Three subtypes of achalasia have been defined through high-resolution esophageal manometry: subtype i shows no pressurization with swallows, subtype ii has increased isobaric panesophageal pressure, and subtype iii has distal esophageal spastic non-isobaric contractions. Studies describing the subtypes based on radiographic findings, clinical symptoms, and stasis scores are limited.
AimTo determine the differences in clinical symptoms, radiographic findings, and stasis scores for the 3 achalasia subtypes.
MethodsPatients undergoing high-resolution esophageal manometry received a questionnaire about current symptoms and previous treatments. The questions included the presence of symptoms and their severity. Barium swallow tests were performed before the high-resolution esophageal manometry study to evaluate the maximum esophageal diameter. Stasis scores were calculated using the transit patterns on high-resolution esophageal manometry.
ResultsOne hundred and eight patients with high-resolution esophageal manometry diagnosis of achalasia (n=8, subtype i; n=84, subtype ii; n=16, subtype iii) within the time frame of 1/2012-6/2015 were included in the study. Sex distribution was similar between the subtypes. Patient age was younger for subtype i (38±16 years), compared with subtypes ii (55±17 years) and iii (63±17 years) (P=.03). Esophageal symptoms did not differ between subtypes regarding the severity of nausea, chest pain, coughing, and heartburn, except for increased vomiting severity in subtype i (2.8±1.4 vs. 1.4±1.4 vs. 1.2±1.2, P<.01). A significant radiographic difference in esophageal dilation was seen between subtypes ii and iii (35.1±14.4 vs. 24.0±7.2mm, P=.023). Stasis scores did not significantly differ between the subtypes.
ConclusionsAchalasia subtypes had similar clinical symptoms, except for increased vomiting severity in subtype i. The maximum esophageal diameter in subtype ii was significantly greater than in subtype iii. Esophageal stasis scores were similar. Thus, high-resolution esophageal manometry remains essential in assessing achalasia subtypes.
Se han definido 3 subtipos de acalasia mediante manometría esofágica de alta resolución: el subtipo i no muestra presurización al deglutir, el subtipo ii presenta presión isobárica pan-esofágica incrementada y el subtipo iii muestra contracciones no isobáricas espásticas en el esófago distal. Los estudios que describen a los subtipos de acalasia basados en hallazgos radiográficos, síntomas clínicos y puntajes de estasis son limitados.
ObjetivoDeterminar las diferencias en los síntomas clínicos, hallazgos radiográficos, y puntajes de estasis para los 3 subtipos de acalasia.
MétodosLos pacientes sometidos a la manometría esofágica de alta resolución recibieron un cuestionario sobre síntomas actuales y tratamientos anteriores que preguntaba acerca de la presencia de síntomas y su severidad. Antes de la manometría esofágica de alta resolución se realizaron esofagogramas con contraste de bario para evaluar el diámetro esofágico máximo. Los puntajes de estasis se calcularon utilizando los patrones de tránsito en la manometría esofágica de alta resolución.
ResultadosSe incluyó en el estudio a 108 pacientes con diagnóstico de acalasia por medio de manometría esofágica de alta resolución (n=8, subtipo i; n=84, subtipo ii; n=16, subtipo iii) dentro del periodo 1/2012-6/2015. La distribución por sexos fue similar entre los subtipos. La edad de los pacientes fue menor para el subtipo i (38±16 años), comparado con los subtipos ii (55±17 años) y iii (63±17 años) (p=0.03). Los síntomas esofágicos entre los subtipos no difirieron en cuanto a severidad de nausea, dolor de pecho, tos y acidez, a excepción de la severidad incrementada del vómito en el subtipo i (2.8±1.4 vs. 1.4±1.4 vs. 1.2±1.2, p<0.01). Se observó una diferencia radiográfica significativa en la dilatación esofágica entre los subtipos ii y iii (35.1±14.4 vs. 24.0±7.2mm, p=0.023). Los puntajes de estasis no difirieron significativamente entre los subtipos.
