The potential benefits of branched-chain amino acids (BCAAs) in cirrhosis extend beyond just the improvement of nutritional status. Their effects include improvement of glucose tolerance, oxidative stress, and inflammatory markers, as has been shown in several studies. A dual nutritional approach of a high-protein, high-fiber diet plus BCAAs in cirrhosis could have additional benefits, compared with BCAAs alone. Such an approach has not been explored and therefore the aim of the present study was to evaluate the effect of a combination of a high-protein, high-fiber diet plus BCAA supplementation over a 6-month period of time on the nutritional status of patients with cirrhosis, as well as its safety and tolerability for those same patients.
MethodsAn open, randomized clinical trial was conducted. Patients were randomized to one of two groups: the BCAAs+HPHF diet intervention group: a high-protein, high-fiber diet with 1.2g/kg protein and 30g of fiber plus supplementation with oral branched-chain amino acids 110g daily and the HPHF diet control group: a high-protein, high-fiber diet with 1.2g/kg protein and 30g of fiber. The differences between the treatment groups were compared using the unpaired T test and the differences at the end of treatment were compared using the paired T test.
ResultsA total of 72 patients were included, 37 in the intervention group and 35 in the control group. At the end of the study period, ammonia and glucose levels showed no significant increase in either group, reflecting the safety of the BCAA supplement. Furthermore, muscle and fat mass were evaluated through triceps skinfold thickness and mid-arm muscle circumference measurements. There was an increase in muscle mass and a decrease in fat mass in the BCAA group, but not in the control group. After the intervention, there were no significant changes in the Psychometric Hepatic Encephalopathy Score or the Critical Flicker Frequency score results in either group, and no episodes of hepatic encephalopathy were observed during the treatment period.
ConclusionSupplementation with branched-chain amino acids plus a high-fiber, high-protein diet is a safe intervention in patients with cirrhosis. It helps increase muscle mass and does not raise the levels of ammonia or glucose, nor is it associated with the development of hepatic encephalopathy.
Los beneficios potenciales de los aminoácidos de cadena ramificada (AACR) en la cirrosis se extienden más allá de solo la mejora del estado nutricional. Sus efectos incluyen la mejora de la tolerancia a la glucosa, estrés oxidativo y los marcadores inflamatorios, como se ha mostrado en varios estudios. Un abordaje nutricional dual de una dieta alta en proteína y fibra más AACR en la cirrosis podría tener beneficios adicionales, comparado con los AACR por sí solos. Tal abordaje no se ha explorado, y por lo tanto el objetivo del presente estudio fue evaluar el efecto de la combinación de una dieta alta en proteína y fibra con la suplementación de AACR durante un periodo de 6 meses sobre el estado nutricional de los pacientes con cirrosis, así como su seguridad y tolerabilidad en dichos pacientes.
MétodosSe realizó un ensayo clínico aleatorizado abierto. Los pacientes se aleatorizaron en uno de 2 grupos: el grupo de intervención con dieta de AACR+APAF: una dieta alta en proteína y fibra con 1.2g/kg de proteína y 30g de fibra más la suplementación con aminoácidos de cadena ramificada orales 110g diarios y el grupo de control con dieta APAF: una dieta alta en proteína y fibra con 1.2g/kg de proteína y 30g de fibra. Las diferencias entre los grupos de tratamiento se compararon utilizando la prueba «t» no pareada, y las diferencias al final del tratamiento se compararon utilizando la prueba «t» pareada.
ResultadosSe incluyó un total de 72 pacientes, 37 en el grupo de intervención y 35 en el grupo de control. Al final del periodo de estudio no se mostró un incremento significativo en los niveles de amonio y glucosa en ambos grupos, reflejando la seguridad del suplemento con AACR. Aún más, se evaluó la masa muscular y la grasa por medio de la medición del espesor del pliegue cutáneo del tríceps y de la circunferencia muscular en la parte media del brazo. En el grupo con AACR hubo un incremento en masa muscular y una disminución en masa grasa, pero no en el grupo de control. Después de la intervención, no hubo cambios significativos en los resultados de puntajes en el puntaje psicométrico de encefalopatía hepática o en la frecuencia crítica de fusión en cualquiera de los grupos, y no se observaron episodios de encefalopatía hepática durante el periodo de tratamiento.
