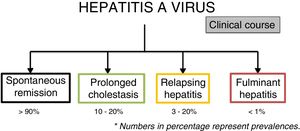
Acute hepatitis due to the hepatitis A virus usually has a short, benign and self-limited course, without causing chronic hepatitis. However, some cases have an atypical presentation, such as relapsing hepatitis, prolonged or persistent cholestasis, fulminant hepatic failure, or liver failure associated with autoimmune hepatitis. The typical clinical course of acute hepatitis A virus infection is spontaneous remission in 90% of the cases, but atypical cases have a prevalence that varies from less than 1 to 20%, depending on the manifestation (overall prevalence ∼7%). There is little information on the atypical clinical courses of hepatitis A virus infection and the lack of recognizing those presentations in clinical practice often results in carrying out numerous studies and treatments that not only are unnecessary, but can also be harmful. The aim of the present article was to describe 3 clinical cases of atypical hepatitis A infection and provide a literature review of such cases.
La hepatitis aguda por virus de hepatitis A usualmente sigue un curso corto, benigno y autolimitado sin ocasionar una hepatitis crónica, sin embargo en algunos casos puede manifestarse mediante formas atípicas como son hepatitis recurrente, colestasis prolongada o persistente, falla hepática fulminante o asociada a hepatitis autoinmune. El curso clínico típico de la infección aguda por el virus de hepatitis A es hacia una remisión espontánea en más del 90% de los casos, sin embargo los cursos atípicos presentan una prevalencia que varía de <1 – 20% según la manifestación (global ∼7%). Existe poca información sobre los cursos clínicos atípicos por la infección del virus de hepatitis A y es relevante mencionar que la falta de reconocimiento de estos, a menudo ocasiona en la práctica clínica la realización de múltiples estudios y tratamientos que además de innecesarios pueden resultar perjudiciales. El presente trabajo tiene como objetivo describir 3 casos clínicos representativos de la infección atípica por virus de hepatitis A y una revisión de la literatura sobre los mismos.
Hepatitis A virus (HAV) infection is generally self-limited and induces permanent active immunity. The clinical course of hepatitis A was described by MacCallum in the mid-twentieth century.1,2 HAV is a nonenveloped ribonucleic acid (RNA) that belongs to the order Picornavirales and the family Picornaviridae.3 The virus is transmitted through oral inoculation of contaminated feces through person-to-person contact or the ingestion of contaminated water and/or food. It has an incubation period that can last from 15 to 50 days. The clinical manifestations of hepatitis A depend on the age of the patient. Only 30% of children under 6 years of age present with nonspecific symptoms with no jaundice and 70% of adults present with nonspecific symptoms, as well as with specific symptoms, such as jaundice.4 The usual clinical course of hepatitis A virus infection begins with a pre-jaundice phase that lasts 5-7 days and is characterized by typical symptoms, such as general malaise, anorexia, nausea, vomiting, fever, abdominal pain, and headache and sometimes with atypical symptoms, such as chills, myalgias, arthralgias, cough, diarrhea, constipation, pruritus, and