ConclusionesLos subtipos de acalasia presentan síntomas clínicos similares, a excepción del incremento de la severidad del vómito en el subtipo i. El diámetro esofágico máximo en el subtipo ii fue significativamente mayor que en el subtipo iii. Los puntajes de estasis esofágica fueron similares. Por lo tanto, la manometría esofágica de alta resolución es esencial en la valoración de los subtipos de acalasia.
Achalasia is an esophageal motility disorder, originally described in 1674 by Sir Thomas Willis. He observed persistent “vomiting” in a patient whose treatment for the dilation of gastroesophageal junction (GEJ) obstruction involved a whale bone.1–3 The hallmark clinical symptom is dysphagia to solids and often to liquids.4 Additional symptoms include regurgitation and chest pain.5
Initial assessment of clinical symptoms is often carried out with barium esophagram, which can show aperistalsis, esophageal dilation, or the classic “bird's beak” appearance.5,6 Esophageal manometry has become the gold standard of diagnosis, with the classic findings of impaired lower esophageal sphincter (LES) relaxation and esophageal aperistalsis.5,6 Diagnosis of achalasia has evolved using high-resolution esophageal manometry (HREM), with the classification of 3 clinical subtypes.7–9 Subtype I shows no pressurization with swallows, subtype II has increased isobaric panesophageal pressure, and subtype III has distal esophageal spastic non-isobaric contractions.5–10 Recently, subtype II has shown more favorable treatment outcomes with pneumatic dilation or laparoscopic Heller myotomy, whereas subtype III has the least favorable outcome.11
Limited studies have been performed comparing the clinical characteristics of the achalasia HREM subtypes. Subtle differences have been seen that include older ages of the patients with subtype III and more severe clinical symptoms.12–14 Previous findings have suggested a different etiology for subtype III or even progression of subtype II disease to subtype I. These earlier studies have been limited by the small number of achalasia cases.
The aim of the present study was to determine the differences in clinical symptoms and radiographic findings for the 3 subtypes of achalasia. We wanted to determine whether the HREM subtypes could be differentiated based on symptoms, transit patterns, or radiographic findings.
Materials and methodsPatientsA total of 108 patients diagnosed with achalasia through HREM from January 2012 to June 2015 at Temple University Hospital were included in the study. Prior to undergoing HREM, each patient was given a questionnaire evaluating current clinical symptoms, medications, and treatments. Patients previously treated with pneumatic dilation, laparoscopic Heller myotomy, or peroral endoscopic myotomy (POEM) were excluded. The questionnaire was designed to identify the presence of symptoms and their severity based on a scale from 0 to 4, expressed in that order as none, mild, moderate, severe, and very severe. Dysphagia and regurgitation frequencies were based on a scale from 1 to 5, described in that order as rarely, several times a month, several times a week, at least once a day, and with every meal. Symptoms of vomiting were clearly defined and differentiated from regurgitation in the survey.
High resolution esophageal manometry protocolPatients were studied after an overnight fast of at least 8hours prior to the study. The patient was in the upright position and the HREM catheter was inserted via nasal intubation. The patient was then placed in the supine position. The catheter was advanced into the stomach and both the upper and lower esophageal sphincters were viewed (ManoScan™ HRM, Given Imaging, Yokneam, Israel). After waiting 5min for the catheter to equilibrate to the body temperature, a 30-s baseline recording was carried out to obtain the baseline GEJ pressure. Twelve wet swallows were performed 30seconds apart with 5 cc of normal saline at room temperature. Following adequate recording, the catheter was removed and the pressure profiles were analyzed (ManoView™, Given Imaging, Yokneam, Israel). LES pressure, upper esophageal sphincter (UES) pressure, and esophageal body contractions were determined. The studies were categorized according to the Chicago Classification for subtype determination.15
Stasis scoresStasis scores were calculated using transit patterns for each individual swallow on the color contour plot view of the high-resolution impedance manometry study. The length of the esophagus extending from the center point of the UES to that of the LES was divided into 3 equal segments (proximal, mid, and distal). Impedance was viewed at each esophageal segment and assigned a score (0 = no stasis, 4=very severe). The sum of these became the total score for each swallow (maximum of 12). Subsequently, the total score for all swallows was calculated and then divided by the number of swallows the patient completed, yielding a final “stasis score” for the entire study. Impedance manometry became available at our institution in 2013, so not all patients had this assessment.