ConclusiónLa suplementación con AACR más una dieta alta en proteína y fibra es una intervención segura en los pacientes con cirrosis. Ayuda al incremento de la masa muscular y no eleva los niveles de amonio o de glucosa, y tampoco se asocia con el desarrollo de encefalopatía hepática.
Malnutrition is highly prevalent in patients with cirrhosis and is seen in up to 40-90% of this population1. The relevance of this complication is the impact on the overall prognosis and other complications of cirrhosis, such as hepatic encephalopathy, ascites, and portal hypertension2,3. Different therapeutic approaches have been used to improve nutritional status in cirrhosis, including high-protein and high-fiber diets, supplementation with micronutrients, such as vitamins and minerals, and exercise4–8. An optimal nutritional approach in cirrhosis includes sufficient energy intake to surpass the daily requirements and overcome the catabolic state, and the intake of high-quality protein and micronutrients8,9.
Amino acid supply to the muscle in this population is impaired through different mechanisms, and to date, only branched-chain amino acids (BCAAs) either together or as individual components, have been proven to be effective as a nutritional supplement in cirrhosis10–13. The 3 main BCAAs are leucine, isoleucine, and valine, and have been used in different clinical trials on cirrhosis, showing an improvement in nutritional status, hepatic encephalopathy, general status, and quality of life, even in cirrhotic patients with hepatocellular carcinoma14–17.
The potential benefits of BCAAs in cirrhosis go beyond just the improvement of nutritional status. Their effects include improvement of glucose tolerance, oxidative stress, and inflammatory markers, as has been shown in several animal studies. Among the theoretic side effects that have been described are the increase in blood ammonia, nausea, and vomiting18,19.
On the other hand, fiber intake affords patients with multiple benefits, functioning as a prebiotic, thus promoting the growth of beneficial bacteria, limiting the growth of harmful bacteria and their production of harmful metabolites, and increasing bowel transit, which can be helpful in cirrhotic patients with hepatic encephalopathy19–21.
It appears that a dual nutritional approach of a high-protein, high-fiber diet plus BCAAs could provide additional benefits to patients with cirrhosis, compared with BCAAs alone. This approach has not been properly explored and therefore the aim of the present study was to observe the effect of the combination of a high-protein, high-fiber diet plus BCAA supplementation over a 6-month period of time on the nutritional status of patients with cirrhosis, as well as its safety and tolerability in those same patients.
Patients and methodsAn open, randomized clinical trial was conducted at a tertiary care center in Mexico City. The study was designed and carried out according to the principles of the Declaration of Helsinki and was approved by our institutional Ethics Committee (Ref. 584).
Study groupsPatients were randomized into one of two groups, in a 1:1 ratio, using simple randomization:
BCAAs + HPHF diet intervention group: high-protein, high-fiber (HPHF) diet with 1.2g/kg protein and 30g of fiber plus supplementation with oral branched-chain amino acids.
HPHF diet control group: high-protein, high-fiber diet with 1.2g/kg protein and 30g of fiber.
The BCAA supplement was provided in individual sachets of 110 grams. The contents of the BCAA supplement were 3.38 grams of L-leucine, 2.75 grams of L-isoleucine, and 2.5 grams of L-valine per 110 grams, totaling 500 Kcal (Enterex Hepatic, Victus Inc.). The patients allocated to the intervention group received 30 sachets every month for the daily consumption of 110g.
Selection criteriaInclusion criteriaWe included patients of both sexes, from 18 to 65 years of age, with an established diagnosis of cirrhosis based on biopsy and/or biochemical and radiologic parameters, with Child-Pugh stage A and B disease.
Exclusion criteriaWe excluded patients with active alcoholism in the previous 6 months, patients that had consumed any type of nutritional supplement in the previous 6 months, decompensated and/or hospitalized patients, patients with hepatocellular carcinoma, patients with TIPS, and patients with uncontrolled thyroid disease.
Elimination criteriaWe eliminated patients with poor adherence to the baseline diet (< 80%) or that did not attend the follow-up visits, and patients with incomplete data.