urticaria.5 The jaundice phase then appears and lasts 4-30 days and is characterized by choluria, acholia, and jaundice.6 The disease course is spontaneous remission in the large majority of cases, but 10-20% of symptomatic patients have an atypical disease course that manifests as relapsing hepatitis, persistent cholestasis, the development of autoimmune hepatitis, or fulminant liver failure. Hepatitis A virus has not yet been reported to progress to chronicity.4 There is little information on the atypical clinical course of HAV infection.
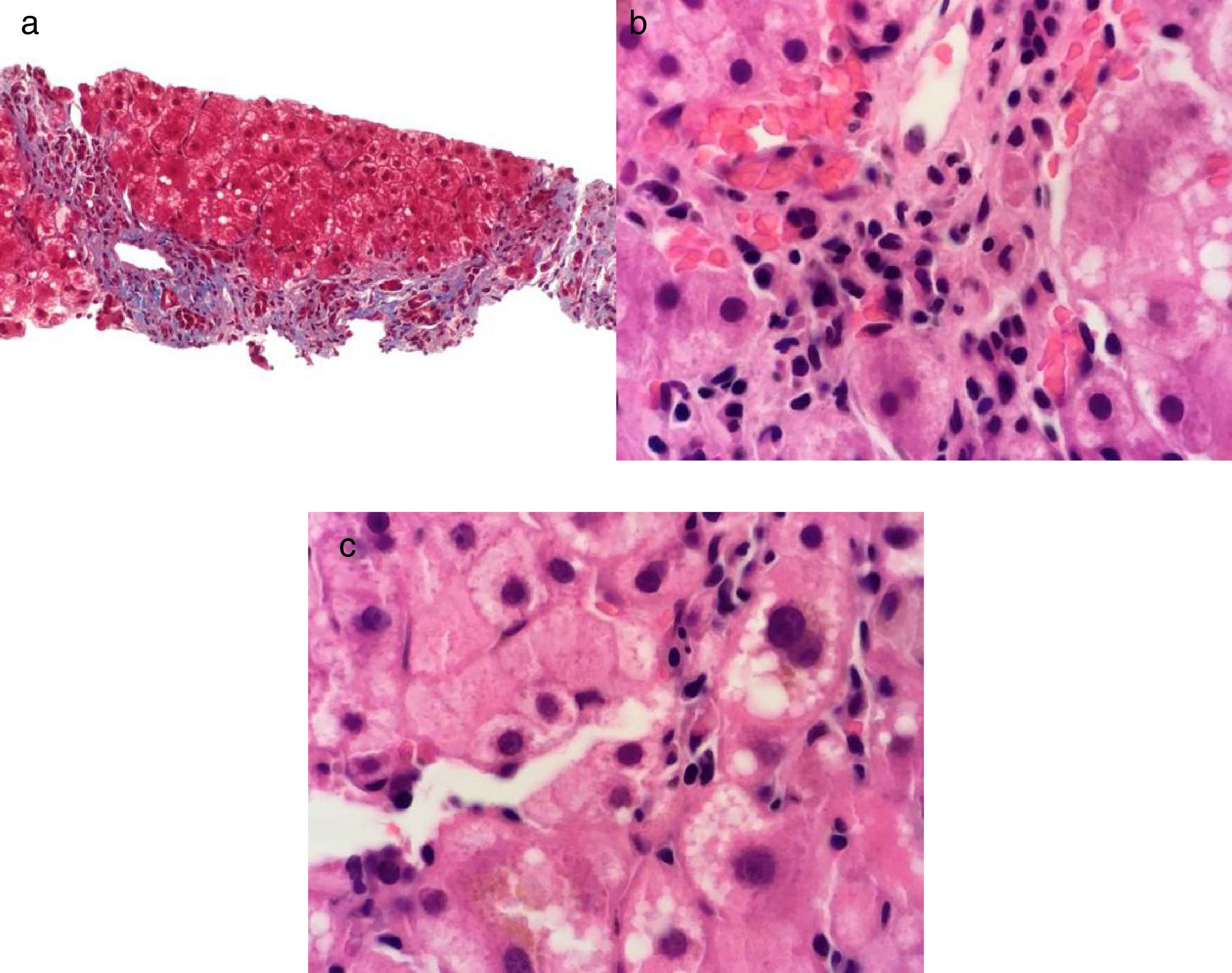
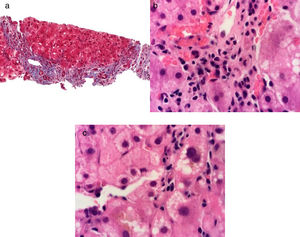
Clinical casesClinical case 1. Acute liver failure due to hepatitis A virus associated with autoimmune hepatitisA 47-year-old woman with a past medical history of asthma controlled with 200 ug of salbutamol as needed and a history of normal laboratory tests (including liver function tests) presented with the sudden onset of abdominal pain, diarrhea, nausea, and vomiting. She received three treatment courses based on antibiotics, with no symptom improvement. Seven days later, she presented with choluria, acholia, jaundice, hepatic encephalopathy, and slight fever. Her evaluation included liver function tests (LFTs) with the following results: total bilirubin (TB) 6.8mg/dl, direct bilirubin (DB) 3.8, ALT 6434 U/l, AST 4355 U/l, ALP 208 U/l, and INR, 3.1. The HAV IgM antibody test in the hepatitis viral panel was positive and a liver and biliary tract imaging study identified nonspecific inflammatory changes, with no other alterations. Hepatitis A and acute liver failure were diagnosed. Surveillance was begun with general support measures along with the IV administration of 10mg of vitamin K every 24h for 3 days. The patient then presented with spontaneous stabilization and general health status improvement, with resolution of coagulopathy and hepatic encephalopathy. The results of a subsequent evaluation 30 days later that included LFTs were: TB 13.8mg/dl; DB 8.0mg/dl; ALT 251 U/l; AST 149 U/l; ALP 194 U/l; and INR 1.3. The patient presented with positive antinuclear antibodies with a cytoplasmic pattern with a 1:80 titer, a thick mottled pattern with a 1:160 titer, an intermediate filament pattern with a 1:320 titer, positive anti-smooth muscle antibodies, and immunoglobulin G of 2628mg/dl. Liver biopsy revealed changes consistent with autoimmune hepatitis (fig. 1). Therefore, the diagnosis of autoimmune hepatitis triggered by HAV was made and treatment was begun with 50mg PO of azathioprine every 24h and 30mg PO of prednisone every 24h, with gradual dose reduction. At month 6 of follow-up, the patient had normal LFTs and she continued treatment with 50mg of azathioprine PO every 24h and 5mg of prednisone PO every 24h.
Masson stain at x40 shows grade III fibrosis due to the presence of portal-portal bridging fibrosis a). Hematoxylin & eosin stain at x100 shows interface hepatitis due to the presence of a lymphoplasmacytic inflammatory infiltrate overlapping the limiting plate b) and chronic lobular hepatitis due to the presence of a lymphoplasmacytic inflammatory infiltrate, hepatocyte vacuolation, and intra-cytoplasmic cholestasis in the hepatic lobule c).
