Chronic diarrhea is one of the most challenging entities in gastroenterology. Since 2012, olmesartan has been recognized as a causal agent for chronic diarrhea that should be included in the differential diagnosis of patients with villous atrophy and negative celiac serologies1.
A 69-year-old woman was taken to the emergency room with a progressive decrease in her level of consciousness, together with lower limb edema. The patient's relatives stated that 6 weeks earlier she began to experience progressive, large-volume, watery stools (5-6 episodes/day), without blood or abdominal pain. Because of anorexia and weight loss (± 2kg), the patient was previously evaluated by her general practitioner and was empirically medicated with metronidazole and ciprofloxacin plus a probiotic, without improvement. A colonoscopy was also performed that identified no macroscopic abnormalities (no biopsies were done). Her past medical history of interest included high blood pressure treated with olmesartan and aliskiren and gastroesophageal reflux disease treated with pantoprazole. The patient had been taking these medications for approximately one year. There were no relevant epidemiologic contacts.
Upon physical examination, she had 8 points on the Glasgow Coma Scale and presented with tachypnea and hemodynamic instability. Her mucosae were dehydrated and her abdomen was not painful. Bilateral edema was more pronounced on the left side. Proctologic examination identified no abnormalities.
The initial blood test results were: Hb: 14.5g/l; WBC: 14.7x109/l; N: 87.9%; platelets: 213x109/l; Na: 153 mmol/l; K: 2.7 mmol/l; BUN: 77mg/dl; creatinine: 3.9mg/dl; CRP: 16mg/l; INR: 1.5; albumin: 2.5g/dl. Arterial blood gas showed metabolic acidosis (pH: 7.247; HCO3: 5.3 mEq/l).
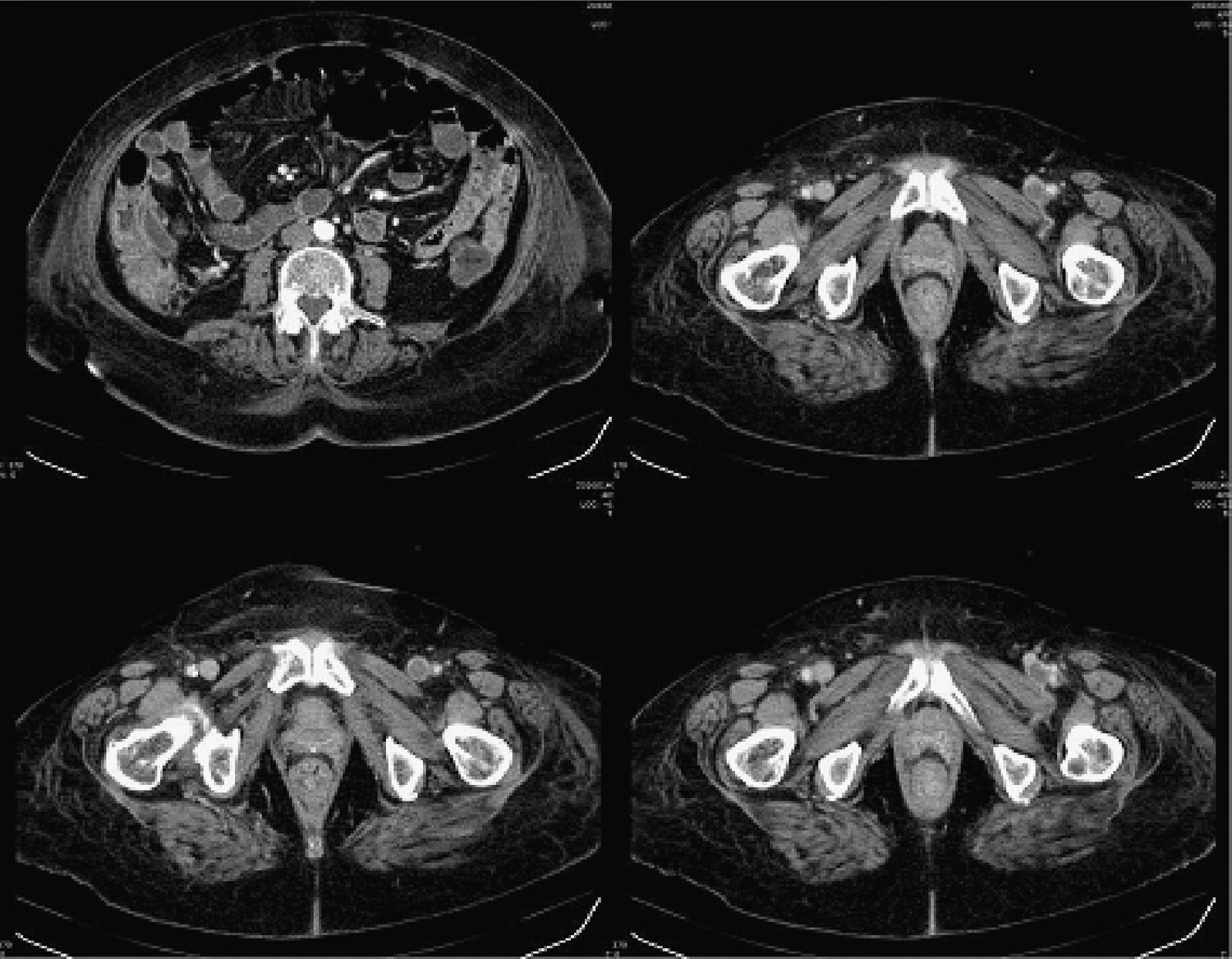
Subsequent ultrasound of the lower limb revealed extensive deep vein thrombosis (DVT) involving the femoral and left popliteal vein. Intravenous anticoagulation was begun.