Evaluation and follow-upPatients that met the selection criteria and signed the statements of informed consent were randomized into the study groups and immediately afterwards were g instructed to follow a standardized diet of 1.2g/kg of protein, 30 grams of fiber and 60% carbohydrates for two weeks. Adherence to the standardized diet was evaluated after two weeks and the patients that had poor adherence were eliminated before the study was begun.
After the 2 weeks of the standardized diet, the patients in both groups received the individualized diet described above. The patients randomized to the intervention received 30 portions of the oral supplement containing 110g each, for daily consumption during one month. The patients then received the same portion each month for 6 months to complete the supplementation regimen.
Patients in both groups were evaluated monthly to assess compliance to diet and/or BCAA supplementation, detect possible side effects from the BCAAs, and evaluate the changes in the clinical, biochemical, and nutritional parameters.
Adherence evaluationTo evaluate adherence to the dietary intervention, food consumption frequency was obtained from each patient, and then compared with the quantities indicated in the dietary plan.
To evaluate adherence to the oral supplement, patients were asked to hand in the empty supplement sachets and to report any lack of compliance and/or side effects.
Statistical analysisThe descriptive variables are presented as mean ± standard deviation and absolute frequencies.
The differences among treatment groups were compared using the unpaired t test and the differences at the end of treatment were compared using the paired t test.
The analysis was carried out with the SPSS v. 21 (IBM, Armonk NY) program.
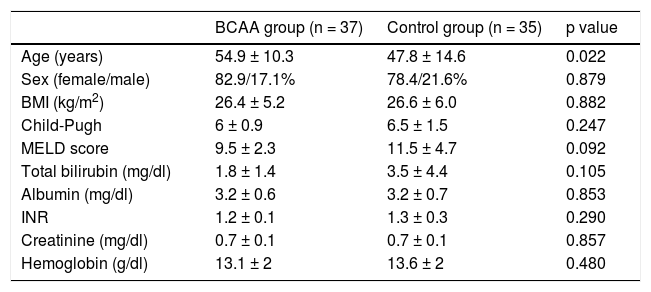
ResultsA total of 72 patients were included in the study and the descriptive characteristics of the population are shown in Table 1. Patients in the control group were younger and had higher Model for End-Stage Liver Disease scores, but no other significant differences were found between the s groups. The main etiologies were hepatitis C virus, primary biliary cirrhosis, and alcohol consumption.
Baseline characteristics of the population.
| BCAA group (n = 37) | Control group (n = 35) | p value | |
|---|---|---|---|
| Age (years) | 54.9 ± 10.3 | 47.8 ± 14.6 | 0.022 |
| Sex (female/male) | 82.9/17.1% | 78.4/21.6% | 0.879 |
| BMI (kg/m2) | 26.4 ± 5.2 | 26.6 ± 6.0 | 0.882 |
| Child-Pugh | 6 ± 0.9 | 6.5 ± 1.5 | 0.247 |
| MELD score | 9.5 ± 2.3 | 11.5 ± 4.7 | 0.092 |
| Total bilirubin (mg/dl) | 1.8 ± 1.4 | 3.5 ± 4.4 | 0.105 |
| Albumin (mg/dl) | 3.2 ± 0.6 | 3.2 ± 0.7 | 0.853 |
| INR | 1.2 ± 0.1 | 1.3 ± 0.3 | 0.290 |
| Creatinine (mg/dl) | 0.7 ± 0.1 | 0.7 ± 0.1 | 0.857 |
| Hemoglobin (g/dl) | 13.1 ± 2 | 13.6 ± 2 | 0.480 |
INR: International normalized ratio; MELD: Model for End-Stage Liver Disease.
Data presented as mean ± SD or absolute frequencies
Ammonia and glucose levels were evaluated at the end of the study period and there was no significant increase in either group, reflecting the safety of the BCAA supplement. Furthermore, muscle and fat masses were evaluated through triceps skinfold thickness and mid-arm muscle circumference (MAMC) measurements. The BCAA group showed a MAMC increase in muscle mass and a decrease in triceps skinfold thickness in fat mass. This finding was not observed in the control group (Table 2).