Barium esophagramDuring the initial outpatient evaluation at Temple University Hospital, most patients underwent a barium swallow test. The tests were performed with the patient in the upright position and drinking a barium solution, followed by a series of x-rays or fluoroscopy. The quantity of barium solution used was dependent on achieving adequate images. Tolerance of the barium solution was variable, depending on the severity of the patient's condition. If dysphagia was the indication for the study, at least 5 single swallows were performed to evaluate motility. Once a diagnosis of achalasia subtype was confirmed, the maximum esophageal diameter was measured between the carina and the LES.
Statistical analysisData were recorded on Microsoft Excel and transferred to the SPSS version 22 (IBM Corporation, Somers, NY, USA) program. Results were expressed as percentages or mean ± standard deviation. The Pearson's chi-square test and Fisher's exact test were used for the categorical variables. The Spearman's rank-order correlation was employed for the nonparametric data to determine an association between 2 ranked variables. The Kruskal-Wallis test was used for the nonparametric data of 3 groups of an independent variable on ordinal dependent variables. One-way analysis of variance was performed on the data that had normal distribution. P values less than 0.05 were considered statistically significant.
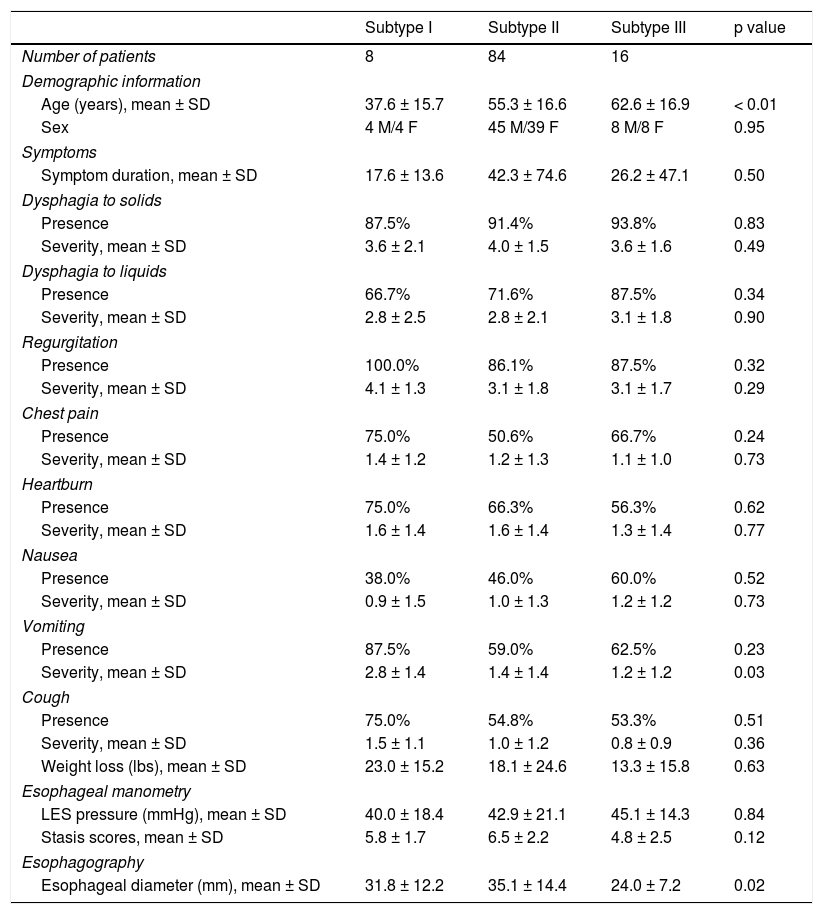
ResultsPatientsA total of 108 patients diagnosed with achalasia through HREM (n=8, subtype I; n=84, subtype II; n=16, subtype III) within the time frame of 1/2012-6/2015 were included in the study. Patients with subtype I were younger (38 ± 16 years), compared with those with subtypes II (55 ± 17 years) (p=0.02) and III (63 ± 17 years) (p<0.01), with statistically significant differences. Sex distribution between the subtypes was similar (p=NS). The mean duration of symptoms between the subtypes was 37.9 ± 68.3 months.