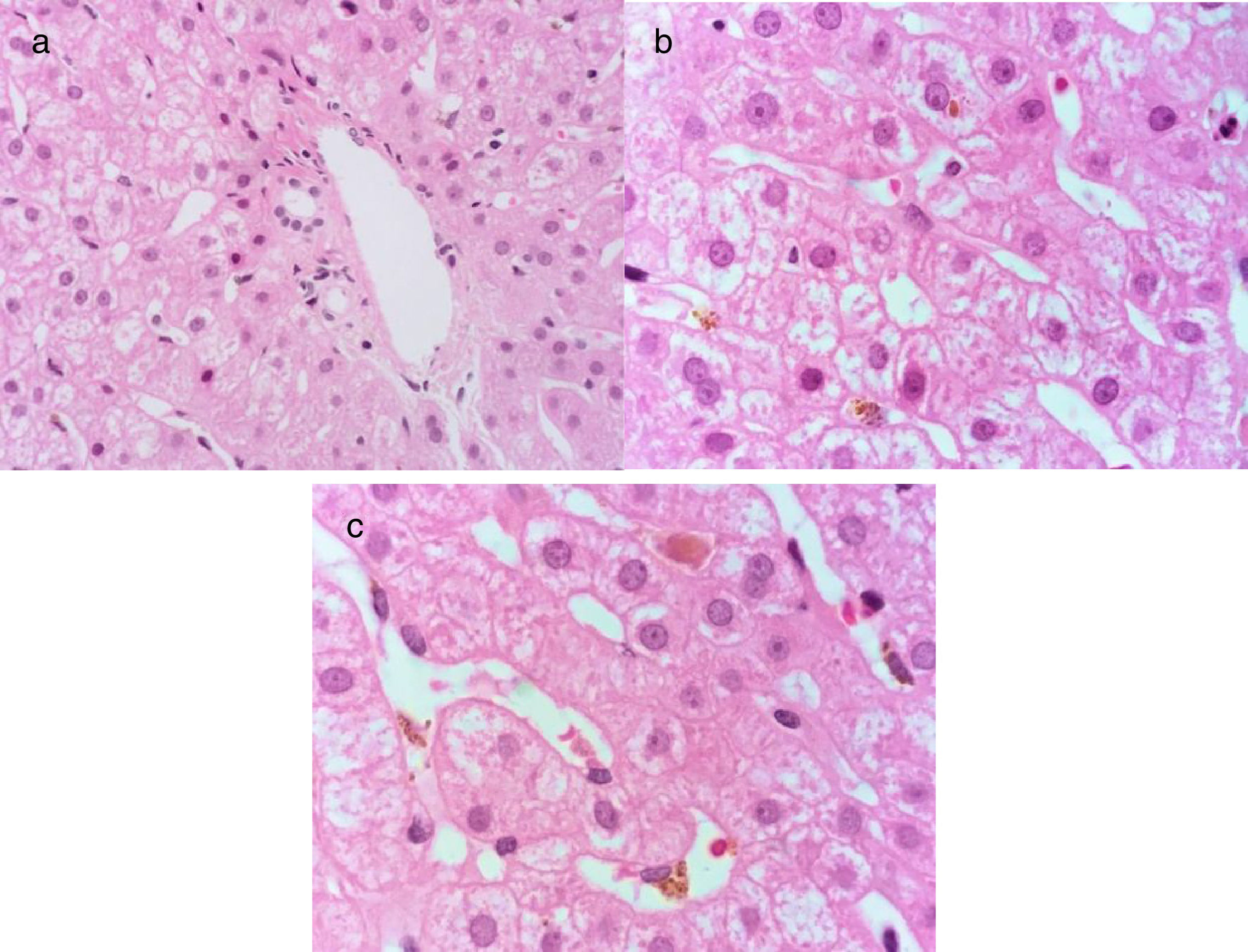
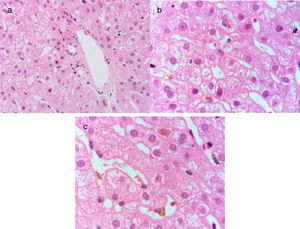
A 21-year-old man, previously in good health, presented with acute onset of nausea and vomiting. At 5 days, he presented with asthenia, adynamia, hyporexia, and jaundice. His evaluation included a positive HAV IgM antibody test in the hepatitis viral panel. Acute hepatitis due to HAV was diagnosed and symptom treatment was begun. At 30 days the patient still presented with jaundice and progressive elevation of TB. HAV with persistent cholestasis was suspected and so he was treated with 400mg of ribavirin PO every 8h and 250mg of ursodeoxycholic acid PO every 8h before his admission to our hospital. At 45 days he presented with symptoms of rhabdomyolysis with pigment-induced acute kidney failure, requiring hospitalization, and was admitted to our center. Follow-up laboratory results, including LFTs, were: TB 37mg/dl, DB 23mg/dl, ALT 63, AST 63, and ALP 116, hemoglobin 7.6g/dl, creatinine 1.7mg/dl, INR 1.0, positive HAV IgM antibody and HAV IgG antibody tests in the hepatitis viral panel, and negative results in the viral panel for HCV, HBV, cytomegalovirus, and Epstein-Barr virus. Liver and biliary tract imaging studies revealed hepatomegaly with no other alterations. Due to LFT alterations lasting more than 12 weeks, the patient was diagnosed with persistent cholestatic hepatitis A and treatment was begun with 40mg of prednisone PO every 24h. After 90 days his LFT results were: TB 3.8mg/dl, DB 1.9mg/dl, ALT 200 U/l, AST 99 U/l, and ALP 393 U/l. The prednisone dose was gradually reduced to 20mg every 24h PO. After 8 months his LFT results were: TB 0.78mg/dl, DB 0.18mg/dl, ALT 87 U/l, AST 42 U/l, and ALP 148 U/l. In the hepatitis viral panel, the HAV IgM antibody test was undetermined and the HAV IgG antibody test was positive. Liver biopsy was carried out and cholestasis with no inflammatory activity was observed (fig. 2).
