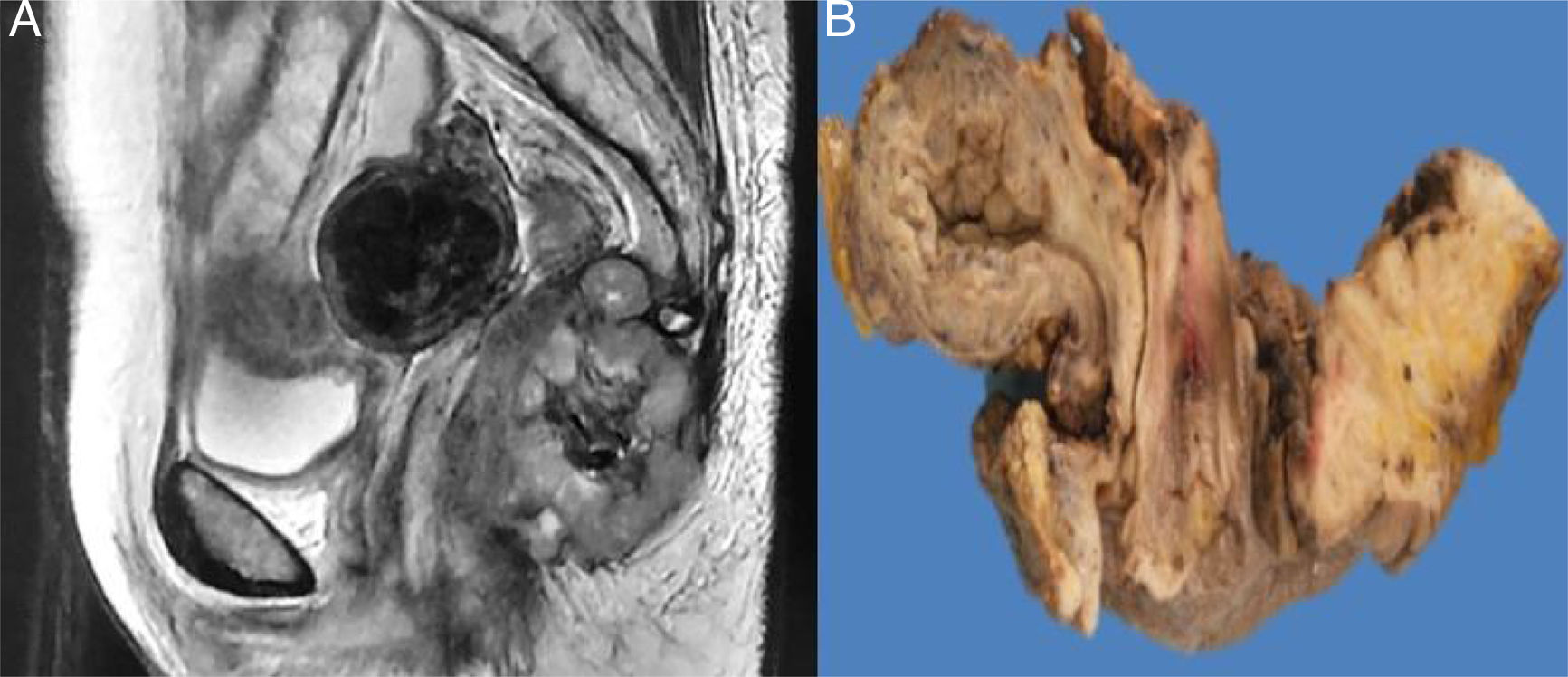
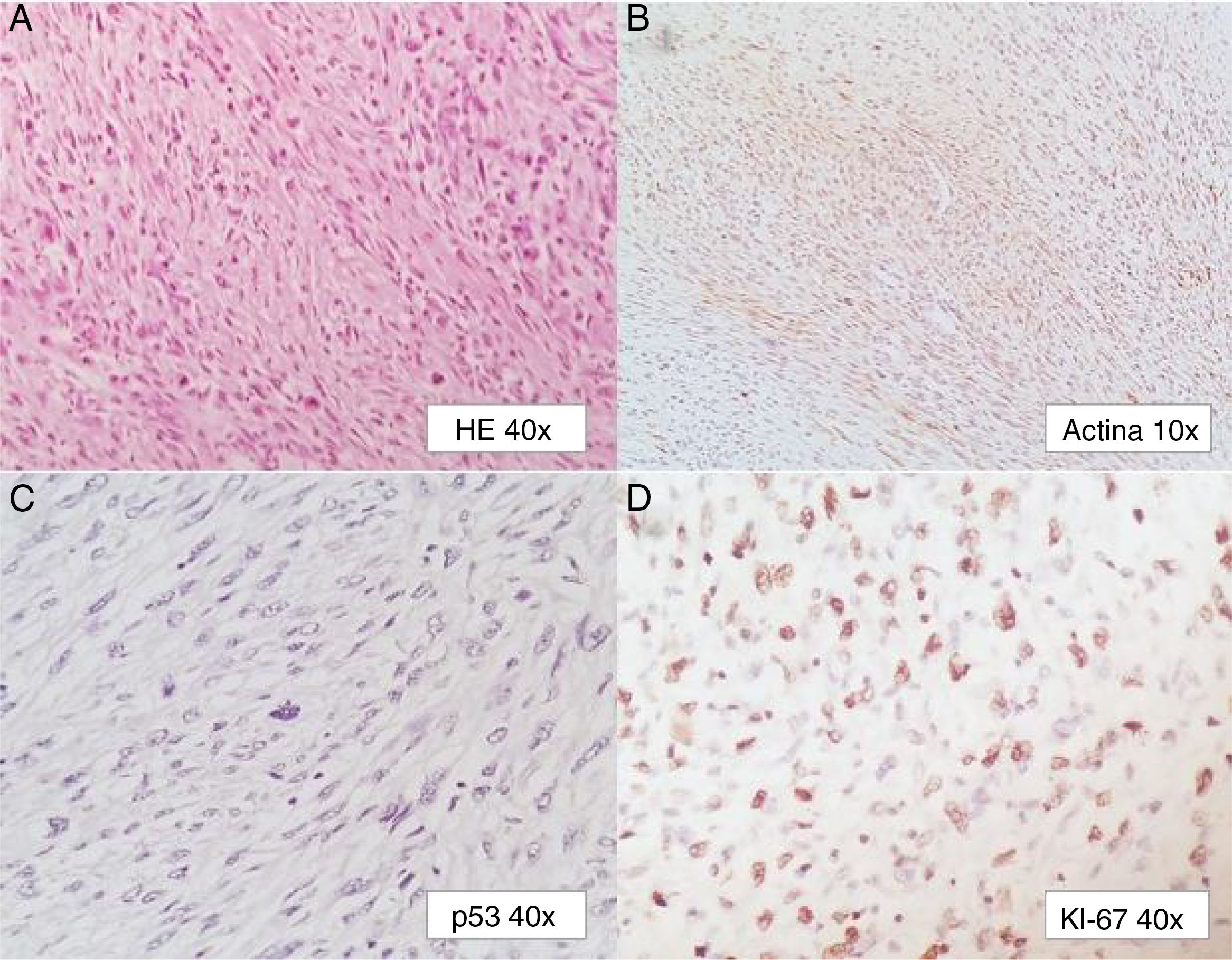
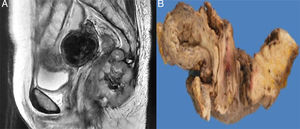
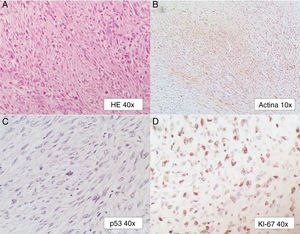
A 58-year-old woman had a past medical history of clinical stage IIB (FIGO) cervical cancer (squamous cell carcinoma), treated with chemotherapy (CT) based on 4 cycles of cisplatin and concomitant radiotherapy (RT) of 50Gy to the pelvis and brachytherapy (BT) of 35Gy. She had complete response and was kept under surveillance. Eight years later, with no apparent recurrence, the patient presented with rectal tenesmus associated with rectal bleeding. Physical examination revealed an ulcerated exophytic tumor 3cm from the anal margin that occluded 50% of the lumen, with no apparent involvement of the rectovaginal septum. Rectosigmoidoscopy identified the tumor at 6cm from the anal margin, occluding up to 70% of the lumen. Biopsy revealed mesenchymal neoplasia with the following immunoprofile: positive actin, negative CD34, CD117, DOG1, PS100, CKAE1/AE3, and P16. The diagnosis was high-grade leiomyosarcoma. Nuclear magnetic resonance (NMR) imaging corroborated the dependence on the rectal wall, as well as the involvement of the muscles of the perineum and the fat of both buttocks in the anal region (fig. 1). The rest of the extension studies were negative for distant disease. The multidisciplinary team began treating the patient with induction CT (doxorubicin and gemcitabine), after performing a sigmoid loop diversion colostomy. Response to the third CT cycle was partial and total pelvic exenteration with vulvectomy was indicated. Intraoperative findings were a tumor in the perineal region involving the gluteus maximus and levator ani muscles, with no separating plane of the uterus and bladder. Urinary reconstruction through ileal conduit and diversion colostomy was performed. The patient had adequate progression with no complications. Surgical specimen examination corroborated the differentiation to high-grade smooth muscle tumor. It measured 7 x 4 x 3cm, compromising the perineal