SymptomsThere was no significant difference in the presence of the esophageal symptoms of nausea, chest pain, heartburn, and coughing (Table 1). The severity of dysphagia with solids and liquids was similar across the subtypes (p=NS). The frequency of regurgitation, location of esophageal dysphagia, chest pain, and the presence/amount of weight loss did not significantly differ between the subtypes. In comparing vomiting severity, there was an increased severity in subtype I compared with subtypes II and III (2.8 ± 1.4 vs. 1.4 ± 1.4 vs. 1.2 ± 1.2, p=0.03). Insufficient data for the symptom of regurgitation did not allow proper analysis. There was no significant difference in the comparison of the duration of symptoms with the diagnosed subtype (17.6 ± 13.7 vs. 42.3 ± 74.6 vs. 37.9 ± 68.3, p=0.50).
Achalasia subtypes: demographics, symptoms, esophageal manometry and impedance, and esophagography.
| Subtype I | Subtype II | Subtype III | p value | |
|---|---|---|---|---|
| Number of patients | 8 | 84 | 16 | |
| Demographic information | ||||
| Age (years), mean ± SD | 37.6 ± 15.7 | 55.3 ± 16.6 | 62.6 ± 16.9 | < 0.01 |
| Sex | 4 M/4 F | 45 M/39 F | 8 M/8 F | 0.95 |
| Symptoms | ||||
| Symptom duration, mean ± SD | 17.6 ± 13.6 | 42.3 ± 74.6 | 26.2 ± 47.1 | 0.50 |
| Dysphagia to solids | ||||
| Presence | 87.5% | 91.4% | 93.8% | 0.83 |
| Severity, mean ± SD | 3.6 ± 2.1 | 4.0 ± 1.5 | 3.6 ± 1.6 | 0.49 |
| Dysphagia to liquids | ||||
| Presence | 66.7% | 71.6% | 87.5% | 0.34 |
| Severity, mean ± SD | 2.8 ± 2.5 | 2.8 ± 2.1 | 3.1 ± 1.8 | 0.90 |
| Regurgitation | ||||
| Presence | 100.0% | 86.1% | 87.5% | 0.32 |
| Severity, mean ± SD | 4.1 ± 1.3 | 3.1 ± 1.8 | 3.1 ± 1.7 | 0.29 |
| Chest pain | ||||
| Presence | 75.0% | 50.6% | 66.7% | 0.24 |
| Severity, mean ± SD | 1.4 ± 1.2 | 1.2 ± 1.3 | 1.1 ± 1.0 | 0.73 |
| Heartburn | ||||
| Presence | 75.0% | 66.3% | 56.3% | 0.62 |
| Severity, mean ± SD | 1.6 ± 1.4 | 1.6 ± 1.4 | 1.3 ± 1.4 | 0.77 |
| Nausea | ||||
| Presence | 38.0% | 46.0% | 60.0% | 0.52 |
| Severity, mean ± SD | 0.9 ± 1.5 | 1.0 ± 1.3 | 1.2 ± 1.2 | 0.73 |
| Vomiting | ||||
| Presence | 87.5% | 59.0% | 62.5% | 0.23 |
| Severity, mean ± SD | 2.8 ± 1.4 | 1.4 ± 1.4 | 1.2 ± 1.2 | 0.03 |
| Cough | ||||
| Presence | 75.0% | 54.8% | 53.3% | 0.51 |
| Severity, mean ± SD | 1.5 ± 1.1 | 1.0 ± 1.2 | 0.8 ± 0.9 | 0.36 |
| Weight loss (lbs), mean ± SD | 23.0 ± 15.2 | 18.1 ± 24.6 | 13.3 ± 15.8 | 0.63 |
| Esophageal manometry | ||||
| LES pressure (mmHg), mean ± SD | 40.0 ± 18.4 | 42.9 ± 21.1 | 45.1 ± 14.3 | 0.84 |
| Stasis scores, mean ± SD | 5.8 ± 1.7 | 6.5 ± 2.2 | 4.8 ± 2.5 | 0.12 |
| Esophagography | ||||
| Esophageal diameter (mm), mean ± SD | 31.8 ± 12.2 | 35.1 ± 14.4 | 24.0 ± 7.2 | 0.02 |
In the maximum esophageal diameter comparison (n=6, subtype I; n=63, subtype II; n=14, subtype III), there was a significant difference between subtype II and subtype III (35.1 ± 14.4 vs. 24.0 ± 7.2mm, p=0.023) and no difference with subtype I (31.8 ± 12.2mm, p=NS).