Hematoxylin & eosin stain at x100 shows normal architecture of the portal area a). Mild cholestasis hepatopathy due to the presence of intracytoplasmic cholestasis and Kupffer cells with phagocytosed biliary pigment can be observed in the hepatic lobule b), as well as intracanalicular cholestasis c).
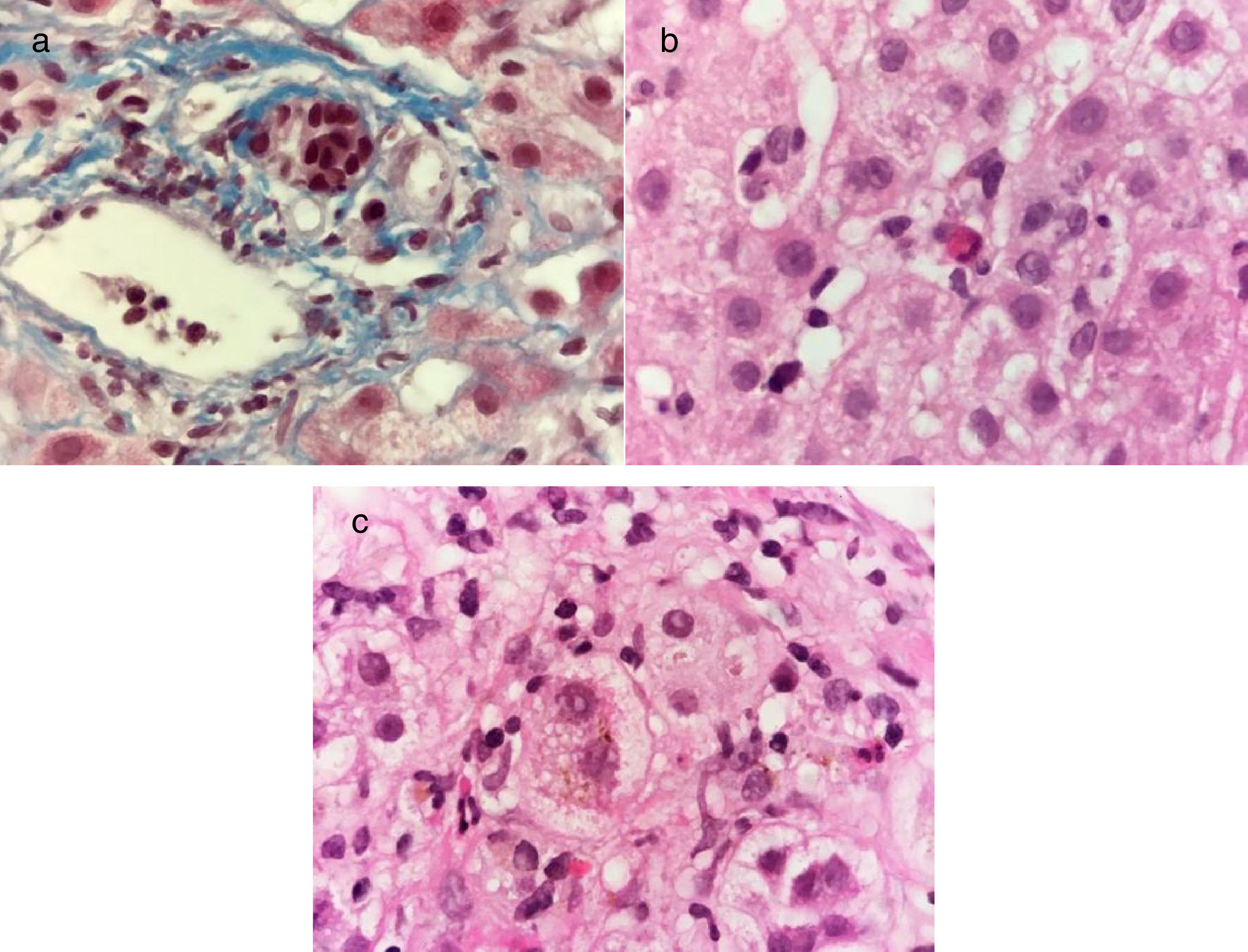
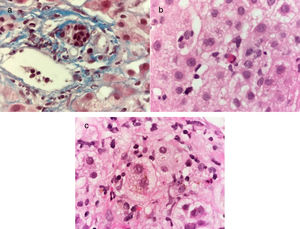
A 29-year-old man, previously in good health, had a remarkable family history of a brother that presented with hepatitis A one month before our patient presented with acute symptoms of general malaise, asthenia, and adynamia. At 5 days he had nausea, vomiting, choluria, acholia, and jaundice. He was diagnosed with acute hepatitis due to HAV, with positive HAV IgM antibodies, and treatment of symptoms was begun. At 30 days there was complete symptom resolution. Seven weeks after the initial symptoms, the patient again presented with general malaise, asthenia, adynamia, nausea, and vomiting, followed by choluria, acholia, and jaundice. Recurrent cholestatic hepatitis A was diagnosed and treatment of symptoms was begun again. Thirty days after the second onset of viral symptoms, the patient continued to have choluria, acholia, and jaundice. His evaluation included LFTs and the results were: TB 43mg/dl, DB 25mg/dl, ALT 1823 U/l, AST 1563 U/l, ALP 122 U/l, and INR 1.1. He had positive HAV IgM and HAV IgG antibodies in the hepatitis viral panel. Liver and biliary tract imaging studies identified hepatosplenomegaly, with no other alterations. Liver biopsy was performed that showed changes consistent with viral hepatitis (fig. 3). Recurrent cholestatic HAV infection diagnosis was confirmed and treatment of symptoms continued. Sixty days after the second presentation of viral symptoms, the patient's LFT results were: TB 18mg/dl, DB 9.5mg/dl, ALT 1116 U/l, AST 1171 U/l, and ALP 179 U/l. Therefore, his disease was considered persistent and recurrent cholestatic hepatitis A and treatment was begun with 40mg of prednisone every 24hours PO with gradual dose reduction. Six months after the second onset of viral symptoms, the patient had normal LFTs.