Kidney function improved, and so a CT scan was performed (fig. 1) that confirmed the extension of the DVP. No other anomalies were observed. Given the characteristics of the diarrhea, an upper endoscopy was done, showing no macroscopic abnormalities (biopsies were taken).
Microbiologic evaluation of stools and Clostridium difficile toxin assay were negative. Subsequent blood tests reported iron deficiency (iron: 28 ug/dl; ferritin: 56 ng/ml; transferrin: 85mg/dl), a low folate value (2.0 ng/ml), and a total cholesterol level of 103mg/dl. HIV screening was also negative. Serum immunoglobulins, fibrinogen, aminotransferases, vitamin B12, TSH, FT4, calcium, magnesium, phosphorus, and anti-TG were normal.
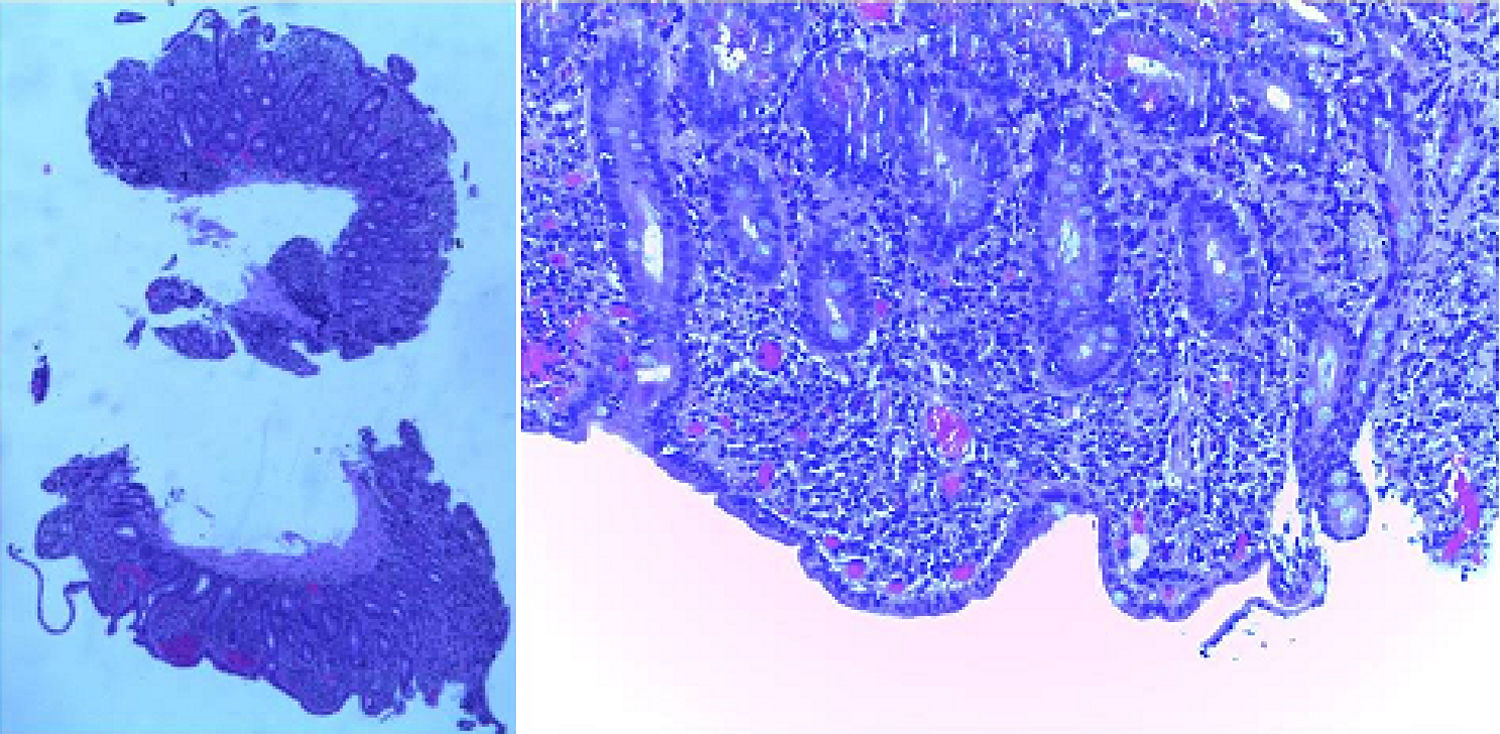
The histologic examination (fig. 2) showed subtotal villous atrophy with lymphocytic infiltration in the lamina propria. The prothrombotic factor study was negative.
The most common cause of villous atrophy is celiac disease. Nevertheless, when dealing with a negative celiac serology other diagnoses should be considered, such as Crohn's disease, enteric infections, collagenous/tropical sprue, common variable immunodeficiency, autoimmune enteropathy, hematologic malignancies, and drug-induced enteropathy2.
After ruling out other possible causes, we established the diagnosis of olmesartan-induced enteropathy.
Improvement was rapid with the suspension of olmesartan, and 9 days after admission, the patient had solid, shaped stools with one bowel movement a day. Olmesartan was substituted by hydrochlorothiazide and the patient was discharged with oral iron therapy, folic acid supplementation, and apixaban. Re-exposure to pantoprazole and aliskiren did not condition symptom reappearance.
Two months later, blood tests confirmed the patient's good progression. Subsequent endoscopy and colonoscopy showed no macroscopic or histologic abnormalities.
Olmesartan is an angiotensin II receptor blocker (ARB) used for treating high blood pressure since 20023. The first report of olmesartan-induced enteropathy was in 2012 and since then, a vast number of case reports and reviews have been published describing this adverse event1,3–7. Olmesartan-induced diarrhea pathogenesis is still unclear. However, the long interval between olmesartan therapy commencement and the development of symptoms suggests a cell-mediated immune mechanism8,9.