Esophageal stasisA total of 80 manometry studies were scored for the degree of esophageal stasis, given that 32 studies could not be scored due to a lack of impedance data. The stasis scores were similar between the subtypes (5.8 ± 1.7 vs. 6.5 ± 2.2 vs. 4.8 ± 2.5, p=NS). These values were comparable to our prior study showing normal values averaging 0.4.16 Stasis scores were compared with dysphagia to solids (r=0.095; p=0.416), dysphagia to liquids (r=0.137; p=0.248), and the severity of vomiting (r=0.081; p=0.486), with no significant relationships.
DiscussionThe present study of achalasia patients shows that the achalasia HREM subtypes had similar clinical symptoms, except for vomiting severity, which was higher in subtype I. The maximum esophageal diameter in subtype II was significantly higher than that of subtype III. Esophageal stasis scores were similar. Thus, HREM continues to be essential in assessing achalasia subtypes, given that clinical symptoms and radiographic findings do not reliably differentiate the 3 subtypes.
Achalasia is a relatively rare condition with no distinct pattern in relation to age and sex distribution. Age distribution has previously been observed to be bimodal or to have an increased risk as age increases, depending on the study.6,17 Previous studies did not differentiate between subtypes. Two recent retrospective studies were conducted that had a similar subtype comparison. A Saudi Arabian study included 32 patients diagnosed with achalasia. There was no significant difference between ages, and subtype I had the lowest age at 34.8 ± 12.2 years. In comparison, in a French study that included 169 patients, ages were similar in subtypes I and II, and patients with subtype III were significantly older.14 In the present study, subtype I patients were significantly younger than subtype II or III patients. A possible explanation for this is that subtype I might progress to subtype II, but the lack of contractility in subtype I suggests it is a later form of the disease. Symptoms, rather than age, could be the trigger for presentation. The variation in age should be considered in relation to the findings of the previous study, as well as to the small number of subtype I patients in the present study.
In the case series of achalasia patients presented herein, the predominant achalasia subtype was subtype II. This predominant subtype II cohort is similar to that observed in other studies, with some variation seen in the smaller studies.12–14 In a study conducted in India that compared pneumatic balloon dilatation and outcomes, subtypes II and III were equally prevalent. Subtype III was generally the least prevalent, with some studies showing the same for subtype I, which was consistent with the present study.12–14,18 A recent study by Rohof et al. included 176 patients with achalasia and utilized an Eckardt score equal to or lower than 3 to determine treatment success. The success rate with pneumatic dilation was significantly higher in subtype II, compared with the other subtypes, and laparoscopic Heller myotomy was considered the probable best treatment in subtype III.11 Prior to this finding, there was little variation in management regarding achalasia subtype determination and further consideration of findings was needed before diagnostic management.