Masson stain at x100 shows a mononuclear inflammatory infiltrate in the portal area and interlobular bile duct damage characterized by nuclear polarity alteration, nuclear contour irregularity, and hyperchromasia a). Hematoxylin & eosin stain at x100 shows cholestatic chronic hepatitis with lobular activity due to the presence of scant mononuclear inflammatory cells and apoptotic hepatocytes b), as well as intracytoplasmic cholestasis and ballooning degeneration of the hepatocytes c).
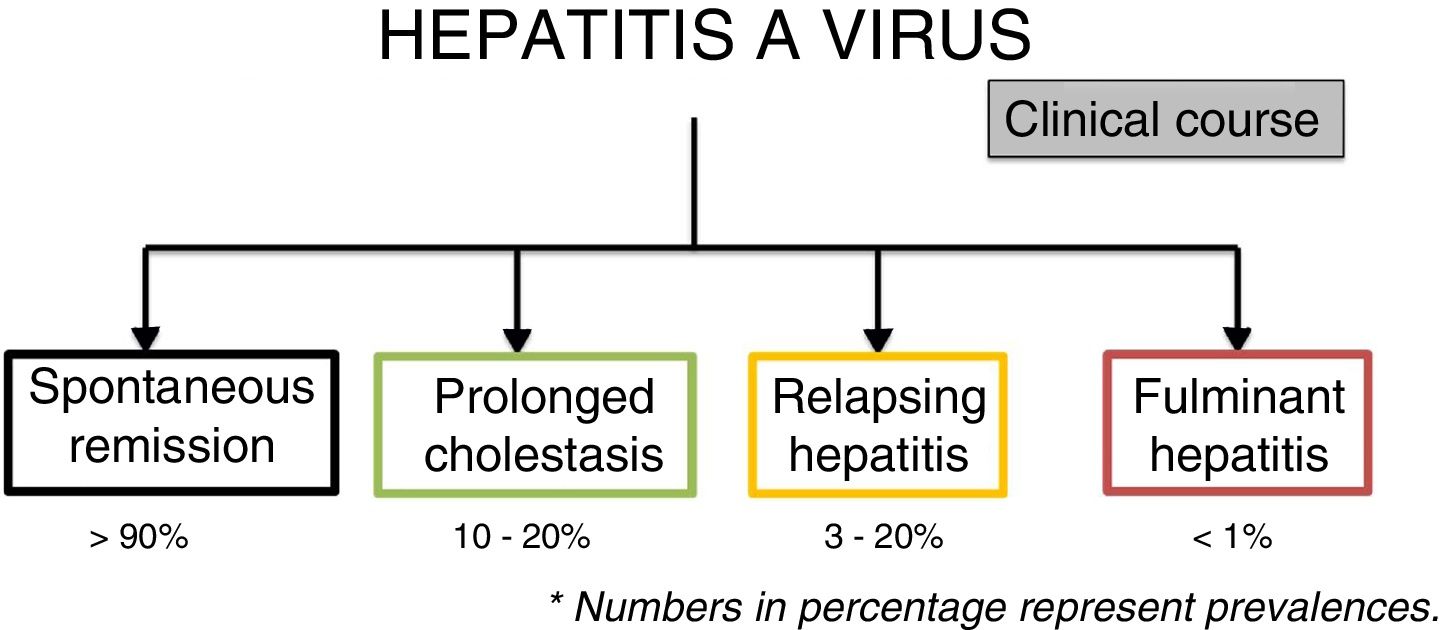
The usual or typical clinical course of acute hepatitis A virus infection is that of spontaneous remission in more than 90% of cases. However, there are atypical courses with a prevalence that varies from <1-20%, according to the manifestation4,7 (fig. 4).