edge up to the rectovaginal septum and infiltrating the anal sphincter muscles and skin. There was also lymphovascular and perineural invasion. The surgical margins were free from neoplasia and the immunohistochemistry pattern was similar to the previous one, adding positive P53 and Ki67 in 1% and 90% of the neoplastic cells, respectively (fig. 2).
Primary leiomyosarcomas of the rectum make up less than 0.1% of colorectal tumors. Not more than 290 cases are reported in the literature and there is still no solid association between rectal leiomyosarcoma and pelvic radiation.7
Due to their rareness, leiomyosarcomas, and radiation-induced sarcomas in general, are difficult to diagnose. They are more aggressive than non-radiation-induced sarcomas and are usually identified at advanced stages. Radiation-induced sarcomas are generally high-grade, thus having poor outcome. The rareness of the tumor and its broad presentation spectrum make high clinical suspicion necessary. Recurrence of the primary tumor with sarcomatoid morphology must be ruled out. This was corroborated in our patient due to the absence of immunohistochemical markers, such as p16 and CKAE1/AE3. Interestingly, despite the fact that there is a relatively frequent marked expression of p53, which is associated with a p53 mutation and worse outcome, its presence is not indispensable for this type of neoplasia. Surgery is the treatment of choice that offers the best oncologic results in these cases. It is also the only viable form of treatment, given that a very low response to systemic treatment, as well as the impossibility of reirradiation has been described.1,8
Financial disclosureNo financial support was received in relation to this study/article.
Conflict of interestThe authors declare that there is no conflict of interest.
Please cite this article as: Garcia-Ortega DY, Reyes-Garcia N, Martinez-Said H, Caro-Sanchez CHS, Cuellar-Hubbe M. Leiomiosarcoma de recto radio inducido posterior a tratamiento de cáncer cérvico uterino. Revista de Gastroenterología de México. 2018;83:465–467.