Achalasia symptoms are typically present for years prior to diagnosis and treatment. The present study showed an average of 38 weeks of symptom presentation before diagnosis. Initial symptoms can be vague and the cardinal symptom is dysphagia, which can progress from solid to liquid dysphagia. Regurgitation that does not respond to adequate proton pump inhibitor treatment is another consistent finding.4 Other clinical symptoms include vomiting, cough, nausea, weight loss, and chest pain.5 The presence of symptoms was similar in the subtype comparison, except for increased vomiting severity in subtype I. Patients with achalasia can present with vomiting, which in retrospect, is occasionally regurgitation of undigested food products from the esophagus. However, the clinical differentiation between subtypes is difficult when there is limited symptom variation. The presence of symptoms was evaluated in other studies, but not their severity. In 2 previous studies, chest pain was more consistently present in subtype III, which was not observed in our study.11,12 In a recent study, a similar pattern of ganglion cell loss was described, but with a greater degree in subtype I than in subtype II, and there was speculation as to the progression from one subtype to another.19 In the present study, there was no significant difference when comparing the duration of symptoms with the diagnosed subtype.
Depending on patient symptoms, barium swallow testing may often precede HREM. A “bird's beak” appearance, aperistalsis, and esophageal dilation are radiographic findings suggestive of achalasia.5 The maximum esophageal dilation measured at the distal third segment of the esophagus was significantly wider in subtype II, compared with subtype III. However, no difference was seen when comparing subtypes II and III with subtype I. This is contrary to other studies that found subtype I significantly wider than subtype III.13 The images used were retrospective and many were not dedicated timed barium swallows, which was a limitation of our study.
Stasis score is a novel feature at our institution to help differentiate these subtypes. A recent study by Midani et al. included 44 subjects undergoing HREM with transit patterns evaluated through the color contour plot view. The stasis score assessment showed that 57% of the patients had normal transit. Other manometry findings included stasis scores of nutcracker esophagus (n=4), jackhammer esophagus (n=6), achalasia (n=2), and absent peristalsis (scleroderma, n=1). Severe stasis score patterns were seen in achalasia and absent peristalsis. The presence of stasis was observed in the evaluation of normal and abnormal manometry and these additional patterns were helpful in determining therapies. The present study utilized this methodology in differentiating the achalasia subtypes.
The average stasis score across all achalasia patients was 6.2 ± 2.3 out of a maximum score of 12. This reinforces previous studies at our center using the stasis score, which demonstrated that patients with achalasia had significantly higher stasis scores than patients with other findings of dysmotility on HREM. The patients with absent peristalsis, as seen in scleroderma, were the exception. This suggests that in achalasia there are still contractions propelling the bolus down to the distal esophagus, which is noticeably not present in absent peristalsis states. There were no significant differences in the stasis scores between the different subtypes. This is consistent with the aperistalsis state of each subtype.
The varying differences in presentation and diagnosis were minor. A weakness of the present study was the limited number of achalasia subtype I and III cases in the cohort. Barium swallow testing was a complementary study for evaluating the initial diagnosis. Regarding our study outcomes, management was unchanged, even after achalasia subtype diagnosis.
In conclusion, achalasia subtype II was the most common subtype found in the achalasia patient cohort of the present study. Clinical symptoms were largely similar, except for the increased severity of vomiting in subtype I. The maximum diameter of the esophagus was larger in subtype II than in subtype III. Overall, the clinical symptoms, radiographic findings, and stasis scores were insufficient for distinguishing the achalasia subtypes. Therefore, HREM continues to be an essential tool in assessing this motility disorder.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that no patient data appear in this article.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Conflict of interestThe authors declare that there is no conflict of interest.
Please cite this article as: Meillier A, Midani D, Caroline D, Saadi M, Parkman H, Schey R. Diferencia de subtipos de acalasia basados en síntomas clínicos, hallazgos radiográficos y puntajes de estasis. Revista de Gastroenterología de México. 2018;83:3–8.