Whether it is a class effect is still doubtful, given that there are reports describing similar effects in patients taking other ARBs, even though those events appear to be less frequent10.
Its clinical spectrum is vast, but the majority of patients experience diarrhea, weight loss, and fatigue1,3–7. One interesting finding in our case was the DVT episode. DVT is a rare side effect of olmesartan, and to the best of our knowledge, there are no reports in the literature correlating olmesartan-induced enteropathy with DVT. The most plausible explanation could be the prothrombotic state secondary to dehydration and patient immobility.
Normocytic normochromic anemia and hypoalbuminemia are the most frequent laboratory abnormalities at presentation. Upper endoscopy may be normal, or show a nodular appearance of the duodenal mucosa. The most common histopathologic finding is flattening of the duodenal villous pattern (either total or partial) and increased duodenal intraepithelial lymphocytes (IELs). Contrary to celiac disease, flattening of the duodenal villous pattern is not always associated with IELs1,3–7.
Regarding treatment, literature reports have demonstrated that withdrawal of olmesartan conditioned clinical remission in all patients and histologic duodenal recovery in most of them1,3–7.
Our case is a typical example of the lack of awareness of this new entity by physicians. Because of the late diagnosis, the clinical course was continued and conditioned a catastrophic clinical set of events that included severe dehydration, hypovolemic shock, acute kidney insufficiency, metabolic acidosis, and DVT.
Conflict of interestThe authors declare that there is no conflict of interest.
Please cite this article as: Gião Antunes A, Vaz AM, Cadillá J, Peixe B, Guerreiro H. Diarrea crónica y trombosis venosa profunda durante el tratamiento con olmesartán. Revista de Gastroenterología de México. 2018;83:200–202.