Persistent cholestasisDefinitionCurrently there is no universal consensus for defining persistent cholestasis secondary to HAV infection and in the studies conducted up to now, it has been defined in different manners. However, historically, persistent cholestasis secondary to HAV infection is that in which the cholestasis, with TB > 10mg/dl and DB levels that are 50% above the TB levels, persists for more than 12 weeks from the initial infection.8–10
EpidemiologyPersistent cholestasis occurs in less than 5% of cases of HAV infection. Its associated factors are chronic hepatitis B virus coinfection, older age, and higher TB levels and prothrombin time.11
PathophysiologyThe mechanisms by which cholestasis persists in such cases is not clear. In lymphocytic cell cultures in animals infected with HAV, an increase in cellular and humoral immune activity has been described.12 In liver biopsies from HAV-infected patients, bile flow obstruction with central-lobular cholestasis, portal inflammation, periportal necrosis, and ductal damage have been observed, and they could be more severe in cases of persistent cholestasis.8,13 A study described the presence of pro-cholestatic polymorphisms of certain hepatocanalicular bile salt transporters, such as the c.3084 [GG] variant of the gene encoding the ABCB11 transporter and the c.711 [AT] variant of the gene encoding for phosphatidylcholine ABCB4, which predispose to a more severe intrahepatic cholestasis in patients infected with HAV, pointing to a probable genetic susceptibility in those patients.14
DiagnosisDiagnosis is clinical and confirmed by an episode of acute viral hepatitis infection due to HAV with positive HAV IgM antibodies and symptoms of jaundice or cholestasis that persist for more than 12 weeks, as presented in our case 2 patient. The most common clinical manifestations are jaundice, pruritus, fever, weight loss, diarrhea, and general malaise.8,9,15 In the LFTs, elevated alkaline phosphatase and TB levels predominate and reach their maximum peak after the 8th week (generally > 10mg/dl), in addition to a slight increase of transaminases and cholesterol.8 It is recommendable to carry out a liver and biliary tract ultrasound to rule out bile duct obstruction that could go unnoticed.8 During the period with cholestasis, the elimination of the virus in feces, as well as IgM antibody positivity, have been observed.9
TreatmentPersistent cholestasis is self-limited.8,9,16 Treatment can solely consist of explaining the favorable nature and clinical course of that disease variant, along with clinical surveillance and support. There are case series with contradictory results with respect to the use of glucocorticoids.
The use of prednisone with rapid tapering has been shown to accelerate symptom resolution of jaundice, pruritus, and general malaise, without presenting relevant adverse effects.8 Case reports have demonstrated that treatment with prednisone at doses of 1mg/kg or 40mg every 24h PO for 4 weeks, with a reduction over 12 weeks, results in significant symptom improvement and the cholestatic pattern of the liver function tests from the first week of treatment to total remission of symptoms and normalization of the LFTs at 6 months.7–17 However, glucocorticoid use has also been described to increase the risk for recurrence in patients that do not have seroconversion to IgG antibodies.9 The glucocorticoid mechanism of action in those cases is due to the stimulus of bile flow from alternate pathways, in addition to their anti-inflammatory effect.18 In our case 2 patient, prednisone management was decided upon because of the persistent cholestatic pattern associated with pigment-induced acute kidney failure to decrease the risk of greater kidney damage from elevated TB levels. Ribavirin was administered prior to the medical evaluation of our team. We do not know why it was used and ribavirin is not described as a therapeutic option in the literature.
OutcomePersistent cholestasis generally resolves spontaneously, with clinical and biochemical recovery at 6 months.9,16
Recurrent cholestasisDefinitionRelapsing hepatitis due to HAV infection is the name given to initial typical symptoms of acute hepatitis due to HAV with symptomatology resolution and biochemical parameter normalization associated with serum clearance of HAV documented through polymerase chain reaction (PCR) of HAV. That usually occurs on average in 3 to 15 weeks after symptom onset, with a later second episode with clinical and biochemical manifestations demonstrated by PCR viral replication of HAV in serum or feces.19
EpidemiologyCurrent epidemiologic data are derived from case reports or small case series in which diagnosis is not supported by evidence of PCR viral replication. Few studies document relapse through PCR viral HAV clearance, making it difficult to establish its precise prevalence. Those studies estimate prevalence at 1.5 to 20% of all HAV infections (case series of up to 200 patients).20–26 For example, in their Italian study on 66 patients with acute HAV infection, Sagnelli et al.27 showed that 5 patients (3.3%) presented with a recurrent disease course in which PCR-detected viremia was parallel to the clinical and biochemical course of the disease.
PathophysiologyThe most widely accepted pathophysiologic mechanism through which recurrence develops is that in which the HAV is not completely eliminated in the first phase of the hepatitis and thus is capable of having a new replication and causing a second disease episode. That could be explained in patients that have an immunity alteration with a decrease in the capacity to produce adequate antibody titers, essential for achieving complete serum clearance of the virus and reducing the liver damage it causes.19–21,23 Studies on patients infected with human immunodeficiency virus that present with acute HAV have shown that they have lower levels of transaminasemia, but higher HAV viral burdens and more prolonged viremia, compared with patients that do not have the human immunodeficiency virus. Such results mean that the activation of the immune system probably plays a role in the liver necrosis developed during acute hepatitis.27–31
A relatively new theory postulates that immunoglobulin A (IgA) is a hepatotropic transporter of HAV, speculating that said mechanism contributes to the different or atypical clinical outcomes of HAV infection.