Changes in nutritional status and metabolic parameters.
| Baseline values in the BCAA group | Final values in the BCAA group | p value | |
|---|---|---|---|
| Triceps skinfold (mm) | 21.1 ± 12.2 | 19.6 ± 7.5 | 0.000 |
| MAMC (cm) | 28.7 ± 5.3 | 30.5 ± 4.6 | 0.000 |
| Ammonia (μg/dl) | 70 ± 46.6 | 73.8 ± 50.4 | 0.484 |
| Glucose (mg/dl) | 110.8 ± 52.9 | 112 ± 52 | 0.725 |
| Baseline values, control group | Final values, control group | p value | |
|---|---|---|---|
| Triceps skinfold (mm) | 20.7 ± 7.3 | 20.3 ± 6.9 | 0.923 |
| MAMC (cm) | 25.6 ± 6.1 | 25.9 ± 6.7 | 0.966 |
| Ammonia (μg/dl) | 65.8 ± 56.8 | 57.1 ± 35.4 | 0.385 |
| Glucose (mg/dl) | 104.3 ± 45.4 | 94.1 ± 17.4 | 0.500 |
MAMC: mid-arm muscle circumference.
Data presented as mean ± SD
Neuropsychometric tests were performed before and after the intervention to evaluate the potential development of covert hepatic encephalopathy. After the intervention, there were no significant changes in the Psychometric Hepatic Encephalopathy Score and Critical Flicker Frequency score results in either group (Table 3).
Changes in neuropsychometric tests.
| Baseline values, BCAA group | Final values, BCAA group | p value | |
|---|---|---|---|
| PHES score | −2 ± 2.8 | −2.2 ± 3.8 | 0.469 |
| CFF (Hz) | 35.5 ± 1.6 | 35.3 ± 1.4 | 0.423 |
| Baseline values, control group | Final values, control group | p value | |
|---|---|---|---|
| PHES score | −1.4 ± 2 | −1.3 ± 2.4 | 0.628 |
| CFF (Hz) | 40.8 ± 3.4 | 40.9 ± 4.2 | 0.738 |
PHES = Psychometrical Hepatic Encephalopathy Score / CFF= critical flicker frequency.
Data presented as mean ± SD.
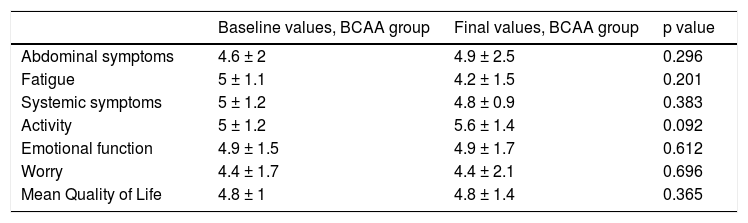
Finally, quality of life was evaluated using the chronic liver disease questionnaire (CLDQ). After the intervention period, the CLDQ score remained stable in the BCAA group and decreased in the control group (Table 4).
Changes in quality of life.
| Baseline values, BCAA group | Final values, BCAA group | p value | |
|---|---|---|---|
| Abdominal symptoms | 4.6 ± 2 | 4.9 ± 2.5 | 0.296 |
| Fatigue | 5 ± 1.1 | 4.2 ± 1.5 | 0.201 |
| Systemic symptoms | 5 ± 1.2 | 4.8 ± 0.9 | 0.383 |
| Activity | 5 ± 1.2 | 5.6 ± 1.4 | 0.092 |
| Emotional function | 4.9 ± 1.5 | 4.9 ± 1.7 | 0.612 |
| Worry | 4.4 ± 1.7 | 4.4 ± 2.1 | 0.696 |
| Mean Quality of Life | 4.8 ± 1 | 4.8 ± 1.4 | 0.365 |
| Baseline values, control group | Final values, control group | p value | |
|---|---|---|---|
| Abdominal symptoms | 4.3 ± 1.4 | 4.7 ± 1.5 | 0.264 |
| Fatigue | 4.3 ± 1.2 | 4.7 ± 1.4 | 0.133 |
| Systemic symptoms | 4.6 ± 0.9 | 4.8 ± 1.2 | 0.420 |
| Activity | 4.7 ± 1.3 | 4.8 ± 1.3 | 0.416 |
| Emotional function | 4.2 ± 1.1 | 4.6 ± 1.4 | 0.125 |
| Worry | 4.2 ± 1.2 | 4.7 ± 1.5 | 0.226 |
| Mean Quality of Life | 5.5 ± 6.6 | 4.7 ± 1.2 | 0.435 |
Data presented as mean ± SD.