That IgA transporter mechanism was studied in a mouse model in 2012 by Andreas Dotzauer et al.32 They showed that the HAV-IgA immunocomplexes efficiently reached the hepatocyte through an enterohepatic pathway mediated by reverse transcystosis via the polymeric immunoglobulin receptor (pIgR) across the intestinal epithelium.33 There could be continuous endogenous reinfection of the liver through that mechanism. The cycle mediated by IgA ends when serum immunoglobulin G (IgG) levels, present in advanced phases of infection, increase, given that they competitively replace the IgA in the HAV-IgA complexes. However, in cases of compromised immunity, that can contribute to lower IgG synthesis and therefore a prolonged illness or recurrent courses of HAV infection.32
DiagnosisDiagnosis is made in a patient that presents with a second episode of acute hepatitis due to HAV, clinically and biochemically manifested and corroborated through HAV PCR (serum or in feces). The patient has the history of a first episode with a later HAV PCR viral clearance and an asymptomatic period of approximately 4 to 10 weeks of duration.24,34
Patients that present with relapse should be considered potentially infectious.5
Immunoglobulin M (IgM) antibodies to HAV can remain detectable up to 3 to 4 months after the event and sometimes up to 12 months.22 The presence of those antibodies does not prove that the manifestations of recurrence are due to HAV.
In our case 3, the patient had persistent cholestatic-type progression of HAV infection, but during that progression he presented with elevated LFT results with transaminasemia. We decided upon liver biopsy, given that we do not have access to PCR for HAV detection in our environment. The biopsy revealed cytopathic changes from the virus attributed to HAV recurrence, since serology was negative for other hepatotropic viruses.
TreatmentManagement is through general measures and support, given there is no specific treatment.
Ursodeoxycholic acid has been used for treating cholestasis and pruritus, as well as the liver damage mediated by bile, thanks to its cytoprotective and immunomodulatory effects at the hepatocyte level. However, its use is not standardized and varies from case to case and pruritus is its most common indication.
Steroid use is controversial. At present there is no indication for the use of steroids in relapsing hepatitis due to HAV and it has been associated with a prolonged course of HAV infection.35 However, improvement of symptomatology with steroid use has been described in other case reports,19,23,36 but more studies are needed to establish their usefulness in that setting. The decision for steroid management in our case 3 patient was made based on the association with a persistent disease course, for which steroid use has been described, as mentioned above.
OutcomeThe recurrence of HAV infection is clinically and biochemically less intense than the initial episode and apparently does not alter the benign outcome of the disease. It sometimes manifests with symptoms of greater cholestasis, compared with the first episode.22,23
As in the typical disease course, behavior does not become chronic after a relapse and liver enzymes normalize approximately 6 months after infection in the majority of cases.37
Fulminant hepatitisDefinitionIt is defined as the presence of acute liver failure (ALF) secondary to HAV infection.38,39
EpidemiologyToday acute HAV infection is the most frequent cause of ALF in populations of developing countries, with reports of 3.1 to 26%.39–44
Mexican epidemiologic studies have reported HAV seroprevalence of up to 80%45 and HAV infection has been documented in some registers to be the cause of ALF in 81.4%.41,42 Although acute HAV infection is the most frequent viral cause of ALF, ALF itself is uncommon in the usual progression of HAV infection, presenting in approximately <1-3% of the cases of HAV infection.38,46–48
Studies have shown that the progression of HAV infection to ALF is more frequent in adults (above 50 years of age), compared with the pediatric population and is more frequent in patients with underlying chronic hepatopathy.40,46,49,50
PathophysiologyDevelopment of ALF due to HAV infection is most likely secondary to an exaggerated immune response and not to the cytopathic effect of the virus in the hepatocyte. That effect has already been observed in in vitro studies.51 That theory is suspected, given that some studies have shown that viral RNA is frequently undetectable in patients with fulminant ALF at the time of presentation, compared with patients that do not present with ALF. Higher viral RNA values are found in the latter (47 vs 16%, p<0.02). Likewise, patients that develop more severe disease present with faster viral clearance.52–54
DiagnosisALF is an abrupt deterioration of liver function, with the development of hepatic encephalopathy (or any alteration in the state of alertness) and coagulopathy, in less than 26 weeks from the onset of jaundice in a patient with no prior history of liver disease. Acute HAV infection is determined as the cause, through finding positive HAV IgM antibodies and characteristically the absence of another cause of ALF.55
TreatmentThe main treatment is medical support, but the only curative management in patients with support management failure and no evidence of spontaneous recovery is orthotopic liver transplantation (OLT). Because of its severity, management should always be carried out in a hospital and if possible in a monitoring unit (preferably in an intensive care unit).55
N-acetyl cysteine (NAC) has been used in ALF, thanks to its different properties. It facilitates the synthesis of depleted glutathione, improves oxygenation by being used as a substrate for microsomal glutathione transferase, and increases blood flow through the cyclase guanylate system by increasing nitric oxide and antioxidant and immunomodulatory action. In a study conducted on 12 pediatric patients with ALF due to HAV, progression after N-acetyl cysteine management was followed. NAC was administered at a daily dose of 10mg/kg, associated with treatment with lactulose, neomycin, and general measures.39 In that study the authors stated that NAC appeared to be an effective measure in ALF due to HAV, because it apparently modified the clinical course of the disease and prevented fatal outcomes. However, at present there is no strong evidence in relation to its use or how long it should be applied.