Nutrition in liver diseases, especially in cirrhosis and hepatocellular carcinoma, has been associated with prognosis and the development of complications. Hence, a logical approach would include interventions aimed at improving nutritional status and, theoretically, survival.
One of the most widely studied nutritional approaches includes the use of BCAAs in cirrhosis, hepatocellular carcinoma, and hepatic encephalopathy. A main finding of the present study was that the changes in the anthropometric parameters were observed only in the BCAA group. They included an increase in muscle mass, determined through MAMC, and a decrease in fat mass, both of which could provide several benefits to the patients. Partial effects of the high-protein, high-fiber diet could be observed in the control group, in which there was no decrease in muscle mass according to the MAMC measurement. However, the clearest benefit was achieved only in the group treated with BCAAs. MAMC is one of the most widely used nutritional markers in cirrhosis, reflecting muscle depletion. An increase in MACM is directly related to an increase in muscle mass, which has a well-proven prognostic implication in cirrhosis. There were no differences in the nutritional parameter results when the analysis was stratified by etiology, an aspect that most certainly could not be assessed due to the small sample size.
For years there was reluctance to prescribe high-protein diets in cirrhosis, based on initial studies showing an increase in ammonia levels after protein intake in portal hypertension. In the present study, we did not see a significant increase in ammonia levels in either group and there were no episodes of covert or overt hepatic encephalopathy during the treatment period. This suggests that BCAA supplementation together with a HPHF diet can be used safely in patients with cirrhosis. Likewise, there were no changes in blood glucose levels after treatment in either group, reflecting the safety of BCAA supplementation. It did not have a deleterious effect on the metabolic parameters, even though baseline glucose levels in the BCAA group were higher than those of the control group.
Some studies have reported a benefit in quality of life when using BCAA supplements, but we found no important changes in the CLDQ scores. The results showed that quality of life remained unchanged after the treatment period in the BCAA group and seemed to decrease in the control groups. However, neither of these findings was statistically significant. This could be because the sample size was most likely too small to detect changes in those parameters.
The strengths of the present study were its methodological design, the use of a HFHP diet as the control, and the evaluation of different aspects of liver disease apart from nutritional status, such as neuropsychometric testing and ammonia quantification. Our study limitations included the facts that BCAA impact was not evaluated in different scenarios, such as hepatocellular carcinoma or as treatment for hepatic encephalopathy, and that the sample size was calculated only for the main outcome, which was nutritional status. This was probably not sufficient to show changes in the quality of life parameters.
ConclusionSupplementation with branched-chain amino acids plus a high-fiber, high-protein diet is a safe intervention in patients with cirrhosis. It helps increase muscle mass, does not increase the levels of ammonia or glucose, and is not associated with the development of hepatic encephalopathy.
Ethical responsibilitiesProtection of persons and animalsThe authors declare that the procedures followed conformed to the ethical standards of the responsible committee on human experimentation and were in accordance with the World Medical Association and the Declaration of Helsinki.
Data confidentialityThe authors declare that they have followed the protocols of their work center in relation to the publication of patient data.
Right to privacy and informed consentThe authors have obtained the informed consent of the patients and/or subjects referred to in the article. This document is in the possession of the corresponding author.
Conflict of interestThe authors declare that there is no conflict of interest.
The authors would like to thank Fabiola Sánchez Meza for her contribution in relation to data collection.
Please cite this article as: Ruiz-Margáin A, Macías-Rodríguez RU, Ríos-Torres SL, Román-Calleja BM, Méndez-Guerrero O, Rodríguez-Córdova P, et al. Efecto de una dieta rica en proteínas y alta en fibra más la suplementación con aminoácidos de cadena ramificada sobre el estado nutricional de pacientes con cirrosis. Revista de Gastroenterología de México. 2018;83:9–15.