OLT is the management of choice in patients with ALF that do not present with spontaneous recovery.40
There are other treatment alternatives, such as with L-ornithine L-aspartate, prostaglandin E, molecular absorbent recirculating system, and plasmapheresis, none of which currently have strong recommendations for their use in cases of ALF.
OutcomeUp to 50-65% of patients have spontaneous recovery through support therapy management, but up to 50% of patients with ALF due to HAV could have unfavorable progression with the need for OLT, or if that is not an option, death.40,54 In their study, Taylor et al.40 observed spontaneous recovery in patients that fit OLT criteria of 55% and outcomes of OLT or death in 45% (OLT in 31% and death in 14%) during a 3-week follow-up. That was reproduced in other studies with spontaneous remission survival of 50-70%.38,40,43,56
The finding of adverse prognostic factors (kidney failure, respiratory failure, hemodynamic failure) are high mortality factors that indicate the need for OLT.57–62
There are studies in which rapid viral clearance apparently is associated with poor outcomes in ALF due to HAV, signifying greater activation of the immune system with greater cytopathic damage to the hepatocyte and thus greater difficulty and longer time for achieving liver regeneration.54
Autoimmune hepatitisDefinitionIn patients that, at the time of acute HAV infection diagnosis, have the concomitant or later diagnosis of autoimmune hepatitis (AIH), there is no criterion that defines the time within which AIH can be associated with HAV infection.63
EpidemiologyAutoimmune hepatitis occurs in 3% of the cases of HAV infection.16 At present, evidence of those cases is limited to isolated case reports.64
PathophysiologyAs occurs with other viral infections, HAV infection has been postulated to be a triggering factor of latent AIH in susceptible individuals. In an experimental study on patients that developed AIH after subclinical HAV infection, prior to the viral infection, the patients were observed to present with a defect in T-lymphocyte immunotolerance toward the asialoglycoprotein receptor in the cellular surface of the hepatocytes. It is considered the main antigen target in type 1 AIH. After infection the patients presented with specific T-lymphocyte CD4 activation, as well as an increase in antibody titers specific for the asialoglycoprotein receptor of the hepatocytes. In those cases, AIH manifested within 5 months after HAV infection.65 The possibility that more AIH cases have been preceded by HAV infection that are not detected because they presented with subclinical infection has also been postulated.
DiagnosisThe AIH diagnostic criteria can more practically be determined utilizing the simplified scoring system for AIH that requires a score ≥ 6 for probable diagnosis and a score ≥ 7 for a definitive diagnosis, with 88% sensitivity and 97% specificity.66
The diagnosis of AIH-associated HAV is made in the acute event. There are also patients that are diagnosed months after acute infection. Due to a lack of studies, there is no well-defined criterion for the time needed for that association. Our case 1 patient clearly presented with ALF due to HAV, given that she had positive IgM serology. Nevertheless, liver biopsy and AIH-specific antibodies were diagnostic for AIH. Here, the same question always arises: Did the patient have subclinical AIH with a minimum elevation of transaminases and no important symptoms and presented with acute HAV infection (which favored presentation with ALF), or was the AIH directly activated by the HAV infection? Advanced fibrosis in the liver biopsy would probably support the first hypothesis, but the doubt remains because the patient had normal LFTs before the event.
TreatmentIn the reported cases of AIH presenting after HAV infection, treatment corresponding to AIH has provided good results.65 Initial treatment with 0.5-1.0mg/kg of prednisone every 24h PO with gradual reduction, together with 1.5-2.0g of mycophenolate mofetil daily in fractioned doses, has recently been described to have response rates that are comparable to those of first-line treatment.67 The treatment aim is to achieve symptom remission, biochemical normalization of the liver function tests and IgG levels, and normalization of the characteristic histologic changes, so that liver damage progression and long-term mortality are reduced, whether or not the patients present with cirrhosis.68
OutcomeThere has been appropriate clinical and biochemical response at 6 months in patients that have documented autoimmune hepatitis concomitant with HAV infection and have received the corresponding immunosuppressant treatment.16
ConclusionsAtypical courses of hepatitis A virus infection have an overall prevalence of 7% that varies depending on disease manifestation.
The importance of vaccination campaigns should be emphasized to reduce the cases of severe HAV infection, always keeping in mind high-risk groups, such as patients with underlying chronic hepatopathy.
Some determinants for the development of atypical disease course that appear to have good association have been studied, but there are not yet enough studies on the topic to be able to make recommendations impacting the surveillance and management of those patients.
In cases of patients with HAV infection that present with a new increase in transaminases or bilirubin, other causes explaining the alteration in the laboratory studies must be ruled out (e.g. autoimmunity, other viral infections [such as hepatitis E virus], etc.) and the probability that the patient presents with an atypical disease course must always be considered to reduce the performance of unnecessary invasive studies, such as liver biopsy, which should be reserved for the cases that cannot be explained after a noninvasive approach.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that no patient data appear in this article.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Financial disclosureNo financial support was received in relation to this article.
Conflict of interestThe authors declare that there is no conflict of interest.
Please cite this article as: Muñoz-Martínez SG, Díaz-Hernández HA, Suárez-Flores D, Sánchez-Ávila JF, Gamboa-Domínguez A, García-Juárez I, et al. Manifestaciones atípicas de la infección por el virus de la hepatitis A. Revista de Gastroenterología de México. 2018;83:134–143.